Solutions to 7.012 Problem Set 1
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Effects of Dietary L-Glutamine Or L-Glutamine Plus L-Glutamic Acid
Brazilian Journal of Poultry Science Revista Brasileira de Ciência Avícola Effects of Dietary L-Glutamine or L-Glutamine Plus ISSN 1516-635X Oct - Dec 2015 / Special Issue L-Glutamic Acid Supplementation Programs on the Nutrition - Poultry feeding additives / 093-098 Performance and Breast Meat Yield Uniformity of http://dx.doi.org/10.1590/1516-635xSpecialIssue 42-d-Old Broilers Nutrition-PoultryFeedingAdditives093-098 Author(s) ABSTRACT Ribeiro Jr VIII This study aimed at evaluating four dietary L-Glutamine (L-Gln) Albino LFTI or L-Gln plus L-Glutamate (L-Glu) supplementation programs on the Rostagno HSI Hannas MII performance, breast yield, and uniformity of broilers. A total of 2,112 Ribeiro CLNIII one-d-old male Cobb 500® broilers were distributed according to a Vieira RAIII randomized block design in a 2 × 4 factorial arrangement (L-Gln or L-Gln Araújo WAG deIV Pessoa GBSII plus L-Glu × 4 supplementation programs), totaling eight treatments Messias RKGIII with 12 replicates of 22 broilers each. The supplementation programs Silva DL daIII consisted of the dietary inclusion or not of 0.4% of L-Gln or L-Gln plus L-Glu for four different periods: 0 days (negative control), 9d, 21d, and 42d. Feed intake (FI, g), body weight gain (BWG, g), feed conversion I Federal University of Viçosa, Department ratio (FCR, kg/kg), coefficient of variation of body weight (CV, %), body of Animal Science, Av PH Rolfs S/N, Viçosa 36570-000, MG, Brazil weight uniformity (UNIF, %), breast weight (BW, g), breast yield (BY, II Ajinomoto do Brasil Ind. e Com. de %), coefficient of variation of breast weight (CVB), breast uniformity Alimentos Ltda. -

Effect of Peptide Histidine Isoleucine on Water and Electrolyte Transport in the Human Jejunum
Gut: first published as 10.1136/gut.25.6.624 on 1 June 1984. Downloaded from Gut, 1984, 25, 624-628 Alimentary tract and pancreas Effect of peptide histidine isoleucine on water and electrolyte transport in the human jejunum K J MORIARTY, J E HEGARTY, K TATEMOTO, V MUTT, N D CHRISTOFIDES, S R BLOOM, AND J R WOOD From the Department of Gastroenterology, St Bartholomew's Hospital, London, The Liver Unit, King's College Hospital, London, Department ofMedicine, Hammersmith Hospital, London, and Department of Biochemistry, Karolinska Institute, Stockholm, Sweden SUMMARY Peptide histidine isoleucine, a 27 amino acid peptide with close amino acid sequence homology to vasoactive intestinal peptide and secretin, is distributed throughout the mammalian intestinal tract, where it has been localised to intramural neurones. An intestinal perfusion technique has been used to study the effect of intravenous peptide histidine isoleucine (44.5 pmol/kg/min) on water and electrolyte transport from a plasma like electrolyte solution in human jejunum in vivo. Peptide histidine isoleucine infusion produced peak plasma peptide histidine isoleucine concentrations in the range 2000-3000 pmolIl, flushing, tachycardia and a reduction in diastolic blood pressure. Peptide histidine isoleucine caused a significant inhibition of net absorption of water, sodium, potassium and bicarbonate and induced a net secretion of chloride, these changes being completely reversed during the post-peptide histidine isoleucine period. These findings suggest that endogenous peptide histidine isoleucine may participate in the neurohumoral regulation of water and electrolyte transport in the human jejunum. http://gut.bmj.com/ Peptide histidine isoleucine, isolated originally from INTESTINAL PERFUSION mammalian small intestine, is a 27-amino acid After an eight hour fast, each subject swallowed a peptide having close amino acid sequence homology double lumen intestinal perfusion tube, incorpo- to vasoactive intestinal peptide and secretin. -
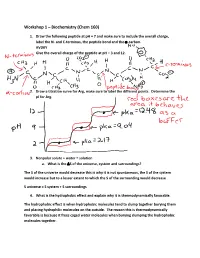
Workshop 1 – Biochemistry (Chem 160)
Workshop 1 – Biochemistry (Chem 160) 1. Draw the following peptide at pH = 7 and make sure to include the overall charge, label the N- and C-terminus, the peptide bond and the -carbon. AVDKY Give the overall charge of the peptide at pH = 3 and 12. 2. Draw a titration curve for Arg, make sure to label the different points. Determine the pI for Arg. 3. Nonpolar solute + water = solution a. What is the S of the universe, system and surroundings? The S of the universe would decrease this is why it is not spontaneous, the S of the system would increase but to a lesser extent to which the S of the surrounding would decrease S universe = S system + S surroundings 4. What is the hydrophobic effect and explain why it is thermodynamically favorable. The hydrophobic effect is when hydrophobic molecules tend to clump together burying them and placing hydrophilic molecules on the outside. The reason this is thermodynamically favorable is because it frees caged water molecules when burying clumping the hydrophobic molecules together. 5. Urea dissolves very readily in water, but the solution becomes very cold as the urea dissolves. How is this possible? Urea dissolves in water because when dissolving there is a net increase in entropy of the universe. The heat exchange, getting colder only reflects the enthalpy (H) component of the total energy change. The entropy change is high enough to offset the enthalpy component and to add up to an overall -G 6. A mutation that changes an alanine residue in the interior of a protein to valine is found to lead to a loss of activity. -

