Synthesis of Novel Extremely Sterically Hindered Tertiary Alkylamines
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Ep 2611776 B1
(19) TZZ ___T (11) EP 2 611 776 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: C07D 211/36 (2006.01) C07D 471/04 (2006.01) 21.09.2016 Bulletin 2016/38 A61K 31/45 (2006.01) A61P 3/10 (2006.01) C07D 211/76 (2006.01) (21) Application number: 11822081.3 (86) International application number: (22) Date of filing: 25.08.2011 PCT/KR2011/006260 (87) International publication number: WO 2012/030106 (08.03.2012 Gazette 2012/10) (54) PRODUCTION METHOD OF INTERMEDIATE COMPOUND FOR SYNTHESIZING MEDICAMENT HERSTELLUNGSVERFAHREN FÜR EINE INTERMEDIATVERBINDUNG ZUR SYNTHESE EINES MEDIKAMENTS PROCÉDÉ DE PRODUCTION D’UN COMPOSÉ INTERMÉDIAIRE POUR LA SYNTHÈSE D’UN MÉDICAMENT (84) Designated Contracting States: EP-A2- 0 279 435 WO-A1-2006/104356 AL AT BE BG CH CY CZ DE DK EE ES FI FR GB WO-A1-2006/104356 US-A- 5 556 982 GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO US-A1- 2008 039 517 PL PT RO RS SE SI SK SM TR • KIM S ET AL: "Free Radical-Mediated (30) Priority: 03.09.2010 KR 20100086619 Carboxylation by Radical Reaction of Alkyl Iodides with Methyl Oxalyl Chloride", (43) Date of publication of application: TETRAHEDRONLETTERS, PERGAMON, GB, vol. 10.07.2013 Bulletin 2013/28 39, no. 40, 1 October 1998 (1998-10-01), pages 7317-7320, XP004133669, ISSN: 0040-4039, DOI: (73) Proprietor: LG Life Sciences Ltd 10.1016/S0040-4039(98)01568-8 Jongno-gu, Seoul 110-062 (KR) • CHRISTOPHE MORIN ET AL: "Synthesis and Evaluation of Boronated Lysine and (72) Inventors: Bis(carboranylated)[gamma]-Amino Acids as • KIM, Bong Chan Monomers for Peptide Assembly", EUROPEAN Daejeon 305-380 (KR) JOURNAL OF ORGANIC CHEMISTRY, vol. -

Carboxylic Acid Functionalized Butyl Rubber: from Synthesis to Applications
Western University Scholarship@Western Electronic Thesis and Dissertation Repository 2-7-2013 12:00 AM Carboxylic Acid Functionalized Butyl Rubber: From Synthesis to Applications Matthew John McEachran The University of Western Ontario Supervisor Dr. Elizabeth R. Gillies The University of Western Ontario Graduate Program in Chemistry A thesis submitted in partial fulfillment of the equirr ements for the degree in Master of Science © Matthew John McEachran 2013 Follow this and additional works at: https://ir.lib.uwo.ca/etd Part of the Materials Chemistry Commons, Medicinal-Pharmaceutical Chemistry Commons, and the Polymer Chemistry Commons Recommended Citation McEachran, Matthew John, "Carboxylic Acid Functionalized Butyl Rubber: From Synthesis to Applications" (2013). Electronic Thesis and Dissertation Repository. 1117. https://ir.lib.uwo.ca/etd/1117 This Dissertation/Thesis is brought to you for free and open access by Scholarship@Western. It has been accepted for inclusion in Electronic Thesis and Dissertation Repository by an authorized administrator of Scholarship@Western. For more information, please contact [email protected]. Carboxylic Acid Functionalized Butyl Rubber: From Synthesis to Applications (Spine title: Carboxylic Acid Functionalized Butyl Rubber) (Thesis format: Integrated Article) by Matthew John McEachran Graduate Program in Chemistry A thesis submitted in partial fulfillment of the requirements for the degree of Master of Science The School of Graduate and Postdoctoral Studies The University of Western Ontario London, -

