Mu Transpososome and Recbcd Nuclease Collaborate in the Repair of Simple Mu Insertions
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Link Between Independent Acquisition of Intracellular Gamma-Endosymbionts and Concerted Evolution in Tremblaya Princeps
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Frontiers - Publisher Connector ORIGINAL RESEARCH published: 25 June 2015 doi: 10.3389/fmicb.2015.00642 The link between independent acquisition of intracellular gamma-endosymbionts and concerted evolution in Tremblaya princeps Sergio López-Madrigal 1, Amparo Latorre 1, 2, Andrés Moya 1, 2 and Rosario Gil 1* 1 Institut Cavanilles de Biodiversitat i Biologia Evolutiva, Universitat de València, València, Spain, 2 Área de Genómica y Salud de la Fundación para el Fomento de la Investigación Sanitaria y Biomédica de la Comunitat Valenciana (FISABIO) – Salud Pública, València, Spain Many insect species establish mutualistic symbiosis with intracellular bacteria that complement their unbalanced diets. The betaproteobacterium “Candidatus Tremblaya” maintains an ancient symbiosis with mealybugs (Hemiptera: Pseudococcidae), which are classified in subfamilies Phenacoccinae and Pseudococcinae. Most Phenacoccinae Edited by: mealybugs have “Candidatus Tremblaya phenacola” as their unique endosymbiont, while Joerg Graf, University of Connecticut, USA most Pseudococcinae mealybugs show a nested symbiosis (a bacterial symbiont placed Reviewed by: inside another one) where every “Candidatus Tremblaya princeps” cell harbors several John M. Chaston, cells of a gammaproteobacterium. Genomic characterization of the endosymbiotic Brigham Young University, USA consortium from Planococcus citri, composed by “Ca. Tremblaya princeps” and Huan Qiu, Rutgers University, USA “Candidatus Moranella endobia,” unveiled several atypical features of the former’s *Correspondence: genome, including the concerted evolution of paralogous loci. Its comparison with the Rosario Gil, genome of “Ca. Tremblaya phenacola” PAVE, single endosymbiont of Phenacoccus Institut Cavanilles de Biodiversitat i avenae, suggests that the atypical reductive evolution of “Ca. -

Characterization of the Multidrug Resistance Protein Expressed in Cell Clones Stably Transfected with the Mouse Mdrl Cdna1
[CANCER RESEARCH 49, 2729-2734, May 15, 1989] Characterization of the Multidrug Resistance Protein Expressed in Cell Clones Stably Transfected with the Mouse mdrl cDNA1 Erwin Schurr,2 Martine Raymond,3 John C. Bell,4 and Philippe Gros5 Department of Biochemistry, Faculty of Medicine, McGill University, Montreal, Canada, H3GIY6 ABSTRACT of the nucleotide and predicted amino acid sequences of this clone indicated that the encoded polypeptide was most likely a Structural features of the multidrug resistance protein encoded by the membrane glycoprotein, highly similar or identical to P-glyco mouse mdrl gene were studied in multidrug-resistant cell clones stably protein, which contained 12 putative transmembrane domains transfected with a biologically active cDNA clone. Independently derived and a cluster of /V-linked glycosylation sites near the amino transfectant cell clones, initially selected in Adriamycin, were shown to be cross-resistant to several drugs, including actinomycin D, amsacrine, terminus. It is formed by the internal duplication of a structural mitoxantrone, VP-16, and vinblastine but remained sensitive to cis- unit which encodes three transmembrane loops and a putative platinum, 5-fluorouracil, ¡trabinocytosine, and bleomycin. In drug-resist nucleotide binding fold (7). The duplicated segment is highly ant transfectants the mdrl gene product was greatly overexpressed as a homologous to the energy coupling subunit of bacterial trans polypeptide of apparent molecular weight 160,000-170,000. This protein port proteins particularly HlyB, a protein that participates in was present in membrane enriched fractions and could be metabolically the extracellular transport of haemolysin in Escherichia coli labeled with |3H]glucosamine, confirming that the transfected mdrl gene (14). -

