Histidine: a Systematic Review on Metabolism and Physiological Effects in Human and Different Animal Species
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Neurotransmitter Resource Guide
NEUROTRANSMITTER RESOURCE GUIDE Science + Insight doctorsdata.com Doctor’s Data, Inc. Neurotransmitter RESOURCE GUIDE Table of Contents Sample Report Sample Report ........................................................................................................................................................................... 1 Analyte Considerations Phenylethylamine (B-phenylethylamine or PEA) ................................................................................................. 1 Tyrosine .......................................................................................................................................................................................... 3 Tyramine ........................................................................................................................................................................................4 Dopamine .....................................................................................................................................................................................6 3, 4-Dihydroxyphenylacetic Acid (DOPAC) ............................................................................................................... 7 3-Methoxytyramine (3-MT) ............................................................................................................................................... 9 Norepinephrine ........................................................................................................................................................................ -

Effect of Peptide Histidine Isoleucine on Water and Electrolyte Transport in the Human Jejunum
Gut: first published as 10.1136/gut.25.6.624 on 1 June 1984. Downloaded from Gut, 1984, 25, 624-628 Alimentary tract and pancreas Effect of peptide histidine isoleucine on water and electrolyte transport in the human jejunum K J MORIARTY, J E HEGARTY, K TATEMOTO, V MUTT, N D CHRISTOFIDES, S R BLOOM, AND J R WOOD From the Department of Gastroenterology, St Bartholomew's Hospital, London, The Liver Unit, King's College Hospital, London, Department ofMedicine, Hammersmith Hospital, London, and Department of Biochemistry, Karolinska Institute, Stockholm, Sweden SUMMARY Peptide histidine isoleucine, a 27 amino acid peptide with close amino acid sequence homology to vasoactive intestinal peptide and secretin, is distributed throughout the mammalian intestinal tract, where it has been localised to intramural neurones. An intestinal perfusion technique has been used to study the effect of intravenous peptide histidine isoleucine (44.5 pmol/kg/min) on water and electrolyte transport from a plasma like electrolyte solution in human jejunum in vivo. Peptide histidine isoleucine infusion produced peak plasma peptide histidine isoleucine concentrations in the range 2000-3000 pmolIl, flushing, tachycardia and a reduction in diastolic blood pressure. Peptide histidine isoleucine caused a significant inhibition of net absorption of water, sodium, potassium and bicarbonate and induced a net secretion of chloride, these changes being completely reversed during the post-peptide histidine isoleucine period. These findings suggest that endogenous peptide histidine isoleucine may participate in the neurohumoral regulation of water and electrolyte transport in the human jejunum. http://gut.bmj.com/ Peptide histidine isoleucine, isolated originally from INTESTINAL PERFUSION mammalian small intestine, is a 27-amino acid After an eight hour fast, each subject swallowed a peptide having close amino acid sequence homology double lumen intestinal perfusion tube, incorpo- to vasoactive intestinal peptide and secretin. -

