Porcn (NM 023638) Mouse Tagged ORF Clone Lentiviral Particle Product Data
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Dual Regulation of Planar Polarization by Secreted Wnts and Vangl2 in the Developing Mouse Cochlea Elvis Huarcaya Najarro1, Jennifer Huang1, Adrian Jacobo2, Lee A
© 2020. Published by The Company of Biologists Ltd | Development (2020) 147, dev191981. doi:10.1242/dev.191981 RESEARCH ARTICLE Dual regulation of planar polarization by secreted Wnts and Vangl2 in the developing mouse cochlea Elvis Huarcaya Najarro1, Jennifer Huang1, Adrian Jacobo2, Lee A. Quiruz1, Nicolas Grillet1 and Alan G. Cheng1,* ABSTRACT on the other. Flamingo (Fmi/Celsr1, Fmi/Celsr2 and Fmi/Celsr3) is Planar cell polarity (PCP) proteins localize asymmetrically to instruct present on both poles of the cell. Defective PCP signaling cell polarity within the tissue plane, with defects leading to deformities represented by a lack of polarized PCP components leads to of the limbs, neural tube and inner ear. Wnt proteins are evolutionarily congenital heart and tracheal abnormalities, skeletal dysplasia, conserved polarity cues, yet Wnt mutants display variable PCP neural tube defects as well as cochlear deformities (Butler and defects; thus, how Wnts regulate PCP remains unresolved. Here, we Wallingford, 2017; White et al., 2018). Despite their crucial roles, have used the developing cochlea as a model system to show that our understanding of upstream signals orchestrating PCP signaling secreted Wnts regulate PCP through polarizing a specific subset of is rather limited. PCP proteins. Conditional deletion of Wntless or porcupine, both of Wnt proteins have been implicated as upstream polarity cues for which are essential for secretion of Wnts, caused misrotated sensory PCP signaling. For example, limb morphogenesis in mice requires a cells and shortened cochlea – both hallmarks of PCP defects. gradient of Wnt5a, which has been reported to act as an instructive Wntless-deficient cochleae lacked the polarized PCP components cue to establish PCP (Gao et al., 2018, 2011). -

The Role of Genetics Mutations in Genes PORCN, TWIST2, HCCS In
ISSN: 2378-3672 Asadi and Jamali. Int J Immunol Immunother 2019, 6:038 DOI: 10.23937/2378-3672/1410038 Volume 6 | Issue 1 International Journal of Open Access Immunology and Immunotherapy REVIEW ARTICLE The Role of Genetics Mutations in Genes PORCN, TWIST2, HCCS in Goltz Syndrome Shahin Asadi1* and Mahsa Jamali2 Check for updates Division of Medical Genetics and Molecular Pathology Research, Molecular Genetics-IRAN-TABRIZ, Iran *Corresponding author: Shahin Asadi, Division of Medical Genetics and Molecular Pathology Research, Molecular Genetics-IRAN-TABRIZ, Iran are usually formed around the nose, the lips, the anal Abstract holes and the genitals of women with Goltz syndrome. Goltz syndrome (focal skin hypoplasia) is a genetic disorder In addition, papillomas may be present in the throat, that primarily affects the skin, skeletal system, eyes and face. People with Goltz syndrome have birth defects. These especially in the esophagus or larynx, and can cause disorders include very thin skin veins (skin hypoplasia), pink swallowing, respiration or sleep poisoning. The papillo- yellow nodules, subcutaneous fat, lack of upper skin layers mas can be removed if necessary by surgery from the (aplasia cutis), small clusters of superficial skin vessels growing regions. People with Goltz syndrome may have (telangiectasia), and veins in dark skin Or bright. Goltz syndrome is caused by mutation genes PORCN, TWIST2, small and abnormal nails on the toes. In addition, hair in HCCS. the scalp of these people can be weak or fragile or not [2] (Figure 2). Keywords Goltz syndrome, PORCN, TWIST2, HCCS genes, Skin and Many people with Goltz syndrome also have hand Skeletal disorders and foot disorders, including abnormalities such as oligodactyly in fingers and toes, fingers and legs (cin- Generalizations of Goltz Syndrome dactyly) or ecd rhodactyly. -