Amino Acids, Glutamine, and Protein Metabolism in Very Low Birth Weight Infants
0031-3998/05/5806-1259 PEDIATRIC RESEARCH Vol. 58, No. 6, 2005 Copyright © 2005 International Pediatric Research Foundation, Inc. Printed in U.S.A. Amino Acids, Glutamine, and Protein Metabolism in Very Low Birth Weight Infants PRABHU S. PARIMI, MARK M. KADROFSKE, LOURDES L. GRUCA, RICHARD W. HANSON, AND SATISH C. KALHAN Department of Pediatrics, Schwartz Center for Metabolism and Nutrition, Case Western Reserve University School of Medicine, MetroHealth Medical Center, Cleveland, Ohio, 44109 ABSTRACT Glutamine has been proposed to be conditionally essential for 5 h (AA1.5) resulted in decrease in rate of appearance (Ra) of premature infants, and the currently used parenteral nutrient phenylalanine and urea, but had no effect on glutamine Ra. mixtures do not contain glutamine. De novo glutamine synthesis Infusion of amino acids at 3.0 g/kg/d for 20 h resulted in increase (DGln) is linked to inflow of carbon into and out of the tricar- in DGln, leucine transamination, and urea synthesis, but had no boxylic acid (TCA) cycle. We hypothesized that a higher supply effect on Ra phenylalanine (AA-Ext). These data show an acute of parenteral amino acids by increasing the influx of amino acid increase in parenteral amino acid–suppressed proteolysis, how- carbon into the TCA cycle will enhance the rate of DGln. Very ever, such an effect was not seen when amino acids were infused low birth weight infants were randomized to receive parenteral for 20 h and resulted in an increase in glutamine synthesis. amino acids either 1.5 g/kg/d for 20 h followed by 3.0 g/kg/d for (Pediatr Res 58: 1259–1264, 2005) 5 h (AA1.5) or 3.0 g/kg/d for 20 h followed by 1.5 g/kg/d for 5 h (AA3.0). -

Mendelian Randomization Study on Amino Acid Metabolism Suggests Tyrosine As Causal Trait for Type 2 Diabetes
nutrients Article Mendelian Randomization Study on Amino Acid Metabolism Suggests Tyrosine as Causal Trait for Type 2 Diabetes Susanne Jäger 1,2,* , Rafael Cuadrat 1,2, Clemens Wittenbecher 1,2,3, Anna Floegel 4, Per Hoffmann 5,6, Cornelia Prehn 7 , Jerzy Adamski 2,7,8,9 , Tobias Pischon 10,11,12 and Matthias B. Schulze 1,2,13 1 Department of Molecular Epidemiology, German Institute of Human Nutrition Potsdam-Rehbruecke, 14558 Nuthetal, Germany; [email protected] (R.C.); [email protected] (C.W.); [email protected] (M.B.S.) 2 German Center for Diabetes Research (DZD), 85764 Neuherberg, Germany; [email protected] 3 Department of Nutrition, Harvard T.H. Chan School of Public Health, Boston, MA 02115, USA 4 Leibniz Institute for Prevention Research and Epidemiology-BIPS, 28359 Bremen, Germany; fl[email protected] 5 Human Genomics Research Group, Department of Biomedicine, University of Basel, 4031 Basel, Switzerland; per.hoff[email protected] 6 Institute of Human Genetics, Division of Genomics, Life & Brain Research Centre, University Hospital of Bonn, 53105 Bonn, Germany 7 Research Unit Molecular Endocrinology and Metabolism, Helmholtz Zentrum München, German Research Center for Environmental Health, 85764 Neuherberg, Germany; [email protected] 8 Chair of Experimental Genetics, Center of Life and Food Sciences Weihenstephan, Technische Universität München, 85354 Freising-Weihenstephan, Germany 9 Department of Biochemistry, Yong Loo Lin School of Medicine, National University of Singapore, 8 Medical Drive, -

