Evolutionary Divergence of the Wsp Signal Transduction System in Β- and Γ-Proteobacteria
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cultivation-Independent Analysis of Pseudomonas Species in Soil and in the Rhizosphere of field-Grown Verticillium Dahliae Host Plants
Blackwell Publishing LtdOxford, UKEMIEnvironmental Microbiology1462-2912© 2006 The Authors; Journal compilation © 2006 Society for Applied Microbiology and Blackwell Publishing Ltd200681221362149Original Article Pseudomonas diversity in the rhizosphereR. Costa, J. F. Salles, G. Berg and K. Smalla Environmental Microbiology (2006) 8(12), 2136–2149 doi:10.1111/j.1462-2920.2006.01096.x Cultivation-independent analysis of Pseudomonas species in soil and in the rhizosphere of field-grown Verticillium dahliae host plants Rodrigo Costa,1 Joana Falcão Salles,2 Gabriele Berg3 rescens lineage and showed closest similarity to and Kornelia Smalla1* culturable Pseudomonas known for displaying anti- 1Federal Biological Research Centre for Agriculture and fungal properties. This report provides a better under- Forestry (BBA), Messeweg 11/12, D-38104 standing of how different factors drive Pseudomonas Braunschweig, Germany. community structure and diversity in bulk and rhizo- 2UMR 5557 Ecologie Microbienne (CNRS – Université sphere soils. Lyon 1), USC 1193 INRA, bâtiment G. Mendel, 43 boulevard du 11 Novembre 1918, F-69622 Villeurbanne, Introduction France. 3Graz University of Technology, Institute of Environmental Verticillium dahliae causes wilt of a broad range of crop Biotechnology, Petersgasse 12, A-8010 Graz, Austria. plants and significant annual yield losses worldwide (Tja- mos et al., 2000). Control of V. dahliae in soil had been largely dependent on the application of methyl bromide in Summary the field. As this ozone-depleting soil fumigant has been Despite their importance for rhizosphere functioning, recently phased-out, the use of alternative, ecologically rhizobacterial Pseudomonas spp. have been mainly friendly practices to combat V. dahliae is a subject of studied in a cultivation-based manner. -

Étude Des Communautés Microbiennes Rhizosphériques De Ligneux Indigènes De Sols Anthropogéniques, Issus D’Effluents Industriels Cyril Zappelini
Étude des communautés microbiennes rhizosphériques de ligneux indigènes de sols anthropogéniques, issus d’effluents industriels Cyril Zappelini To cite this version: Cyril Zappelini. Étude des communautés microbiennes rhizosphériques de ligneux indigènes de sols anthropogéniques, issus d’effluents industriels. Sciences agricoles. Université Bourgogne Franche- Comté, 2018. Français. NNT : 2018UBFCD057. tel-01902775 HAL Id: tel-01902775 https://tel.archives-ouvertes.fr/tel-01902775 Submitted on 23 Oct 2018 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. UNIVERSITÉ DE BOURGOGNE FRANCHE-COMTÉ École doctorale Environnement-Santé Laboratoire Chrono-Environnement (UMR UFC/CNRS 6249) THÈSE Présentée en vue de l’obtention du titre de Docteur de l’Université Bourgogne Franche-Comté Spécialité « Sciences de la Vie et de l’Environnement » ÉTUDE DES COMMUNAUTES MICROBIENNES RHIZOSPHERIQUES DE LIGNEUX INDIGENES DE SOLS ANTHROPOGENIQUES, ISSUS D’EFFLUENTS INDUSTRIELS Présentée et soutenue publiquement par Cyril ZAPPELINI Le 3 juillet 2018, devant le jury composé de : Membres du jury : Vera SLAVEYKOVA (Professeure, Univ. de Genève) Rapporteure Bertrand AIGLE (Professeur, Univ. de Lorraine) Rapporteur & président du jury Céline ROOSE-AMSALEG (IGR, Univ. de Rennes) Examinatrice Karine JEZEQUEL (Maître de conférences, Univ. de Haute Alsace) Examinatrice Nicolas CAPELLI (Maître de conférences HDR, UBFC) Encadrant Christophe GUYEUX (Professeur, UBFC) Co-directeur de thèse Michel CHALOT (Professeur, UBFC) Directeur de thèse « En vérité, le chemin importe peu, la volonté d'arriver suffit à tout. -