Based on Network Pharmacology Tools to Investigate the Molecular Mechanism of Cordyceps Sinensis on the Treatment of Diabetic Nephropathy
Hindawi Journal of Diabetes Research Volume 2021, Article ID 8891093, 12 pages https://doi.org/10.1155/2021/8891093 Research Article Based on Network Pharmacology Tools to Investigate the Molecular Mechanism of Cordyceps sinensis on the Treatment of Diabetic Nephropathy Yan Li,1 Lei Wang,2 Bojun Xu,1 Liangbin Zhao,1 Li Li,1 Keyang Xu,3 Anqi Tang,1 Shasha Zhou,1 Lu Song,1 Xiao Zhang,1 and Huakui Zhan 1 1Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu, 610072 Sichuan, China 2Key Laboratory of Chinese Internal Medicine of Ministry of Education and Dongzhimen Hospital, Beijing University of Chinese Medicine, Beijing 100700, China 3Zhejiang Chinese Medical University, Hangzhou, 310053 Zhejiang, China Correspondence should be addressed to Huakui Zhan; [email protected] Received 27 August 2020; Revised 17 January 2021; Accepted 24 January 2021; Published 8 February 2021 Academic Editor: Michaelangela Barbieri Copyright © 2021 Yan Li et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Background. Diabetic nephropathy (DN) is one of the most common complications of diabetes mellitus and is a major cause of end- stage kidney disease. Cordyceps sinensis (Cordyceps, Dong Chong Xia Cao) is a widely applied ingredient for treating patients with DN in China, while the molecular mechanisms remain unclear. This study is aimed at revealing the therapeutic mechanisms of Cordyceps in DN by undertaking a network pharmacology analysis. Materials and Methods. In this study, active ingredients and associated target proteins of Cordyceps sinensis were obtained via Traditional Chinese Medicine Systems Pharmacology Database (TCMSP) and Swiss Target Prediction platform, then reconfirmed by using PubChem databases. -

A Uniform Human Wnt Expression Library Reveals a Shared Secretory Pathway and Unique Signaling Activities
Differentiation 84 (2012) 203–213 Contents lists available at SciVerse ScienceDirect Differentiation journal homepage: www.elsevier.com/locate/diff A uniform human Wnt expression library reveals a shared secretory pathway and unique signaling activities Rani Najdi a, Kyle Proffitt b, Stephanie Sprowl a, Simran Kaur b, Jia Yu b, Tracy M. Covey b, David M. Virshup b,1, Marian L. Waterman a,n a Department of Microbiology and Molecular Genetics, University of California, Irvine, CA, USA b Program in Cancer and Stem Cell Biology, Duke-NUS Graduate Medical School, Singapore article info abstract Article history: Wnt ligands are secreted morphogens that control multiple developmental processes during embry- Received 24 April 2012 ogenesis and adult homeostasis. A diverse set of receptors and signals have been linked to individual Accepted 16 June 2012 Wnts, but the lack of tools for comparative analysis has limited the ability to determine which of these Available online 9 July 2012 signals are general for the entire Wnt family, and which define subsets of differently acting ligands. We Keywords: have created a versatile Gateway library of clones for all 19 human Wnts. An analysis comparing Wnt signaling epitope-tagged and untagged versions of each ligand shows that despite their similar expression at the b-catenin mRNA level, Wnts exhibit considerable variation in stability, processing and secretion. At least 14 out of Wnt modification and secretion the 19 Wnts activate b-catenin-dependent signaling, an activity that is cell type-dependent and tracks with the stabilization of b-catenin and LRP6 phosphorylation. We find that the core Wnt modification and secretion proteins Porcupine (PORCN) and Wntless (WLS) are essential for all Wnts to signal through b-catenin-dependent and independent pathways. -