Isoleucine, an Essential Amino Acid, Prevents Liver Metastases of Colon Cancer by Antiangiogenesis Kazumoto Murata1 and Masami Moriyama2
Research Article Isoleucine, an Essential Amino Acid, Prevents Liver Metastases of Colon Cancer by Antiangiogenesis Kazumoto Murata1 and Masami Moriyama2 1The First Department of Internal Medicine, Mie University School of Medicine, Tsu, Mie, Japan and 2Microbiology and Immunology, Keio University School of Medicine, Tokyo, Japan Abstract infection in the airways of cystic fibrosis (8), and susceptibility to salmonella infection in mouse intestinal tracts (9). In addition to In spite of recent advances in the treatment of colon cancer, h multiple liver metastases of colon cancer are still difficult to their direct antimicrobial activities, -defensins are strong treat. Some chemotherapeutic regimens have been reported to chemotactic factors for memory T cells and dendritic cells, be efficient, but there is a high risk of side effects associated suggesting that they also play an important role in acquired immunity (10–12). h-defensins are also inducible by inflammatory with these. Here, we show that isoleucine, an essential amino a h acid, prevents liver metastases in a mouse colon cancer cytokines, such as tumor necrosis factor- and interleukin-1 (13, 14). Recently, Fehlbaum et al. (15) reported that isoleucine metastatic model. Because isoleucine is a strong inducer of h B-defensin, we first hypothesized that it prevented liver and its analogues are highly specific inducers of -defensins. We thus originally hypothesized that isoleucine may contribute to metastases via the accumulation of dendritic cells or memory B tumor immunity through both innate and acquired immunity by T cells through up-regulation of -defensin. However, neither h B-defensin nor immunologic responses were induced by induction of -defensins. -
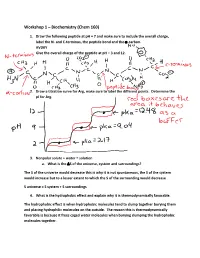
Workshop 1 – Biochemistry (Chem 160)
Workshop 1 – Biochemistry (Chem 160) 1. Draw the following peptide at pH = 7 and make sure to include the overall charge, label the N- and C-terminus, the peptide bond and the -carbon. AVDKY Give the overall charge of the peptide at pH = 3 and 12. 2. Draw a titration curve for Arg, make sure to label the different points. Determine the pI for Arg. 3. Nonpolar solute + water = solution a. What is the S of the universe, system and surroundings? The S of the universe would decrease this is why it is not spontaneous, the S of the system would increase but to a lesser extent to which the S of the surrounding would decrease S universe = S system + S surroundings 4. What is the hydrophobic effect and explain why it is thermodynamically favorable. The hydrophobic effect is when hydrophobic molecules tend to clump together burying them and placing hydrophilic molecules on the outside. The reason this is thermodynamically favorable is because it frees caged water molecules when burying clumping the hydrophobic molecules together. 5. Urea dissolves very readily in water, but the solution becomes very cold as the urea dissolves. How is this possible? Urea dissolves in water because when dissolving there is a net increase in entropy of the universe. The heat exchange, getting colder only reflects the enthalpy (H) component of the total energy change. The entropy change is high enough to offset the enthalpy component and to add up to an overall -G 6. A mutation that changes an alanine residue in the interior of a protein to valine is found to lead to a loss of activity. -

Diphenhydramine Hydrochloride (CASRN 147-24-0) in F344/N Rats
NATIONAL TOXICOLOGY PROGRAM Technical Report Series No. 355 TOXICOLOGY AND CARCINOGENESIS STUDIES OF DIPHENHYDRAMINE HYDROCHLORIDE (CAS NO. 147-24-0) IN F344/N RATS AND B6C3F1 MICE (FEED STUDIES) LJ.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Public Health Service National Institutes of Health NTP ‘TECHNICAL REPORT ON THE TOXICOLOGY AND CARCINOGENESIS STUDIES OF DIPHENHYDRAMINE HYDROCHLORIDE (CAS NO. 147-24-0) IN F344/N RATS AND B6C3F1 MICE (FEED STUDIES) R. Melnick, Ph.D., Study Scientist NATIONAL TOXICOLOGY PROGRAM P.O. Box 12233 Research Triangle Park, NC 27709 September 1989 NTP TR 355 NIH Publication No. 89-2810 U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Public Health Service National Institutes of Health CONTENTS PAGE ABSTRACT ................................................................ 3 EXPLANATION OF LEVELS OF EVIDENCE OF CARCINOGENIC ACTIVITY .................. 6 CONTRIBUTORS ............................................................ 7 PEERREVIEWPANEL ........................................................ 8 SUMMARY OF PEER REVIEW COMMENTS ......................................... 9 I. INTRODUCTION ........................................................ 11 I1. MATERIALS AND METHODS .............................................. 21 III. RESULTS ............................................................. 35 RATS ............................................................. 36 MICE ............................................................. 45 GENETIC TOXICOLOGY ............................................... 53 IV. -