Marta Dora Phd Thesis
MOLECULAR MONITORING OF MEAT SPOILING PSEUDOMONAS SPECIES AND ANALYSIS OF STAPHYLOCOCCAL ENTEROTOXIN EXPRESSION AND FORMATION Dóra Márta Doctoral Thesis Corvinus University of Budapest Faculty of Food Science Department of Microbiology and Biotechnology Budapest, 2011 ii According to the Doctoral Council of Life Sciences of Corvinus University of Budapest on 29 th November 2011, the following committee was designated for defence. Committee: Chair: Tibor Deák, D.Sc. Members: Péter Biacs, D.Sc. László Abrankó, Ph.D. Judit Tornai-Lehoczki, Ph.D. Judit Beczner, C.Sc. Opponents: Adrienn Micsinai, Ph.D. László Varga, Ph.D. Secretary: László Abrankó, Ph.D. iii “I am among those who think that science has great beauty. A scientist in his laboratory is not only a technichian: he is also a child placed before natural phenomena which impress him like a fairy tale.” Marie Curie (1867-1934) iv CONTENTS LIST OF ABBREVIATION............................................................................................................vii 1. INTRODUCTION..........................................................................................................................1 2. LITERATURE REVIEW..............................................................................................................3 2.1. Characterization of red meat.....................................................................................................3 2.1.1. Microbiological aspects of food spoilage especially on pork............................................4 2.2. Food -

Aquatic Microbial Ecology 80:15
The following supplement accompanies the article Isolates as models to study bacterial ecophysiology and biogeochemistry Åke Hagström*, Farooq Azam, Carlo Berg, Ulla Li Zweifel *Corresponding author: [email protected] Aquatic Microbial Ecology 80: 15–27 (2017) Supplementary Materials & Methods The bacteria characterized in this study were collected from sites at three different sea areas; the Northern Baltic Sea (63°30’N, 19°48’E), Northwest Mediterranean Sea (43°41'N, 7°19'E) and Southern California Bight (32°53'N, 117°15'W). Seawater was spread onto Zobell agar plates or marine agar plates (DIFCO) and incubated at in situ temperature. Colonies were picked and plate- purified before being frozen in liquid medium with 20% glycerol. The collection represents aerobic heterotrophic bacteria from pelagic waters. Bacteria were grown in media according to their physiological needs of salinity. Isolates from the Baltic Sea were grown on Zobell media (ZoBELL, 1941) (800 ml filtered seawater from the Baltic, 200 ml Milli-Q water, 5g Bacto-peptone, 1g Bacto-yeast extract). Isolates from the Mediterranean Sea and the Southern California Bight were grown on marine agar or marine broth (DIFCO laboratories). The optimal temperature for growth was determined by growing each isolate in 4ml of appropriate media at 5, 10, 15, 20, 25, 30, 35, 40, 45 and 50o C with gentle shaking. Growth was measured by an increase in absorbance at 550nm. Statistical analyses The influence of temperature, geographical origin and taxonomic affiliation on growth rates was assessed by a two-way analysis of variance (ANOVA) in R (http://www.r-project.org/) and the “car” package. -

Control of Phytopathogenic Microorganisms with Pseudomonas Sp. and Substances and Compositions Derived Therefrom