Tumour-Stroma Signalling in Cancer Cell Motility and Metastasis
Tumour-Stroma Signalling in Cancer Cell Motility and Metastasis by Valbona Luga A thesis submitted in conformity with the requirements for the degree of Doctor of Philosophy, Department of Molecular Genetics, University of Toronto © Copyright by Valbona Luga, 2013 Tumour-Stroma Signalling in Cancer Cell Motility and Metastasis Valbona Luga Doctor of Philosophy Department of Molecular Genetics University of Toronto 2013 Abstract The tumour-associated stroma, consisting of fibroblasts, inflammatory cells, vasculature and extracellular matrix proteins, plays a critical role in tumour growth, but how it regulates cancer cell migration and metastasis is poorly understood. The Wnt-planar cell polarity (PCP) pathway regulates convergent extension movements in vertebrate development. However, it is unclear whether this pathway also functions in cancer cell migration. In addition, the factors that mobilize long-range signalling of Wnt morphogens, which are tightly associated with the plasma membrane, have yet to be completely characterized. Here, I show that fibroblasts secrete membrane microvesicles of endocytic origin, termed exosomes, which promote tumour cell protrusive activity, motility and metastasis via the exosome component Cd81. In addition, I demonstrate that fibroblast exosomes activate autocrine Wnt-PCP signalling in breast cancer cells as detected by the association of Wnt with Fzd receptors and the asymmetric distribution of Fzd-Dvl and Vangl-Pk complexes in exosome-stimulated cancer cell protrusive structures. Moreover, I show that Pk expression in breast cancer cells is essential for fibroblast-stimulated cancer cell metastasis. Lastly, I reveal that trafficking in cancer cells promotes tethering of autocrine Wnt11 to fibroblast exosomes. These studies further our understanding of the role of ii the tumour-associated stroma in cancer metastasis and bring us closer to a more targeted approach for the treatment of cancer spread. -
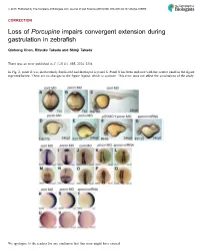
Porcupine Impairs Convergent Extension During Gastrulation in Zebrafish
ß 2015. Published by The Company of Biologists Ltd | Journal of Cell Science (2015) 000, 000–000 doi:10.1242/jcs.168989 CORRECTION Loss of Porcupine impairs convergent extension during gastrulation in zebrafish Qiuhong Chen, Ritsuko Takada and Shinji Takada There was an error published in J. Cell Sci. 125, 2224–2234. In Fig. 2, panel R was inadvertently duplicated and displayed as panel S. Panel S has been replaced with the correct panel in the figure reprinted below. There are no changes to the figure legend, which is accurate. This error does not affect the conclusions of the study. We apologise to the readers for any confusion that this error might have caused. 1 Journal of Cell Science JCS168989.3d 22/1/15 10:57:22 The Charlesworth Group, Wakefield +44(0)1924 369598 - Rev 9.0.225/W (Oct 13 2006) 2224 Research Article Loss of Porcupine impairs convergent extension during gastrulation in zebrafish Qiuhong Chen1, Ritsuko Takada1 and Shinji Takada1,2,* 1Okazaki Institute for Integrative Bioscience and National Institute for Basic Biology, National Institutes of Natural Sciences, Okazaki, Aichi 444-8787, Japan 2Department of Molecular Biomechanics, The Graduate University for Advanced Studies (SOKENDAI), Okazaki, Aichi 444-8787, Japan *Author for correspondence ([email protected]) Accepted 9 January 2012 Journal of Cell Science 125, 2224–2234 ß 2012. Published by The Company of Biologists Ltd doi: 10.1242/jcs.098368 Summary Porcupine (Porcn), an O-acyltransferase located in the endoplasmic reticulum (ER), is required for lipidation of Wnt proteins to enable their trafficking from the ER in mammalian cell culture. -

Microrna-124-3P Suppresses Mouse Lip Mesenchymal Cell Proliferation
Suzuki et al. BMC Genomics (2019) 20:852 https://doi.org/10.1186/s12864-019-6238-4 RESEARCH ARTICLE Open Access MicroRNA-124-3p suppresses mouse lip mesenchymal cell proliferation through the regulation of genes associated with cleft lip in the mouse Akiko Suzuki1,2, Hiroki Yoshioka1,2, Dima Summakia1, Neha G. Desai1,3, Goo Jun3,4, Peilin Jia5, David S. Loose4,6, Kenichi Ogata1,2, Mona V. Gajera1,3, Zhongming Zhao3,4,5 and Junichi Iwata1,2,4* Abstract Background: Cleft lip (CL), one of the most common congenital birth defects, shows considerable geographic and ethnic variation, with contribution of both genetic and environmental factors. Mouse genetic studies have identified several CL-associated genes. However, it remains elusive how these CL-associated genes are regulated and involved in CL. Environmental factors may regulate these genes at the post-transcriptional level through the regulation of non-coding microRNAs (miRNAs). In this study, we sought to identify miRNAs associated with CL in mice. Results: Through a systematic literature review and a Mouse Genome Informatics (MGI) database search, we identified 55 genes that were associated with CL in mice. Subsequent bioinformatic analysis of these genes predicted that a total of 33 miRNAs target multiple CL-associated genes, with 20 CL-associated genes being potentially regulated by multiple miRNAs. To experimentally validate miRNA function in cell proliferation, we conducted cell proliferation/viability assays for the selected five candidate miRNAs (miR-124-3p, let-7a-5p, let-7b-5p, let-7c-5p, and let-7d-5p). Overexpression of miR-124-3p, but not of the others, inhibited cell proliferation through suppression of CL-associated genes in cultured mouse embryonic lip mesenchymal cells (MELM cells) isolated from the developing mouse lip region. -