H-Tyrosine-Glycine-Phenylalanine-Glycine-Glycine-OH for the Treatment of Chronic Idiopathic Myelofibrosis
European Medicines Agency Pre-authorisation Evaluation of Medicines for Human Use Document Date: London, 5 October 2009 Doc.Ref.: EMEA/COMP/1579/2003 Rev.1 Please note that this product was withdrawn from the Community Register of designated Orphan Medicinal Products in March 2009 on request of the Sponsor. Committee for Orphan Medicinal Products Public summary of positive opinion for orphan designation of H-Tyrosine-Glycine-Phenylalanine-Glycine-Glycine-OH for the treatment of chronic idiopathic myelofibrosis On 20 October 2003, orphan designation (EU/3/03/167) was granted by the European Commission to Abiogen Pharma S.p.A, Italy, for H-Tyrosine-Glycine-Phenylalanine-Glycine-Glycine-OH for the treatment of chronic idiopathic myelofibrosis. What is chronic idiopathic myelofibrosis? Chronic idiopathic myelofibrosis is a disease in which cancer cells are found in the blood and in the bone marrow. The bone marrow is the spongy tissue inside the large bones in the body. Normally the bone marrow makes cells called “blasts” that mature into several different types of blood cells that have specific functions in the body. These include red cells, white cells and platelets. Red blood cells carry oxygen and other materials to all tissues of the body. White blood cells fight infection. Platelets make the blood clot. When myelofibrosis develops, the bone marrow produces large number of abnormal blood cells. In chronic idiopathic myelofibrosis the abnormal population of cells produce substances that alter the growth media of bone marrow and makes bone marrow very dense and rigid. As the abnormal bone marrow environment is no more adequate for the cells, some migrate to other sites where proliferation and maturation take place. -

Amino Acid Recognition by Aminoacyl-Trna Synthetases
www.nature.com/scientificreports OPEN The structural basis of the genetic code: amino acid recognition by aminoacyl‑tRNA synthetases Florian Kaiser1,2,4*, Sarah Krautwurst3,4, Sebastian Salentin1, V. Joachim Haupt1,2, Christoph Leberecht3, Sebastian Bittrich3, Dirk Labudde3 & Michael Schroeder1 Storage and directed transfer of information is the key requirement for the development of life. Yet any information stored on our genes is useless without its correct interpretation. The genetic code defnes the rule set to decode this information. Aminoacyl-tRNA synthetases are at the heart of this process. We extensively characterize how these enzymes distinguish all natural amino acids based on the computational analysis of crystallographic structure data. The results of this meta-analysis show that the correct read-out of genetic information is a delicate interplay between the composition of the binding site, non-covalent interactions, error correction mechanisms, and steric efects. One of the most profound open questions in biology is how the genetic code was established. While proteins are encoded by nucleic acid blueprints, decoding this information in turn requires proteins. Te emergence of this self-referencing system poses a chicken-or-egg dilemma and its origin is still heavily debated 1,2. Aminoacyl-tRNA synthetases (aaRSs) implement the correct assignment of amino acids to their codons and are thus inherently connected to the emergence of genetic coding. Tese enzymes link tRNA molecules with their amino acid cargo and are consequently vital for protein biosynthesis. Beside the correct recognition of tRNA features3, highly specifc non-covalent interactions in the binding sites of aaRSs are required to correctly detect the designated amino acid4–7 and to prevent errors in biosynthesis5,8. -