(19) TZZ Z_Z_T (11) EP 2 820 140 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A01N 63/02 (2006.01) A01N 37/06 (2006.01) 10.01.2018 Bulletin 2018/02 A01N 37/36 (2006.01) A01N 43/08 (2006.01) C12P 1/04 (2006.01) (21) Application number: 13754767.5 (86) International application number: (22) Date of filing: 27.02.2013 PCT/US2013/028112 (87) International publication number: WO 2013/130680 (06.09.2013 Gazette 2013/36) (54) CONTROL OF PHYTOPATHOGENIC MICROORGANISMS WITH PSEUDOMONAS SP. AND SUBSTANCES AND COMPOSITIONS DERIVED THEREFROM BEKÄMPFUNG VON PHYTOPATHOGENEN MIKROORGANISMEN MIT PSEUDOMONAS SP. SOWIE DARAUS HERGESTELLTE SUBSTANZEN UND ZUSAMMENSETZUNGEN RÉGULATION DE MICRO-ORGANISMES PHYTOPATHOGÈNES PAR PSEUDOMONAS SP. ET DES SUBSTANCES ET DES COMPOSITIONS OBTENUES À PARTIR DE CELLE-CI (84) Designated Contracting States: • O. COUILLEROT ET AL: "Pseudomonas AL AT BE BG CH CY CZ DE DK EE ES FI FR GB fluorescens and closely-related fluorescent GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO pseudomonads as biocontrol agents of PL PT RO RS SE SI SK SM TR soil-borne phytopathogens", LETTERS IN APPLIED MICROBIOLOGY, vol. 48, no. 5, 1 May (30) Priority: 28.02.2012 US 201261604507 P 2009 (2009-05-01), pages 505-512, XP55202836, 30.07.2012 US 201261670624 P ISSN: 0266-8254, DOI: 10.1111/j.1472-765X.2009.02566.x (43) Date of publication of application: • GUANPENG GAO ET AL: "Effect of Biocontrol 07.01.2015 Bulletin 2015/02 Agent Pseudomonas fluorescens 2P24 on Soil Fungal Community in Cucumber Rhizosphere (73) Proprietor: Marrone Bio Innovations, Inc. -

Health Hazard Evaluation Report 2014-0050-3234
Evaluation of an Unpleasant Odor at an Aircraft Ejection Seat Manufacturer Kendra Broadwater, MPH Marie A. de Perio, MD Scott E. Brueck, MS, CIH Nancy C. Burton, PhD, CIH Angela R. Lemons, MS Brett J. Green, PhD HHE Report No. 2014-0050-3234 April 2015 U.S. Department of Health and Human Services Centers for Disease Control and Prevention Page 1 NationalHealth Institute Hazard forEvaluation Occupational Report 2014-0050-3234 Safety and Health Contents Highlights ...............................................i Abbreviations ..................................... iii Introduction ......................................... 1 Methods ............................................... 2 Results and Discussion ........................ 4 Conclusions ........................................ 14 Recommendations............................. 15 Appendix A ........................................ 18 Appendix B......................................... 22 Appendix C ........................................ 25 Appendix D ........................................27 References......................................... 28 Acknowledgement............................33 The employer is required to post a copy of this report for 30 days at or near the workplace(s) of affected employees. The employer must take steps to ensure that the posted report is not altered, defaced, or covered by other material. The cover photo is a close-up image of sorbent tubes, which are used by the HHE Program to measure airborne exposures. This photo is an artistic representation that may not be related to this Health Hazard Evaluation. Photo by NIOSH. Page 2 Health Hazard Evaluation Report 2014-0050-3234 Highlights of this Evaluation The Health Hazard Evaluation Program received a request from the employer at a facility where aircraft ejection seats are made. Employees were concerned about a lingering odor that was first noticed in April 2013 and continued to be a problem. The odor remained in materials and goods after they left the facility. What We Did ● We visited the facility in May 2014. -