Effects of Single Amino Acid Deficiency on Mrna Translation Are Markedly
www.nature.com/scientificreports OPEN Efects of single amino acid defciency on mRNA translation are markedly diferent for methionine Received: 12 December 2016 Accepted: 4 May 2018 versus leucine Published: xx xx xxxx Kevin M. Mazor, Leiming Dong, Yuanhui Mao, Robert V. Swanda, Shu-Bing Qian & Martha H. Stipanuk Although amino acids are known regulators of translation, the unique contributions of specifc amino acids are not well understood. We compared efects of culturing HEK293T cells in medium lacking either leucine, methionine, histidine, or arginine on eIF2 and 4EBP1 phosphorylation and measures of mRNA translation. Methionine starvation caused the most drastic decrease in translation as assessed by polysome formation, ribosome profling, and a measure of protein synthesis (puromycin-labeled polypeptides) but had no signifcant efect on eIF2 phosphorylation, 4EBP1 hyperphosphorylation or 4EBP1 binding to eIF4E. Leucine starvation suppressed polysome formation and was the only tested condition that caused a signifcant decrease in 4EBP1 phosphorylation or increase in 4EBP1 binding to eIF4E, but efects of leucine starvation were not replicated by overexpressing nonphosphorylatable 4EBP1. This suggests the binding of 4EBP1 to eIF4E may not by itself explain the suppression of mRNA translation under conditions of leucine starvation. Ribosome profling suggested that leucine deprivation may primarily inhibit ribosome loading, whereas methionine deprivation may primarily impair start site recognition. These data underscore our lack of a full -

Role of Dietary Histidine in the Prevention of Obesity and Metabolic Syndrome
Open access Editorial Open Heart: first published as 10.1136/openhrt-2017-000676 on 1 July 2018. Downloaded from Role of dietary histidine in the prevention of obesity and metabolic syndrome James J DiNicolantonio,1 Mark F McCarty,2 James H OKeefe 1 To cite: DiNicolantonio JJ, HISTIDINE SUPPLEMENTATION AMELIORATES histidine dose dependently increases hypo- McCarty MF, OKeefe JH. Role of METABOLIC SYNDROME thalamic levels of histamine as well as hypo- dietary histidine in the prevention of obesity and A recent Chinese supplementation study, in thalamic activity of histidine decarboxylase, metabolic syndrome. Open Heart which obese middle-aged women diagnosed the enzyme which converts histidine to hista- 10 2018;5:e000676. doi:10.1136/ with metabolic syndrome received 12 weeks mine. Such administration also inhibits food openhrt-2017-000676 of supplemental histidine (2 g, twice daily) or consumption—an effect that is blocked in matching placebo, achieved remarkable find- animals pretreated with an irreversible inhib- 1 Accepted 24 April 2018 ings. Insulin sensitivity improved significantly itor of histidine decarboxylase. in the histidine-supplemented subjects, and Neuronal histamine release in the hypo- this may have been partially attributable to thalamus is subject to feedback regulation loss of body fat. Body mass index (BMI), waist by presynaptic H3 receptors. In rodent circumference and body fat declined in the studies, antagonists and inverse agonists for histidine-supplemented group relative to the these receptors have been shown to mark- placebo group; the average fat loss in the histi- edly amplify hypothalamic histamine levels, dine group was a robust 2.71 kg. Markers of suppress feeding, decrease body weight and systemic inflammation such as serum tumour enhance metabolic rate.11–15 Such agents may necrosis factor-alpha (TNF-α) and inter- have clinical potential for managing obesity. -