Molecule Based on Evans Blue Confers Superior Pharmacokinetics and Transforms Drugs to Theranostic Agents
Novel “Add-On” Molecule Based on Evans Blue Confers Superior Pharmacokinetics and Transforms Drugs to Theranostic Agents Haojun Chen*1,2, Orit Jacobson*2, Gang Niu2, Ido D. Weiss3, Dale O. Kiesewetter2, Yi Liu2, Ying Ma2, Hua Wu1, and Xiaoyuan Chen2 1Department of Nuclear Medicine, Xiamen Cancer Hospital of the First Affiliated Hospital of Xiamen University, Xiamen, China; 2Laboratory of Molecular Imaging and Nanomedicine, National Institute of Biomedical Imaging and Bioengineering, National Institutes of Health, Bethesda, Maryland; and 3Laboratory of Molecular Immunology, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, Maryland One of the major design considerations for a drug is its The goal of drug development is to achieve high activity and pharmacokinetics in the blood. A drug with a short half-life in specificity for a desired biologic target. However, many potential the blood is less available at a target organ. Such a limitation pharmaceuticals that meet these criteria fail as therapeutics because dictates treatment with either high doses or more frequent doses, of unfavorable pharmacokinetics, in particular, rapid blood clearance, both of which may increase the likelihood of undesirable side effects. To address the need for additional methods to improve which prevents the achievement of therapeutic concentrations. For the blood half-life of drugs and molecular imaging agents, we some drugs, the administration of large or frequently repeated doses developed an “add-on” molecule that contains 3 groups: a trun- is required to achieve and maintain therapeutic levels (1) but can, in cated Evans blue dye molecule that binds to albumin with a low turn, increase the probability of undesired side effects. -

A Review of Dietary (Phyto)Nutrients for Glutathione Support
nutrients Review A Review of Dietary (Phyto)Nutrients for Glutathione Support Deanna M. Minich 1,* and Benjamin I. Brown 2 1 Human Nutrition and Functional Medicine Graduate Program, University of Western States, 2900 NE 132nd Ave, Portland, OR 97230, USA 2 BCNH College of Nutrition and Health, 116–118 Finchley Road, London NW3 5HT, UK * Correspondence: [email protected] Received: 8 July 2019; Accepted: 23 August 2019; Published: 3 September 2019 Abstract: Glutathione is a tripeptide that plays a pivotal role in critical physiological processes resulting in effects relevant to diverse disease pathophysiology such as maintenance of redox balance, reduction of oxidative stress, enhancement of metabolic detoxification, and regulation of immune system function. The diverse roles of glutathione in physiology are relevant to a considerable body of evidence suggesting that glutathione status may be an important biomarker and treatment target in various chronic, age-related diseases. Yet, proper personalized balance in the individual is key as well as a better understanding of antioxidants and redox balance. Optimizing glutathione levels has been proposed as a strategy for health promotion and disease prevention, although clear, causal relationships between glutathione status and disease risk or treatment remain to be clarified. Nonetheless, human clinical research suggests that nutritional interventions, including amino acids, vitamins, minerals, phytochemicals, and foods can have important effects on circulating glutathione which may translate to clinical benefit. Importantly, genetic variation is a modifier of glutathione status and influences response to nutritional factors that impact glutathione levels. This narrative review explores clinical evidence for nutritional strategies that could be used to improve glutathione status. -

Convenient Preparation of Poly(L-Histidine) by the Direct Polymerization of L-Histidine Or Nim Benzyl-L-Histidine with Diphenylphosphoryl Azide
Polymer Journal, Vol. 13, No. 12, pp 1151-1154 (1981) SHORT COMMUNICATION Convenient Preparation of Poly(L-histidine) by the Direct Polymerization of L-Histidine or Nim_Benzyl-L-histidine with Diphenylphosphoryl Azide Takumi NARUSE, Bun-ichiro NAKAJIMA, Akihiro TSUTSUMI, and Norio NISHI Department of Polymer Science, Faculty of Science, Hokkaido University, Nishi 8-chome, Kita 10-jo, Kita-ku, Sapporo 060, Japan. (Received August 14, 1981) KEY WORDS Polymerization I Diphenylphosphoryl Azide (DPPA) I L- Histidine I N'm-Benzyl-L-histidine I Poly(L-histidine) I Poly(N'm-benzyl-L histidine) I IR Spectrum I 13C NMR Spectrum I Intrinsic Viscosity I Poly(L-histidine) is a very interesting poly(a histidine) by the polymerization of L-histidine with amino acid) as a synthetic functional polymer or as iodine-phosphonic acid esters was unsuccessful. The a model for the functional biopolymer such as simplification of the synthetic route for poly(L enzymes. It is known, however, that poly(L histidine) is still necessary. histidine) can not be prepared by the Fuchs Diphenylphosphoryl azide (DPPA) has been used Farthing method 1 which is very popular for prepar as a convenient reagent for racemization-free pep ing poly(a-amino acid)s. A synthetic route with tide synthesis, since it was synthesized by Shioiri et several steps involving the synthesis of NCA (a a!. in 1972.6 Recently, we reported that this reagent amino acid N-carboxyanhydride) by the rather can also be used successfully for the polymerization classical Leuchs method/ have been used for the of amino acids or peptides. -