Production of Malonic Acid Through the Fermentation of Glucose
University of Pennsylvania ScholarlyCommons Department of Chemical & Biomolecular Senior Design Reports (CBE) Engineering 4-20-2018 Production of Malonic Acid through the Fermentation of Glucose Emily P. Peters University of Pennsylvania, [email protected] Gabrielle J. Schlakman University of Pennsylvania, [email protected] Elise N. Yang University of Pennsylvania, [email protected] Follow this and additional works at: https://repository.upenn.edu/cbe_sdr Part of the Biochemical and Biomolecular Engineering Commons Peters, Emily P.; Schlakman, Gabrielle J.; and Yang, Elise N., "Production of Malonic Acid through the Fermentation of Glucose" (2018). Senior Design Reports (CBE). 107. https://repository.upenn.edu/cbe_sdr/107 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/cbe_sdr/107 For more information, please contact [email protected]. Production of Malonic Acid through the Fermentation of Glucose Abstract The overall process to produce malonic acid has not drastically changed in the past 50 years. The current process is damaging to the environment and costly, requiring high market prices. Lygos, Inc., a lab in Berkeley, California, has published a patent describing a way to produce malonic acid through the biological fermentation of genetically modified easty cells. This proposed technology is appealing as it is both better for the environment and economically friendly. For the process discussed in this report, genetically modified Pichia Kudriavzevii yeast cells will be purchased from the Lygos lab along with the negotiation of exclusive licensing rights to the technology. The cells will be grown in fermentation vessels, while being constantly fed oxygen, glucose and fermentation media. The cells will excrete malonic acid in the 101 hour fermentation process. -

Preparation of Amine Alanes
Europaisches Patentamt J European Patent Office 00 Publication number: 0 273 229 Office europeen des brevets A2 EUROPEAN PATENT APPLICATION © Application number: 87117901.6 © mt. ci.<: C01B 6/06 , C07C 87/00 © Date of filing: 03.12.87 © Priority: 22.12.86 US 945286 © Applicant: ETHYL CORPORATION Ethyl Tower 451 Florida Boulevard ® Date of publication of application: Baton Rouge Louisiana 70801 (US) 06.07.88 Bulletin 88/27 @ Inventor: Marlett, Everett Michael ® Designated Contracting States: 9355 Greenbriar Drive AT BE CH DE ES FR GB IT LI LU NL SE Baton Rouge Louisiana 7081 5(US) 0 Representative: Sandmair, Kurt, Dr. Patentanwalte Schwabe, Sandmair, Marx Stuntzstrasse 16 D-8000 MUnchen 80(DE) © Preparation of amine alanes. © Process for the preparation of an amine alane, said process comprising reacting: (a) an alkali metal aluminum tetrahydride having the formula MAIH4, wherein M is an alkali metal selected from the class consisting of lithium, sodium and potassium, (b) a hydrogen halide, and (c) a complexing tertiary amine. In this process, NaAlhU, HCI, and (C2H5)3N are preferred reactants. CM CM a. LU Xerox Copy Centre 0 273 229 PREPARATION OF AM1NE ALANES The invention pertains to the formation of amine alanes AIH3«NR3. These materials are useful as chemical intermediates. For example, they can be reacted with a silicon halide such as SiCU or SiF4 to produce silane. Reference is made to U.S. 4,474,743. It pertains to production of silane and aluminum fluoride from silicon tetrafluoride and amine alanes. Reference is also made to U.S. 4,006,095. It teaches, inter alia, that SiCU reacts with a toluene solution of AIH3 containing dimethyl ether or a stabilizing amine. -