Histidine Decarboxylase Knockout Mice Production and Signal
Nitric Oxide Mediates T Cell Cytokine Production and Signal Transduction in Histidine Decarboxylase Knockout Mice This information is current as Agnes Koncz, Maria Pasztoi, Mercedesz Mazan, Ferenc of October 1, 2021. Fazakas, Edit Buzas, Andras Falus and Gyorgy Nagy J Immunol 2007; 179:6613-6619; ; doi: 10.4049/jimmunol.179.10.6613 http://www.jimmunol.org/content/179/10/6613 Downloaded from References This article cites 41 articles, 8 of which you can access for free at: http://www.jimmunol.org/content/179/10/6613.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average by guest on October 1, 2021 Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2007 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Nitric Oxide Mediates T Cell Cytokine Production and Signal Transduction in Histidine Decarboxylase Knockout Mice1 Agnes Koncz,*† Maria Pasztoi,* Mercedesz Mazan,* Ferenc Fazakas,‡ Edit Buzas,* Andras Falus,*§ and Gyorgy Nagy2,3*¶ Histamine is a key regulator of the immune system. -

Amino Acid Recognition by Aminoacyl-Trna Synthetases
www.nature.com/scientificreports OPEN The structural basis of the genetic code: amino acid recognition by aminoacyl‑tRNA synthetases Florian Kaiser1,2,4*, Sarah Krautwurst3,4, Sebastian Salentin1, V. Joachim Haupt1,2, Christoph Leberecht3, Sebastian Bittrich3, Dirk Labudde3 & Michael Schroeder1 Storage and directed transfer of information is the key requirement for the development of life. Yet any information stored on our genes is useless without its correct interpretation. The genetic code defnes the rule set to decode this information. Aminoacyl-tRNA synthetases are at the heart of this process. We extensively characterize how these enzymes distinguish all natural amino acids based on the computational analysis of crystallographic structure data. The results of this meta-analysis show that the correct read-out of genetic information is a delicate interplay between the composition of the binding site, non-covalent interactions, error correction mechanisms, and steric efects. One of the most profound open questions in biology is how the genetic code was established. While proteins are encoded by nucleic acid blueprints, decoding this information in turn requires proteins. Te emergence of this self-referencing system poses a chicken-or-egg dilemma and its origin is still heavily debated 1,2. Aminoacyl-tRNA synthetases (aaRSs) implement the correct assignment of amino acids to their codons and are thus inherently connected to the emergence of genetic coding. Tese enzymes link tRNA molecules with their amino acid cargo and are consequently vital for protein biosynthesis. Beside the correct recognition of tRNA features3, highly specifc non-covalent interactions in the binding sites of aaRSs are required to correctly detect the designated amino acid4–7 and to prevent errors in biosynthesis5,8. -

Electrochemical Studies of Dl-Leucine, L-Proline and L
ELECTROCHEMICAL STUDIES OF DL -LEUCINE, 60 L-PROLINE AND L-TRYPTOPHAN AND THEIR INTERACTION WITH COPPER AND IRON 30 c b A) M. A. Jabbar, R. J. Mannan, S. Salauddin and B. µ a Rashid 0 Department of Chemistry, University of Dhaka, ( Current Dhaka-1000, Bangladesh -30 Introduction -60 In vitro study of the charge transfer reactions coupled -800 -400 0 400 800 with chemical reactions can give important indication of Potential vs. Ag/AgCl (mV) about actual biological processes occurring in human Fig.1. Comparison of the cyclic voltammogram of system. Understanding of such charge-transfer 5.0mM (a) DL -Leucine, (b) Cu-DL -Leucine ion and mechanism will help to determine the effectiveness of (c) [Fe-DL -Leucine] in 0.1M KCl solution at a Pt- nutrition, metabolism and treatment of various biological button electrode. Scan rates 50 mV/s. disorders. In the previous research, the redox behaviour of 40 various amino acids and biochemically important compounds and their charge transfer reaction and their b interaction of metal ions were studied [1,2]. In the present 20 ) a c research, the redox behavior and the charge transfer µΑ kinetics of DL -Leucine, L-Proline and L-Tryptophan in 0 the presence and absence of copper and iron will be investigated. Current ( -20 Experimental A computerized electrochemistry system developed by -40 -800 -400 0 400 800 Advanced Analytics, Virginia, USA, (Model-2040) Potential vs. Ag/AgCl (mV) consisting of three electrodes micro-cell with a saturated Ag/AgCl reference, a Pt-wire auxiliary and a pretreated Fig.2 . Comparison of the cyclic voltammogram of Pt-button working electrode is employed to investigate 5.0mM (a) L-Proline, (b) Cu-L-Proline and (c) Fe-L- Proline in 0.1M KCl solution at a Pt-button different amino acids and metal-amino acid systems. -