Expanding the Phenotypic Spectrum of PORCN Variants in Two Males with Syndromic Microphthalmia
European Journal of Human Genetics (2015) 23, 551–554 & 2015 Macmillan Publishers Limited All rights reserved 1018-4813/15 www.nature.com/ejhg SHORT REPORT Expanding the phenotypic spectrum of PORCN variants in two males with syndromic microphthalmia Paul D Brady1, Hilde Van Esch1, Nathalie Fieremans1,GuyFroyen1, Anne Slavotinek2, Jan Deprest3,4, Koenraad Devriendt1 and Joris R Vermeesch*,1 Variants in PORCN are a cause of Goltz-Gorlin syndrome or Focal Dermal Hypoplasia, an X-linked dominant disorder affecting heterozygous females and until now considered to be embryonic lethal in males. Exome sequencing was performed in a family in which two male siblings were characterized by microphthalmia and additional congenital anomalies including diaphragmatic hernia, spina bifida and cardiac defects. Surprisingly, we identified a maternally inherited variant in PORCN present in both males as well as in two female siblings. This represents the first finding of a PORCN variant in non-mosaic males affected with Goltz-Gorlin syndrome. The apparently asymptomatic mother showed extreme skewing of X-inactivation (90%), an asymptomatic female sibling showed skewing of 88%, and the second female sibling affected with cutis aplasia of the scalp showed X-inactivation considered within the normal range. European Journal of Human Genetics (2015) 23, 551–554; doi:10.1038/ejhg.2014.135; published online 16 July 2014 INTRODUCTION The skin was normal and no other digital or skeletal abnormalities were Microphthalmia occurs with an incidence of 0.9–2.3 per 10 000,1 and observed. He was deceased at day 0, with confirmation of the clinical findings is estimated to occur in association with additional congenital by pathology examination. -

Variation in Protein Coding Genes Identifies Information Flow
bioRxiv preprint doi: https://doi.org/10.1101/679456; this version posted June 21, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Animal complexity and information flow 1 1 2 3 4 5 Variation in protein coding genes identifies information flow as a contributor to 6 animal complexity 7 8 Jack Dean, Daniela Lopes Cardoso and Colin Sharpe* 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 Institute of Biological and Biomedical Sciences 25 School of Biological Science 26 University of Portsmouth, 27 Portsmouth, UK 28 PO16 7YH 29 30 * Author for correspondence 31 [email protected] 32 33 Orcid numbers: 34 DLC: 0000-0003-2683-1745 35 CS: 0000-0002-5022-0840 36 37 38 39 40 41 42 43 44 45 46 47 48 49 Abstract bioRxiv preprint doi: https://doi.org/10.1101/679456; this version posted June 21, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Animal complexity and information flow 2 1 Across the metazoans there is a trend towards greater organismal complexity. How 2 complexity is generated, however, is uncertain. Since C.elegans and humans have 3 approximately the same number of genes, the explanation will depend on how genes are 4 used, rather than their absolute number. -