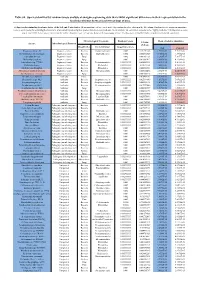
Table S8. Species Identified by Random Forests Analysis of Shotgun Sequencing Data That Exhibit Significant Differences In
Table S8. Species identified by random forests analysis of shotgun sequencing data that exhibit significant differences in their representation in the fecal microbiomes between each two groups of mice. (a) Species discriminating fecal microbiota of the Soil and Control mice. Mean importance of species identified by random forest are shown in the 5th column. Random forests assigns an importance score to each species by estimating the increase in error caused by removing that species from the set of predictors. In our analysis, we considered a species to be “highly predictive” if its importance score was at least 0.001. T-test was performed for the relative abundances of each species between the two groups of mice. P-values were at least 0.05 to be considered statistically significant. Microbiological Taxonomy Random Forests Mean of relative abundance P-Value Species Microbiological Function (T-Test) Classification Bacterial Order Importance Score Soil Control Rhodococcus sp. 2G Engineered strain Bacteria Corynebacteriales 0.002 5.73791E-05 1.9325E-05 9.3737E-06 Herminiimonas arsenitoxidans Engineered strain Bacteria Burkholderiales 0.002 0.005112829 7.1580E-05 1.3995E-05 Aspergillus ibericus Engineered strain Fungi 0.002 0.001061181 9.2368E-05 7.3057E-05 Dichomitus squalens Engineered strain Fungi 0.002 0.018887472 8.0887E-05 4.1254E-05 Acinetobacter sp. TTH0-4 Engineered strain Bacteria Pseudomonadales 0.001333333 0.025523638 2.2311E-05 8.2612E-06 Rhizobium tropici Engineered strain Bacteria Rhizobiales 0.001333333 0.02079554 7.0081E-05 4.2000E-05 Methylocystis bryophila Engineered strain Bacteria Rhizobiales 0.001333333 0.006513543 3.5401E-05 2.2044E-05 Alteromonas naphthalenivorans Engineered strain Bacteria Alteromonadales 0.001 0.000660472 2.0747E-05 4.6463E-05 Saccharomyces cerevisiae Engineered strain Fungi 0.001 0.002980726 3.9901E-05 7.3043E-05 Bacillus phage Belinda Antibiotic Phage 0.002 0.016409765 6.8789E-07 6.0681E-08 Streptomyces sp. -

Pseudomonas Versuta Sp. Nov., Isolated from Antarctic Soil 1 Wah
*Manuscript 1 Pseudomonas versuta sp. nov., isolated from Antarctic soil 1 2 3 1,2 3 1 2,4 1,5 4 2 Wah Seng See-Too , Sergio Salazar , Robson Ee , Peter Convey , Kok-Gan Chan , 5 6 3 Álvaro Peix 3,6* 7 8 4 1Division of Genetics and Molecular Biology, Institute of Biological Sciences, Faculty of 9 10 11 5 Science University of Malaya, 50603 Kuala Lumpur, Malaysia 12 13 6 2National Antarctic Research Centre (NARC), Institute of Postgraduate Studies, University of 14 15 16 7 Malaya, 50603 Kuala Lumpur, Malaysia 17 18 8 3Instituto de Recursos Naturales y Agrobiología. IRNASA -CSIC, Salamanca, Spain 19 20 4 21 9 British Antarctic Survey, NERC, High Cross, Madingley Road, Cambridge CB3 OET, UK 22 23 10 5UM Omics Centre, University of Malaya, Kuala Lumpur, Malaysia 24 25 11 6Unidad Asociada Grupo de Interacción Planta-Microorganismo Universidad de Salamanca- 26 27 28 12 IRNASA ( CSIC) 29 30 13 , IRNASA-CSIC, 31 32 33 14 c/Cordel de Merinas 40 -52, 37008 Salamanca, Spain. Tel.: +34 923219606. 34 35 15 E-mail address: [email protected] (A. Peix) 36 37 38 39 16 Abstract: 40 41 42 43 17 In this study w e used a polyphas ic taxonomy approach to analyse three bacterial strains 44 45 18 coded L10.10 T, A4R1.5 and A4R1.12 , isolated in the course of a study of quorum -quenching 46 47 19 bacteria occurring Antarctic soil . The 16S rRNA gene sequence was identical in the three 48 49 50 20 strains and showed 99.7% pairwise similarity with respect to the closest related species 51 52 21 Pseudomonas weihenstephanensis WS4993 T, and the next closest related species were P. -