DNA Unwinding Step-Size of E. Coli Recbcd Helicase Determined from Single Turnover Chemical Quenched-flow Kinetic Studies
B doi:10.1016/S0022-2836(02)01067-7 available online at http://www.idealibrary.com on w J. Mol. Biol. (2002) 324, 409–428 DNA Unwinding Step-size of E. coli RecBCD Helicase Determined from Single Turnover Chemical Quenched-flow Kinetic Studies Aaron L. Lucius1, Alessandro Vindigni1, Razmic Gregorian1 Janid A. Ali1, Andrew F. Taylor2, Gerald R. Smith2 and Timothy M. Lohman1* 1Department of Biochemistry The mechanism by which Escherichia coli RecBCD DNA helicase unwinds and Molecular Biophysics duplex DNA was examined in vitro using pre-steady-state chemical Washington University School quenched-flow kinetic methods. Single turnover DNA unwinding experi- of Medicine, 660 S. Euclid Ave. ments were performed by addition of ATP to RecBCD that was pre- Box 8231, St. Louis, MO 63110 bound to a series of DNA substrates containing duplex DNA regions USA ranging from 24 bp to 60 bp. In each case, the time-course for formation of completely unwound DNA displayed a distinct lag phase that 2Fred Hutchinson Cancer increased with duplex length, reflecting the transient formation of Research Center, 1100 Fairview partially unwound DNA intermediates during unwinding catalyzed by Ave. North, Seattle, WA 98109 RecBCD. Quantitative analysis of five independent sets of DNA unwind- USA ing time courses indicates that RecBCD unwinds duplex DNA in discrete steps, with an average unwinding “step-size”, m ¼ 3.9(^1.3) bp step21, ^ 21 with an average unwinding rate of kU ¼ 196( 77) steps s ^ 21 (mkU ¼ 790( 23) bp s ) at 25.0 8C (10 mM MgCl2, 30 mM NaCl (pH 7.0), 5% (v/v) glycerol). -

Capture of a Recombination Activating Sequence from Mammalian Cells
Gene Therapy (1999) 6, 1819–1825 1999 Stockton Press All rights reserved 0969-7128/99 $15.00 http://www.stockton-press.co.uk/gt Capture of a recombination activating sequence from mammalian cells P Olson and R Dornburg The Dorrance H Hamilton Laboratories, Center for Human Virology, Division of Infectious Diseases, Jefferson Medical College, Thomas Jefferson University, Jefferson Alumni Hall, 1020 Locust Street, Rm 329, Philadelphia, PA 19107, USA We have developed a genetic trap for identifying revealed a putative recombination signal (CCCACCC). sequences that promote homologous DNA recombination. When this heptamer or an abbreviated form (CCCACC) The trap employs a retroviral vector that normally disables were reinserted into the vector, they stimulated vector itself after one round of replication. Insertion of defined repair and other DNA rearrangements. Mutant forms of DNA sequences into the vector induced the repair of a 300 these oligomers (eg CCCAACC or CCWACWS) did not. base pair deletion, which restored its ability to replicate. Our data suggest that the recombination events occurred Tests of random sequence libraries made in the vector within 48 h after transfection. Keywords: recombination; retroviral vector; vector stability; gene conversion; gene therapy Introduction scripts are still made from the intact left LTR, but reverse transcription copies the deletion to both LTRs, disabling Recognition of cis-acting DNA signals occupies a central the daughter provirus.15–17 We found that the SIN vectors role in both site-specific and general recombination path- that could escape this programmed disablement did so 1–6 ways. Signals in site-specific pathways define the by recombinationally repairing the U3-deleted LTR. -

Thermodynamics of DNA Binding by DNA Polymerase I and Reca
Louisiana State University LSU Digital Commons LSU Doctoral Dissertations Graduate School 2014 Thermodynamics of DNA Binding by DNA Polymerase I and RecA Recombinase from Deinococcus radiodurans Jaycob Dalton Warfel Louisiana State University and Agricultural and Mechanical College Follow this and additional works at: https://digitalcommons.lsu.edu/gradschool_dissertations Recommended Citation Warfel, Jaycob Dalton, "Thermodynamics of DNA Binding by DNA Polymerase I and RecA Recombinase from Deinococcus radiodurans" (2014). LSU Doctoral Dissertations. 2382. https://digitalcommons.lsu.edu/gradschool_dissertations/2382 This Dissertation is brought to you for free and open access by the Graduate School at LSU Digital Commons. It has been accepted for inclusion in LSU Doctoral Dissertations by an authorized graduate school editor of LSU Digital Commons. For more information, please [email protected]. THERMODYNAMICS OF DNA BINDING BY DNA POLYMERASE I AND RECA RECOMBINASE FROM DEINOCOCCUS RADIODURANS A Dissertation Submitted to the Graduate Faculty of the Louisiana State University and Agricultural and Mechanical College in partial fulfillment of the requirements for the degree of Doctor of Philosophy in The Department of Biological Sciences by Jaycob Dalton Warfel B.S. Louisiana State University, 2006 May 2015 ACKNOWLEDGEMENTS I would like to express my utmost gratitude to the myriad of individuals who have lent their support during the time it has taken to complete this dissertation. First and foremost is due glory to God, The Father, The Son and The Holy Spirit, through whom all is accomplished. It is with extreme thankfulness for the blessings bestowed upon me, and with vast appreciation for the beauty of God’s creation that I have pursued a scientific education. -