Chem 341 • Organic Chemistry I Lecture Summary 07 • September 07, 2007
Chem 341 • Organic Chemistry I Lecture Summary 07 • September 07, 2007 Chapter 2 - Polar Covalent Bonds; Acids and Bases Alkyl Substituents When an alkane is a smaller part of a larger molecule (a substituent), it is called an ‘alkyl’ group. The parent name for the number of carbons remains the same, but it is given a ‘yl’ ending indicating it is a sub-part of the molecule. See for example several common names for some organic molecules shown below. Note that for chlorobutane, there are four different constitutional isomers possible. Normal butane refers to the straight chain. If the chlorine is moved to a secondary carbin it is called a secondary chloride. If the carbon skeleton is changed, it becomes an iso-butyl group and if the chlorine is attached to the tertiary carbon it is a tert-butyl group. These common names are still in use, though there is a more exact systematic naming system that is easier to understand (but longer names). H3C Cl OH methyl chloride ethyl alcohol Cl Cl Cl Cl n-butyl chloride sec-butyl chloride iso-butyl chloride tert-butyl chloride 1-chlorobutane 2-chlorobutane 1-chloro-2-methylpropane 2-chloro-2-methylpropane Degree of Alkyl Substitution We designate a kind of carbon (or kind of functional group attached to that carbon) according to how many other alkyl groups are attached to it. For example, a carbon on the end of a chain (with one alkyl group) would be a primary carbon. The hydrogens attached to that carbon would be called primary hydrogens. An alcohol attached to a primary carbon would be called a primary alcohol. -

WO 2013/089962 Al 20 June 2013 (20.06.2013) W P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2013/089962 Al 20 June 2013 (20.06.2013) W P O P C T (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every B01J 31/04 (2006.01) B01J 31/18 (2006.01) kind of national protection available): AE, AG, AL, AM, B01J 31/14 (2006.01) B01J 31/22 (2006.01) AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, (21) Number: International Application DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, PCT/US20 12/065285 HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, (22) International Filing Date: KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, 15 November 2012 (15.1 1.2012) ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, (25) Filing Language: English RW, SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, (26) Publication Language: English TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: 13/323,328 12 December 201 1 (12. 12.201 1) US (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (71) Applicant (for all designated States except US): CHEV¬ GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, SZ, TZ, RON PHILLIPS CHEMICAL COMPANY LP UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, TJ, [US/US]; 10001 Six Pines Drive, The Woodlands, Texas TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, 77380 (US). -

PESTICIDES Criteria for a Recommended Standard
CRITERIA FOR A RECOMMENDED STANDARD OCCUPATIONAL EXPOSURE DURING THE MANUFACTURE AND FORMULATION OF PESTICIDES criteria for a recommended standard... OCCUPATIONAL EXPOSURE DURING THE MANUFACTURE AND FORMULATION OF PESTICIDES * U.S. DEPARTMENT OF HEALTH, EDUCATION, AND WELFARE Public Health Service Center for Disease Control National Institute for Occupational Safety and Health July 1978 For sale by the Superintendent of Documents, U.S. Government Printing Office, Washington, D.C. 20402 DISCLAIMER Mention of company names or products does not constitute endorsement by the National Institute for Occupational Safety and Health. DHEW (NIOSH) Publication No. 78-174 PREFACE The Occupational Safety and Health Act of 1970 emphasizes the need for standards to protect the health and provide for the safety of workers occupationally exposed to an ever-increasing number of potential hazards. The National Institute for Occupational Safety and Health (NIOSH) has implemented a formal system of research, with priorities determined on the basis of specified indices, to provide relevant data from which valid criteria for effective standards can be derived. Recommended standards for occupational exposure, which are the result of this work, are based on the effects of exposure on health. The Secretary of Labor will weigh these recommendations along with other considerations, such as feasibility and means of implementation, in developing regulatory standards. Successive reports will be presented as research and epideiriologic studies are completed and as sampling and analytical methods are developed. Criteria and standards will be reviewed periodically to ensure continuing protection of workers. The contributions to this document on pesticide manufacturing and formulating industries by NIOSH staff members, the review consultants, the reviewer selected by the American Conference of Governmental Industrial Hygienists (ACGIH), other Federal agencies, and by Robert B. -