Pseudomonas Versuta Sp. Nov., Isolated from Antarctic Soil
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by NERC Open Research Archive Accepted Manuscript Title: Pseudomonas versuta sp. nov., isolated from Antarctic soil Authors: Wah Seng See-Too, Sergio Salazar, Robson Ee, Peter Convey, Kok-Gan Chan, Alvaro´ Peix PII: S0723-2020(17)30039-5 DOI: http://dx.doi.org/doi:10.1016/j.syapm.2017.03.002 Reference: SYAPM 25827 To appear in: Received date: 12-1-2017 Revised date: 20-3-2017 Accepted date: 24-3-2017 Please cite this article as: Wah Seng See-Too, Sergio Salazar, Robson Ee, Peter Convey, Kok-Gan Chan, Alvaro´ Peix, Pseudomonas versuta sp.nov., isolated from Antarctic soil, Systematic and Applied Microbiologyhttp://dx.doi.org/10.1016/j.syapm.2017.03.002 This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain. Pseudomonas versuta sp. nov., isolated from Antarctic soil Wah Seng See-Too1,2, Sergio Salazar3, Robson Ee1, Peter Convey 2,4, Kok-Gan Chan1,5, Álvaro Peix3,6* 1Division of Genetics and Molecular Biology, Institute of Biological Sciences, Faculty of Science University of Malaya, 50603 Kuala Lumpur, Malaysia 2National Antarctic Research Centre (NARC), Institute of Postgraduate Studies, University of Malaya, 50603 Kuala Lumpur, Malaysia 3Instituto de Recursos Naturales y Agrobiología. -
Engineering Pseudomonas Protegens Pf-5 for Nitrogen Fixation and Its Application to Improve Plant Growth Under Nitrogen-Deficient Conditions
Engineering Pseudomonas protegens Pf-5 for Nitrogen Fixation and its Application to Improve Plant Growth under Nitrogen-Deficient Conditions Lorena Setten1, Gabriela Soto1,2, Matteo Mozzicafreddo3, Ana Romina Fox2, Christian Lisi1, Massimiliano Cuccioloni3, Mauro Angeletti3, Elba Pagano1, Antonio Dı´az-Paleo1, Nicola´s Daniel Ayub1,2* 1 Instituto de Gene´tica Ewald A. Favret (CICVyA-INTA), Castelar, Buenos Aires, Argentina, 2 Consejo Nacional de Investigaciones Cientı´ficas y Te´cnicas (CONICET), Cuidad Auto´noma de Buenos Aires, Argentina, 3 School of Biosciences and Biotechnology, University of Camerino, Camerino (MC), Italy Abstract Nitrogen is the second most critical factor for crop production after water. In this study, the beneficial rhizobacterium Pseudomonas protegens Pf-5 was genetically modified to fix nitrogen using the genes encoding the nitrogenase of Pseudomonas stutzeri A1501 via the X940 cosmid. Pf-5 X940 was able to grow in L medium without nitrogen, displayed high nitrogenase activity and released significant quantities of ammonium to the medium. Pf-5 X940 also showed constitutive expression and enzymatic activity of nitrogenase in ammonium medium or in nitrogen-free medium, suggesting a constitutive nitrogen fixation. Similar to Pseudomonas protegens Pf-5, Pseudomonas putida, Pseudomonas veronii and Pseudomonas taetrolens but not Pseudomonas balearica and Pseudomonas stutzeri transformed with cosmid X940 showed constitutive nitrogenase activity and high ammonium production, suggesting that this phenotype depends on the genome context and that this technology to obtain nitrogen-fixing bacteria is not restricted to Pf-5. Interestingly, inoculation of Arabidopsis, alfalfa, tall fescue and maize with Pf-5 X940 increased the ammonium concentration in soil and plant productivity under nitrogen-deficient conditions. -

Diversité Des Bactéries Halophiles Dans L'écosystème Fromager Et