Insights from the Reanalysis of High-Throughput Chemical Genomics Data For
bioRxiv preprint doi: https://doi.org/10.1101/2020.07.16.206243; this version posted July 16, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. 1 1 Title: Insights from the reanalysis of high-throughput chemical genomics data for 2 Escherichia coli K-12 3 Authors: Peter I-Fan Wu1, Curtis Ross1, Deborah A. Siegele2 and James C. Hu1,3 4 5 Affiliations: 6 1. Department of Biochemistry and Biophysics, Texas A&M University and Texas 7 Agrilife Research, College Station, TX 77843-2128 8 2. Department of Biology, Texas A&M University, College Station, TX 77843-3258 9 3. Deceased 10 11 Correspondence: [email protected] 12 13 14 Key words: phenotypic profiling, functional genomics, microbial genomics, biostatistics, 15 Escherichia coli, bacterial genetics 16 17 bioRxiv preprint doi: https://doi.org/10.1101/2020.07.16.206243; this version posted July 16, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. 2 18 ABSTRACT 19 Despite the demonstrated success of genome-wide genetic screens and chemical 20 genomics studies at predicting functions for genes of unknown function or predicting 21 new functions for well-characterized genes, their potential to provide insights into gene 22 function hasn't been fully explored. -

( 12 ) United States Patent
US010428349B2 (12 ) United States Patent ( 10 ) Patent No. : US 10 , 428 ,349 B2 DeRosa et al . (45 ) Date of Patent: Oct . 1 , 2019 ( 54 ) MULTIMERIC CODING NUCLEIC ACID C12N 2830 / 50 ; C12N 9 / 1018 ; A61K AND USES THEREOF 38 / 1816 ; A61K 38 /45 ; A61K 38/ 44 ; ( 71 ) Applicant: Translate Bio , Inc ., Lexington , MA A61K 38 / 177 ; A61K 48 /005 (US ) See application file for complete search history . (72 ) Inventors : Frank DeRosa , Lexington , MA (US ) ; Michael Heartlein , Lexington , MA (56 ) References Cited (US ) ; Daniel Crawford , Lexington , U . S . PATENT DOCUMENTS MA (US ) ; Shrirang Karve , Lexington , 5 , 705 , 385 A 1 / 1998 Bally et al. MA (US ) 5 ,976 , 567 A 11/ 1999 Wheeler ( 73 ) Assignee : Translate Bio , Inc ., Lexington , MA 5 , 981, 501 A 11/ 1999 Wheeler et al. 6 ,489 ,464 B1 12 /2002 Agrawal et al. (US ) 6 ,534 ,484 B13 / 2003 Wheeler et al. ( * ) Notice : Subject to any disclaimer , the term of this 6 , 815 ,432 B2 11/ 2004 Wheeler et al. patent is extended or adjusted under 35 7 , 422 , 902 B1 9 /2008 Wheeler et al . 7 , 745 ,651 B2 6 / 2010 Heyes et al . U . S . C . 154 ( b ) by 0 days. 7 , 799 , 565 B2 9 / 2010 MacLachlan et al. (21 ) Appl. No. : 16 / 280, 772 7 , 803 , 397 B2 9 / 2010 Heyes et al . 7 , 901, 708 B2 3 / 2011 MacLachlan et al. ( 22 ) Filed : Feb . 20 , 2019 8 , 101 ,741 B2 1 / 2012 MacLachlan et al . 8 , 188 , 263 B2 5 /2012 MacLachlan et al . (65 ) Prior Publication Data 8 , 236 , 943 B2 8 /2012 Lee et al . -