Chapter 16 the Chemistry of Benzene and Its Derivatives
Instructor Supplemental Solutions to Problems © 2010 Roberts and Company Publishers Chapter 16 The Chemistry of Benzene and Its Derivatives Solutions to In-Text Problems 16.1 (b) o-Diethylbenzene or 1,2-diethylbenzene (d) 2,4-Dichlorophenol (f) Benzylbenzene or (phenylmethyl)benzene (also commonly called diphenylmethane) 16.2 (b) (d) (f) (h) 16.3 Add about 25 °C per carbon relative to toluene (110.6 C; see text p. 743): (b) propylbenzene: 161 °C (actual: 159 °C) 16.4 The aromatic compound has NMR absorptions with greater chemical shift in each case because of the ring current (Fig. 16.2, text p. 745). (b) The chemical shift of the benzene protons is at considerably greater chemical shift because benzene is aromatic and 1,4-cyclohexadiene is not. 16.6 (b) Among other features, the NMR spectrum of 1-bromo-4-ethylbenzene has a typical ethyl quartet and a typical para-substitution pattern for the ring protons, as shown in Fig. 16.3, text p. 747, whereas the spectrum of (2- bromoethyl)benzene should show a pair of triplets for the methylene protons and a complex pattern for the ring protons. If this isn’t enough to distinguish the two compounds, the integral of the ring protons relative to the integral of the remaining protons is different in the two compounds. 16.7 (b) The IR spectrum indicates the presence of an OH group, and the chemical shift of the broad NMR resonance (d 6.0) suggests that this could be a phenol. The splitting patterns of the d 1.17 and d 2.58 resonances show that the compound also contains an ethyl group, and the splitting pattern of the ring protons shows that the compound is a para-disubstituted benzene derivative. -

Docw R S Acquired by ERIC Include Many Informal Unpublished * Material - T Available Fromother Sources
DOCUMENT RESUME ED 135 584 SE 021 575 AUTHOR Zdravkovich, V. TITLE Organic Chemistry Self Instructional Package 4: Alkanes-Nomenclature. INSTITUTION Prince George's Community Coll., Largo, Md. PUB DATE 76 NOTE 41p.; For related Packages 1-17, see SE 021 572-588; Not available in hard copy due to copyright restrictions AVAILABLE FROM Prince George's Community College Bookstore, Largo, Maryland 20870 ($17.00 a set, $1.00 each) EDRS PRICE MF-$0.83 Plus Postage. MC Not Available from EDRS. DESCRIPTORS *Autoinstructional Aids; *Chemistry; *College Science; Higher Education; *Independent Study; Individualized Instruction; Individualized Programs; *Organic Chemistry; Science Education; Self Help Programs IDENTIFIERS Alkanes; Prince Georges Community College ABSTRACT This booklet, one of a series of 17 developed at Prince George's Community College, largo, Maryland, provides an individualized, self-paced undergraduate organic chemistry instruction module designed to augment any course in organic chemistry but particularly those taught using the text "Organic Chemistry" by Morrison and Boyd. The entire series of modules covers the first 13 chapters of the Morrison-Boyd text in great detail. Each module has been provided with from one to three audiotapes, available from PrinOe George's Community College, to provide students additional explanations of particular concepts. Each module includes a self-evaluation exercise, a reference guide, worksheets to be completed with the audiotapes, answer sheets for the worksheets, a progress evaluation, an answer sheet for the progress evaluation, an answer sheet for the self-evaluation exercise, an introduction to the topic covered by the module, and student performance objectives for the module. The topic of this module is alkanes. -

Organic and Biological Chemistry
CHAPTER 23 Organic and Biological Chemistry CONTENTS HO H 23.1 ▶ Organic Molecules and Their C Structures: Alkanes H H C 23.2 ▶ Families of Organic Compounds: HO O O C C Functional Groups 23.3 ▶ Naming Organic Compounds H CC 23.4 ▶ Carbohydrates:HO A Biological Example HO OH of Isomers H 23.5 ▶ Valence Bond TCheory and Orbital OverlapH Pictures H C 23.6 ▶ Lipids:HO A Biological EOxample ofO Cis–Trans IsomerismC C 23.7 ▶ Formal Charge and Resonance in Organic CompoundsH CC 23.8 ▶ Conjugated SystemsHO OH 23.9 ▶ Proteins: A Biological Example of Conjugation 23.10 ▶ Aromatic Compounds and Molecular Ascorbic acid, also known as vitamin C, is an essential nutrient in the human diet Orbital Theory because it is not synthesized in our body. We can eat citrus fruits or take pills that contain vitamin C to maintain health. 23.11 ▶ Nucleic Acids: A Biological Example of Aromaticity ? Which Is Better, Natural or Synthetic? The answer to this question can be found in the INQUIRY ▶▶▶ on page 1021. STUDY GUIDE M23_MCMU3170_07_SE_C23.indd 978 27/11/14 2:11 AM f the ultimate goal of chemistry is to understand the world around us on a molecular level, then a knowledge of biochemistry—the chemistry of living organisms—is a central part Iof that goal. Biochemistry, in turn, is a branch of organic chemistry, a term originally used to mean the study of compounds from living organisms while inorganic chemistry was used for the study of compounds from nonliving sources. Today, however, we know that there are no fundamental differences between organic and inorganic compounds; the same principles apply to both. -