Diversité des bactéries halophiles dans l'écosystème fromager et étude de leurs impacts fonctionnels Diversity of halophilic bacteria in the cheese ecosystem and the study of their functional impacts Thèse de doctorat de l'université Paris-Saclay École doctorale n° 581 Agriculture, Alimentation, Biologie, Environnement et Santé (ABIES) Spécialité de doctorat: Microbiologie Unité de Recherche : Micalis Institute, Jouy-en-Josas, France Référent : AgroParisTech Thèse présentée et soutenue à Paris-Saclay, le 01/04/2021 par Caroline Isabel KOTHE Composition du Jury Michel-Yves MISTOU Président Directeur de Recherche, INRAE centre IDF - Jouy-en-Josas - Antony Monique ZAGOREC Rapporteur & Examinatrice Directrice de Recherche, INRAE centre Pays de la Loire Nathalie DESMASURES Rapporteur & Examinatrice Professeure, Université de Caen Normandie Françoise IRLINGER Examinatrice Ingénieure de Recherche, INRAE centre IDF - Versailles-Grignon Jean-Louis HATTE Examinateur Ingénieur Recherche et Développement, Lactalis Direction de la thèse Pierre RENAULT Directeur de thèse Directeur de Recherche, INRAE (centre IDF - Jouy-en-Josas - Antony) 2021UPASB014 : NNT Thèse de doctorat de Thèse “A master in the art of living draws no sharp distinction between her work and her play; her labor and her leisure; her mind and her body; her education and her recreation. She hardly knows which is which. She simply pursues her vision of excellence through whatever she is doing, and leaves others to determine whether she is working or playing. To herself, she always appears to be doing both.” Adapted to Lawrence Pearsall Jacks REMERCIEMENTS Remerciements L'opportunité de faire un doctorat, en France, à l’Unité mixte de recherche MICALIS de Jouy-en-Josas a provoqué de nombreux changements dans ma vie : un autre pays, une autre langue, une autre culture et aussi, un nouveau domaine de recherche. -

Food Microbiology Comparative Evaluation of Spoilage-Related
Food Microbiology 77 (2019) 166–172 Contents lists available at ScienceDirect Food Microbiology journal homepage: www.elsevier.com/locate/fm Comparative evaluation of spoilage-related bacterial diversity and metabolite profiles in chilled beef stored under air and vacuum packaging T Ahmad Rois Mansura,d,1, Eun-Ji Songb,d,1, Yong-Sun Choa, Young-Do Namb,d, Yun-Sang Choic, ∗ ∗∗ Dae-Ok Kime, Dong-Ho Seob, , Tae Gyu Nama, a Food Analysis Center, Korea Food Research Institute, Wanju, 55365, Republic of Korea b Research Group of Healthcare, Korea Food Research Institute, Wanju, 55365, Republic of Korea c Food Processing Research Center, Korea Food Research Institute, Wanju, 55365, Republic of Korea d Department of Food Biotechnology, Korea University of Science and Technology, Daejeon, 34113, Republic of Korea e Department of Food Science and Biotechnology, Kyung Hee University, Yongin, 17104, Republic of Korea ARTICLE INFO ABSTRACT Keywords: Microbial spoilage is a complex event to which different bacterial populations and metabolites can contribute Beef spoilage depending on the storage conditions. This study explored the evolution of spoilage and related volatile organic Bacterial communities compounds (VOCs) in chilled beef under air and vacuum packaging (VP). The results suggested that different Bacterial metabolites storage conditions affected changes in bacterial communities and metabolites in beef and consequently affected Volatile compounds the odor properties of the stored beef, thereby leading to spoilage. Bacterial species belonging to Pseudomonadaceae (Pseudomonas spp.) and lactic acid bacteria (Lactobacillus sp.) dominated the bacterial com- munities in beef stored under air and VP, respectively, with several VOCs associated with off-odors of the stored beef and most likely produced by both bacteria.