Supplementary Information
Supplementary information (a) (b) Figure S1. Resistant (a) and sensitive (b) gene scores plotted against subsystems involved in cell regulation. The small circles represent the individual hits and the large circles represent the mean of each subsystem. Each individual score signifies the mean of 12 trials – three biological and four technical. The p-value was calculated as a two-tailed t-test and significance was determined using the Benjamini-Hochberg procedure; false discovery rate was selected to be 0.1. Plots constructed using Pathway Tools, Omics Dashboard. Figure S2. Connectivity map displaying the predicted functional associations between the silver-resistant gene hits; disconnected gene hits not shown. The thicknesses of the lines indicate the degree of confidence prediction for the given interaction, based on fusion, co-occurrence, experimental and co-expression data. Figure produced using STRING (version 10.5) and a medium confidence score (approximate probability) of 0.4. Figure S3. Connectivity map displaying the predicted functional associations between the silver-sensitive gene hits; disconnected gene hits not shown. The thicknesses of the lines indicate the degree of confidence prediction for the given interaction, based on fusion, co-occurrence, experimental and co-expression data. Figure produced using STRING (version 10.5) and a medium confidence score (approximate probability) of 0.4. Figure S4. Metabolic overview of the pathways in Escherichia coli. The pathways involved in silver-resistance are coloured according to respective normalized score. Each individual score represents the mean of 12 trials – three biological and four technical. Amino acid – upward pointing triangle, carbohydrate – square, proteins – diamond, purines – vertical ellipse, cofactor – downward pointing triangle, tRNA – tee, and other – circle. -

Metabolic Complementarity and Genomics of the Dual Bacterial Symbiosis of Sharpshooters
UC Davis UC Davis Previously Published Works Title Metabolic complementarity and genomics of the dual bacterial symbiosis of sharpshooters Permalink https://escholarship.org/uc/item/3wf162dk Journal PLOS Biology, 4(6) ISSN 1544-9173 Authors Wu, Dongying Daugherty, Sean C Van Aken, Susan E et al. Publication Date 2006-06-01 Peer reviewed eScholarship.org Powered by the California Digital Library University of California PLoS BIOLOGY Metabolic Complementarity and Genomics of the Dual Bacterial Symbiosis of Sharpshooters Dongying Wu1, Sean C. Daugherty1, Susan E. Van Aken2, Grace H. Pai2, Kisha L. Watkins1, Hoda Khouri1, Luke J. Tallon1, Jennifer M. Zaborsky1, Helen E. Dunbar3, Phat L. Tran3, Nancy A. Moran3, Jonathan A. Eisen1*¤ 1 The Institute for Genomic Research, Rockville, Maryland, United States of America, 2 J. Craig Venter Institute, Joint Technology Center, Rockville, Maryland, United States of America, 3 Department of Ecology and Evolutionary Biology, University of Arizona, Tucson, Arizona, United States of America Mutualistic intracellular symbiosis between bacteria and insects is a widespread phenomenon that has contributed to the global success of insects. The symbionts, by provisioning nutrients lacking from diets, allow various insects to occupy or dominate ecological niches that might otherwise be unavailable. One such insect is the glassy-winged sharpshooter (Homalodisca coagulata), which feeds on xylem fluid, a diet exceptionally poor in organic nutrients. Phylogenetic studies based on rRNA have shown two types of bacterial symbionts to be coevolving with sharpshooters: the gamma-proteobacterium Baumannia cicadellinicola and the Bacteroidetes species Sulcia muelleri. We report here the sequencing and analysis of the 686,192–base pair genome of B. -