Deletion of Mouse Porcn Blocks Wnt Ligand Secretion and Reveals an Ectodermal Etiology of Human Focal Dermal Hypoplasia/Goltz Syndrome
Deletion of mouse Porcn blocks Wnt ligand secretion and reveals an ectodermal etiology of human focal dermal hypoplasia/Goltz syndrome Jared J. Barrotta, Gabriela M. Casha, Aaron P. Smithb, Jeffery R. Barrowb, and L. Charles Murtaugha,1 aDepartment of Human Genetics, University of Utah, Salt Lake City, UT 84112; and bDepartment of Physiology and Developmental Biology, Brigham Young University, Provo, UT 84602 Edited* by Clifford J. Tabin, Harvard Medical School, Boston, MA, and approved June 16, 2011 (received for review May 12, 2010) The Drosophila porcupine gene is required for secretion of wingless drome is never transmitted to male offspring, suggesting male- and other Wnt proteins, and sporadic mutations in its unique specific embryonic lethality. The congenital abnormalities asso- human ortholog, PORCN, cause a pleiotropic X-linked dominant ciated with FDH are highly pleiotropic and variable, including disorder, focal dermal hypoplasia (FDH, also known as Goltz syn- multiple aspects of skin and skeletal development (20), and con- drome). We generated a conditional allele of the X-linked mouse siderably overlap defects observed in mouse Wnt pathway Porcn gene and analyzed its requirement in Wnt signaling and em- mutants (Table S1). Given the connections between porcupine/ bryonic development. We find that Porcn-deficient cells exhibit Porcn and Wnt signaling, we developed a conditional allele of a cell-autonomous defect in Wnt ligand secretion but remain re- mouse Porcn that provides a unique genetic tool to block Wnt sponsive to exogenous Wnts. Consistent with the female-specific ligand biogenesis. inheritance pattern of FDH, Porcn hemizygous male embryos arrest during early embryogenesis and fail to generate mesoderm, a phe- Results notype previously associated with loss of Wnt activity. -

PORCN Gene Porcupine O-Acyltransferase
PORCN gene porcupine O-acyltransferase Normal Function The PORCN gene provides instructions for making a protein that belongs to a group of related proteins called the porcupine (Porc) family. Although the precise function of the PORCN protein is unknown, proteins in the Porc family are involved in the process of transferring a molecule called palmitoleic acid to Wnt proteins. Wnt proteins participate in chemical signaling pathways in the body and play critical roles in development before birth. Members of the Porc family are located in the endoplasmic reticulum, which is a structure inside the cell that is involved in protein processing and transport. The transfer of palmitoleic acid to Wnt proteins facilitates the release of these proteins from the cell so they can regulate development of the skin, bones, and other structures. Researchers are working to determine the specific role of the PORCN protein within human cells. Health Conditions Related to Genetic Changes Focal dermal hypoplasia At least 29 mutations in the PORCN gene have been found to cause focal dermal hypoplasia. These mutations may alter the protein's structure, lead to the production of an abnormally short version of the protein, or delete the entire PORCN gene. All of these mutations appear to result in the absence of any functional PORCN protein. Researchers believe Wnt proteins cannot be released from the cell without the PORCN protein. When Wnt proteins are unable to leave the cell, they cannot participate in the chemical signaling pathways that are critical for normal -

RNF43 Is Frequently Mutated in Colorectal and Endometrial Cancers
RNF43 is frequently mutated in colorectal and endometrial cancers The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation Giannakis, Marios et al. “RNF43 Is Frequently Mutated in Colorectal and Endometrial Cancers.” Nature Genetics 46, 12 (October 2014): 1264–1266 © 2014 Nature America, Inc As Published http://dx.doi.org/10.1038/NG.3127 Publisher Nature Publishing Group Version Author's final manuscript Citable link http://hdl.handle.net/1721.1/116687 Terms of Use Article is made available in accordance with the publisher's policy and may be subject to US copyright law. Please refer to the publisher's site for terms of use. NIH Public Access Author Manuscript Nat Genet. Author manuscript; available in PMC 2015 June 01. NIH-PA Author ManuscriptPublished NIH-PA Author Manuscript in final edited NIH-PA Author Manuscript form as: Nat Genet. 2014 December ; 46(12): 1264–1266. doi:10.1038/ng.3127. RNF43 is frequently mutated in colorectal and endometrial cancers Marios Giannakis1,2,3,*, Eran Hodis1,3,4,5,*, Xinmeng Jasmine Mu1,3, Mai Yamauchi1, Joseph Rosenbluh1,3, Kristian Cibulskis3, Gordon Saksena3, Michael S. Lawrence3, ZhiRong Qian1, Reiko Nishihara1,6,7,8, Eliezer M. Van Allen1,2,3, William C. Hahn1,2,3, Stacey B. Gabriel3, Eric S. Lander3,9,10, Gad Getz3,11, Shuji Ogino1,6,12, Charles S. Fuchs1,13, and Levi A. Garraway1,2,3 1Department of Medical Oncology, Dana-Farber Cancer Institute, Harvard Medical School, Boston, Massachusetts, USA 2Department of Medicine, Brigham