In Vitro Pharmacology of Clinically Used Central Nervous System-Active Drugs As Inverse H1 Receptor Agonists
0022-3565/07/3221-172–179$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 322, No. 1 Copyright © 2007 by The American Society for Pharmacology and Experimental Therapeutics 118869/3215703 JPET 322:172–179, 2007 Printed in U.S.A. In Vitro Pharmacology of Clinically Used Central Nervous System-Active Drugs as Inverse H1 Receptor Agonists R. A. Bakker,1 M. W. Nicholas,2 T. T. Smith, E. S. Burstein, U. Hacksell, H. Timmerman, R. Leurs, M. R. Brann, and D. M. Weiner Department of Medicinal Chemistry, Leiden/Amsterdam Center for Drug Research, Vrije Universiteit Amsterdam, Amsterdam, The Netherlands (R.A.B., H.T., R.L.); ACADIA Pharmaceuticals Inc., San Diego, California (R.A.B., M.W.N., T.T.S., E.S.B., U.H., M.R.B., D.M.W.); and Departments of Pharmacology (M.R.B.), Neurosciences (D.M.W.), and Psychiatry (D.M.W.), University of California, San Diego, California Received January 2, 2007; accepted March 30, 2007 Downloaded from ABSTRACT The human histamine H1 receptor (H1R) is a prototypical G on this screen, we have reported on the identification of 8R- protein-coupled receptor and an important, well characterized lisuride as a potent stereospecific partial H1R agonist (Mol target for the development of antagonists to treat allergic con- Pharmacol 65:538–549, 2004). In contrast, herein we report on jpet.aspetjournals.org ditions. Many neuropsychiatric drugs are also known to po- a large number of varied clinical and chemical classes of drugs tently antagonize this receptor, underlying aspects of their side that are active in the central nervous system that display potent effect profiles. -

Convenient Preparation of Poly(L-Histidine) by the Direct Polymerization of L-Histidine Or Nim Benzyl-L-Histidine with Diphenylphosphoryl Azide
Polymer Journal, Vol. 13, No. 12, pp 1151-1154 (1981) SHORT COMMUNICATION Convenient Preparation of Poly(L-histidine) by the Direct Polymerization of L-Histidine or Nim_Benzyl-L-histidine with Diphenylphosphoryl Azide Takumi NARUSE, Bun-ichiro NAKAJIMA, Akihiro TSUTSUMI, and Norio NISHI Department of Polymer Science, Faculty of Science, Hokkaido University, Nishi 8-chome, Kita 10-jo, Kita-ku, Sapporo 060, Japan. (Received August 14, 1981) KEY WORDS Polymerization I Diphenylphosphoryl Azide (DPPA) I L- Histidine I N'm-Benzyl-L-histidine I Poly(L-histidine) I Poly(N'm-benzyl-L histidine) I IR Spectrum I 13C NMR Spectrum I Intrinsic Viscosity I Poly(L-histidine) is a very interesting poly(a histidine) by the polymerization of L-histidine with amino acid) as a synthetic functional polymer or as iodine-phosphonic acid esters was unsuccessful. The a model for the functional biopolymer such as simplification of the synthetic route for poly(L enzymes. It is known, however, that poly(L histidine) is still necessary. histidine) can not be prepared by the Fuchs Diphenylphosphoryl azide (DPPA) has been used Farthing method 1 which is very popular for prepar as a convenient reagent for racemization-free pep ing poly(a-amino acid)s. A synthetic route with tide synthesis, since it was synthesized by Shioiri et several steps involving the synthesis of NCA (a a!. in 1972.6 Recently, we reported that this reagent amino acid N-carboxyanhydride) by the rather can also be used successfully for the polymerization classical Leuchs method/ have been used for the of amino acids or peptides.