Amino Acid Chemistry
Handout 4 Amino Acid and Protein Chemistry ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES Amino Acid Chemistry I. Chemistry of amino acids A. General amino acid structure + HN3- 1. All amino acids are carboxylic acids, i.e., they have a –COOH group at the #1 carbon. 2. All amino acids contain an amino group at the #2 carbon (may amino acids have a second amino group). 3. All amino acids are zwitterions – they contain both positive and negative charges at physiological pH. II. Essential and nonessential amino acids A. Nonessential amino acids: can make the carbon skeleton 1. From glycolysis. 2. From the TCA cycle. B. Nonessential if it can be made from an essential amino acid. 1. Amino acid "sparing". 2. May still be essential under some conditions. C. Essential amino acids 1. Branched chain amino acids (isoleucine, leucine and valine) 2. Lysine 3. Methionine 4. Phenyalanine 5. Threonine 6. Tryptophan 1 Handout 4 Amino Acid and Protein Chemistry D. Essential during rapid growth or for optimal health 1. Arginine 2. Histidine E. Nonessential amino acids 1. Alanine (from pyruvate) 2. Aspartate, asparagine (from oxaloacetate) 3. Cysteine (from serine and methionine) 4. Glutamate, glutamine (from α-ketoglutarate) 5. Glycine (from serine) 6. Proline (from glutamate) 7. Serine (from 3-phosphoglycerate) 8. Tyrosine (from phenylalanine) E. Nonessential and not required for protein synthesis 1. Hydroxyproline (made postranslationally from proline) 2. Hydroxylysine (made postranslationally from lysine) III. Acidic, basic, polar, and hydrophobic amino acids A. Acidic amino acids: amino acids that can donate a hydrogen ion (proton) and thereby decrease pH in an aqueous solution 1. -

L -Glutamic Acid (G1251)
L-Glutamic acid Product Number G 1251 Store at Room Temperature Product Description Precautions and Disclaimer Molecular Formula: C5H9NO4 For Laboratory Use Only. Not for drug, household or Molecular Weight: 147.1 other uses. CAS Number: 56-86-0 pI: 3.081 Preparation Instructions 1 pKa: 2.10 (α-COOH), 9.47 (α-NH2), 4.07 (ϕ-COOH) This product is soluble in 1 M HCl (100 mg/ml), with 2 Specific Rotation: D +31.4 ° (6 N HCl, 22.4 °C) heat as needed, yielding a clear, colorless solution. Synonyms: (S)-2-aminoglutaric acid, (S)-2- The solubility in water at 25 °C has been reported to aminopentanedioic acid, 1-aminopropane-1,3- be 8.6 mg/ml.2 dicarboxylic acid, Glu2 Storage/Stability L-Glutamic acid is one of the two amino acids that Aqueous glutamic acid solutions will form contains a carboxylic acid group in its side chains. pyrrolidonecarboxylic acid slowly at room temperature Glutamic acid is commonly referred to as "glutamate", and more rapidly at 100 °C.9 because its carboxylic acid side chain will be deprotonated and thus negatively charged in its References anionic form at physiological pH. In amino acid 1. Molecular Biology LabFax, Brown, T. A., ed., BIOS metabolism, glutamate is formed from the transfer of Scientific Publishers Ltd. (Oxford, UK: 1991), p. amino groups from amino acids to α-ketoglutarate. It 29. thus acts as an intermediary between ammonia and 2. The Merck Index, 12th ed., Entry# 4477. the amino acids in vivo. Glutamate is converted to 3. Biochemistry, 3rd ed., Stryer, L., W.