Synthesis of Insecticidal Mono- and Diacylhydrazines for Disruption of K+ Voltage-Gated Channels, Elucidation of Regiochemistry, And
Synthesis of Insecticidal Mono- and Diacylhydrazines for Disruption of K+ Voltage-Gated Channels, Elucidation of Regiochemistry, and Description of Conformational Isomerism by NMR Spectroscopy and Computation Joseph Shelby Clements II Dissertation submitted to the faculty of the Virginia Polytechnic Institute and State University in partial fulfillment of the requirements for the degree of Doctor of Philosophy In Chemistry Paul R. Carlier, Chair David G. I. Kingston James M. Tanko Gordon T. Yee May 2, 2017 Blacksburg, VA Keywords: Hydrazine, acylation, α-effect, regioselectivity, hydrazide, voltage- gated, (E)-/(Z)-, 19F NMR, single electron transfer (SET), rotational barrier, N-N bond rotation, rotational isomerism, helicity, transition state, dihedral angle Copyright 2017, Joseph S. Clements II Synthesis of Insecticidal Mono- and Diacylhydrazines for Disruption of K+ Voltage-Gated Channels, Elucidation of Regiochemistry, and Description of Conformational Isomerism by NMR Spectroscopy and Computation Joseph Shelby Clements II ABSTRACT Based on the success of diacyl-tert-butylhydrazines RH-5849 and RH-1266 in controlling agricultural crop pests, we endeavored to synthesize our own diacylbenzyl- and arylhydrazine derivatives for use against the malaria vector Anopheles gambiae. In the process of producing a library of compounds for assay against An. gambiae, it became clear that employing regioselective acylation techniques (in molecules that feature two nucleophilic, acyclic nitrogen atoms α to one another) would be imperative. Synthesis of the library derivatives proceeded rapidly and after topical assay, we found three compounds that were more toxic than the RH- series leads. One of the three displayed an LD50 value of half that of RH-1266, though patch clamp assay concluded that toxicity was not necessarily linked to inhibition of mosquito K+ channel Kv2.1. -
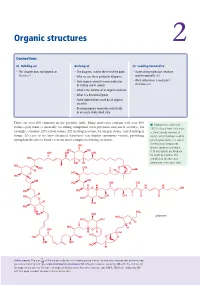
Organic Structures 2
Organic structures 2 Connections Building on Arriving at Looking forward to • This chapter does not depend on • The diagrams used in the rest of the book • Ascertaining molecular structure Chapter 1 • Why we use these particular diagrams spectroscopically ch3 • How organic chemists name molecules • What determines a molecule’s in writing and in speech structure ch4 • What is the skeleton of an organic molecule • What is a functional group • Some abbreviations used by all organic chemists • Drawing organic molecules realistically in an easily understood style There are over 100 elements in the periodic table. Many molecules contain well over 100 ■ Palytoxin was isolated in atoms—palytoxin (a naturally occurring compound with potential anticancer activity), for 1971 in Hawaii from Limu make example, contains 129 carbon atoms, 221 hydrogen atoms, 54 oxygen atoms, and 3 nitrogen o Hane (‘deadly seaweed of atoms. It’s easy to see how chemical structures can display enormous variety, providing Hana’), which had been used to enough molecules to build even the most complicated living creatures. poison spear points. It is one of the most toxic compounds OH known, requiring only about HO OH 0.15 micrograms per kilogram OH OH OH for death by injection. The HO complicated structure was O OH H determined a few years later. HO OH OH O H H O O HO HO H OH OH HO OH OH H H H H O N N OH HO HO H HO OH OH O O O HO OH H OH HO OH NH2 HO OH HO palytoxin HO OH O H HO H OH O H H H HO HO OH O O OH OH HO H Online support.