Intrinsic Sequence Specificity of the Cas1 Integrase Directs New
RESEARCH ARTICLE elifesciences.org Intrinsic sequence specificity of the Cas1 integrase directs new spacer acquisition Clare Rollie1, Stefanie Schneider2, Anna Sophie Brinkmann3, Edward L Bolt3, Malcolm F White1* 1Biomedical Sciences Research Complex, University of St Andrews, St Andrews, United Kingdom; 2Faculty of Medicine, Institute of Cell Biology, University of Duisburg-Essen, Essen, Germany; 3School of Life Sciences, Queen’s Medical Centre, University of Nottingham, Nottingham, United Kingdom Abstract The adaptive prokaryotic immune system CRISPR-Cas provides RNA-mediated protection from invading genetic elements. The fundamental basis of the system is the ability to capture small pieces of foreign DNA for incorporation into the genome at the CRISPR locus, a process known as Adaptation, which is dependent on the Cas1 and Cas2 proteins. We demonstrate that Cas1 catalyses an efficient trans-esterification reaction on branched DNA substrates, which represents the reverse- or disintegration reaction. Cas1 from both Escherichia coli and Sulfolobus solfataricus display sequence specific activity, with a clear preference for the nucleotides flanking the integration site at the leader-repeat 1 boundary of the CRISPR locus. Cas2 is not required for this activity and does not influence the specificity. This suggests that the inherent sequence specificity of Cas1 is a major determinant of the adaptation process. DOI: 10.7554/eLife.08716.001 Introduction The CRISPR-Cas system is an adaptive immune system present in many archaeal and bacterial species. *For correspondence: It provides immunity against viruses and other mobile genetic elements mediated through sequence [email protected] homology-directed detection and destruction of foreign nucleic acid species (reviewed in Sorek et al., 2013; Barrangou and Marraffini, 2014; van der Oost et al., 2014). -
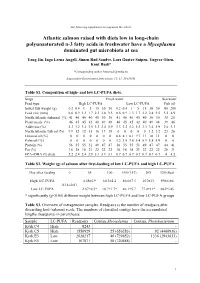
Q 297 Suppl USE
The following supplement accompanies the article Atlantic salmon raised with diets low in long-chain polyunsaturated n-3 fatty acids in freshwater have a Mycoplasma dominated gut microbiota at sea Yang Jin, Inga Leena Angell, Simen Rød Sandve, Lars Gustav Snipen, Yngvar Olsen, Knut Rudi* *Corresponding author: [email protected] Aquaculture Environment Interactions 11: 31–39 (2019) Table S1. Composition of high- and low LC-PUFA diets. Stage Fresh water Sea water Feed type High LC-PUFA Low LC-PUFA Fish oil Initial fish weight (g) 0.2 0.4 1 5 15 30 50 0.2 0.4 1 5 15 30 50 80 200 Feed size (mm) 0.6 0.9 1.3 1.7 2.2 2.8 3.5 0.6 0.9 1.3 1.7 2.2 2.8 3.5 3.5 4.9 North Atlantic fishmeal (%) 41 40 40 40 40 30 30 41 40 40 40 40 30 30 35 25 Plant meals (%) 46 45 45 42 40 49 48 46 45 45 42 40 49 48 39 46 Additives (%) 3.3 3.2 3.2 3.5 3.3 3.4 3.9 3.3 3.2 3.2 3.5 3.3 3.4 3.9 2.6 3.3 North Atlantic fish oil (%) 9.9 12 12 15 16 17 18 0 0 0 0 0 1.2 1.2 23 26 Linseed oil (%) 0 0 0 0 0 0 0 6.8 8.1 8.1 9.7 11 10 11 0 0 Palm oil (%) 0 0 0 0 0 0 0 3.2 3.8 3.8 5.4 5.9 5.8 5.9 0 0 Protein (%) 56 55 55 51 49 47 47 56 55 55 51 49 47 47 44 41 Fat (%) 16 18 18 21 22 22 22 16 18 18 21 22 22 22 28 31 EPA+DHA (% diet) 2.2 2.4 2.4 2.9 3.1 3.1 3.1 0.7 0.7 0.7 0.7 0.7 0.7 0.7 4 4.2 Table S2. -

Molecular Interactions Between Phage and the Catfish Pathogen Edwardsiella Ictaluri and Comparative Genomics of Epidemic Strains of Aeromonas Hydrophila
Molecular Interactions between phage and the catfish pathogen Edwardsiella ictaluri and Comparative Genomics of Epidemic strains of Aeromonas hydrophila by Mohammad Jahangir Hossain A dissertation submitted to the Graduate Faculty of Auburn University in partial fulfillment of the requirements for the Degree of Doctor of Philosophy Auburn, Alabama December 8, 2012 Keywords: Bacteriophages, Edwardsiella ictaluri, Epidemic Outbreak, Aeromonas hydrophila, Comparative Genomics, Channel Catfish Copyright 2012 by Mohammad Jahangir Hossain Approved by Mark R. Liles, Chair, Associate Professor of Biological Sciences Jeffery S. Terhune, Associate Professor of Fisheries and Allied Aquaculture Sharon R. Roberts, Associate Professor of Biological Sciences John F. Murphy, Professor of Plant Pathology Abstract Edwardsiella ictaluri causes Enteric Septicemia of Catfish (ESC) which is responsible for significant economic loss of the catfish aquaculture industry in the southeastern United States. A better diagnostic tool is required for the identification of Edw. ictaluri from ESC-affected catfish. Bacteriophages ΦeiAU and ΦeiDWF are specifically lytic to Edw. ictaluri strains and have tremendous potential to be used as diagnostic agents for the identification of Edw. ictaluri from ESC-affected catfish. However, bacteriophages ΦeiAU and ΦeiDWF demonstrate varying degree of lytic activity to different Edw. ictaluri strains. To identify the basis for variation and understand the mechanisms of phage-host interactions, phage resistant Edw. ictaluri mutants were generated by transposon mutagenesis of wild type Edw. ictaluri strains. Characterization of phage resistant Edw. ictaluri mutants revealed different host factors including integral membrane proteins, molecular chaperon, protein involved in LPS biosynthesis, proteins with proteolytic and regulatory activities, and proteins of unknown functions contribute to phage infection.