1 Office Safe Handling of Cryogenic Materials Guidelines
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Liquid Argon
Safetygram 8 Liquid argon Liquid argon is tasteless, colorless, odorless, noncorrosive, nonflammable, and extremely cold. Belonging to the family of rare gases, argon is the most plentiful, making up approximately 1% of the earth’s atmosphere. It is monatomic and extremely inert, forming no known chemical compounds. Since argon is inert, special materials of construction are not required. However, materials of construction must be selected to withstand the low temperature of liquid argon. Vessels and piping should be designed to American Society of Mechanical Engineers (ASME) specifications or the Department of Transportation (DOT) codes for the pressures and temperatures involved. Although used more commonly in the gaseous state, argon is commonly stored and transported as a liquid, affording a more cost-effective way of providing product supply. Liquid argon is a cryogenic liquid. Cryogenic liquids are liquefied gases that have a normal boiling point below –130°F (–90°C). Liquid argon has a boiling point of –303°F (–186°C). The temperature difference between the product and the surrounding environment, even in winter, is substantial. Keeping this surrounding heat from the product requires special equipment to store and handle cryogenic liquids. A typical system consists of the following components: a cryogenic storage tank, one or more vaporizers, a pressure control system, and all of the piping required for fill, vaporization. The cryogenic tank is constructed, in principle, like a vacuum bottle. It is designed to keep heat away from the liquid that is contained in the inner vessel. Vaporizers convert the liquid argon to its gaseous state. A pressure control manifold controls the pressure at which the gas is fed to the process. -

Cryogenic Liquid Nitrogen Vehicles (ZEV's)
International Journal of Scientific and Research Publications, Volume 6, Issue 9, September 2016 562 ISSN 2250-3153 Cryogenic Liquid Nitrogen Vehicles (ZEV’S) K J Yogesh Department Of Mechanical Engineering, Jain Engineering College, Belagavi Abstract- As a result of widely increasing air pollution available zero emission vehicle (ZEV) meeting it's standards are throughout the world & vehicle emissions having a major the electrically recharged ones, however these vehicles are also contribution towards the same, it makes its very essential to not a great success in the society due to its own limitations like engineer or design an alternative to the present traditional initial cost, slow recharge, speeds etc. Lead acid & Ni-Cd gasoline vehicles. Liquid nitrogen fueled vehicles can act as an batteries are the past of major technologies in the electric excellent alternative for the same. Liquefied N2 at cryogenic vehicles. They exhibit specific energy in the range of 30-40 W- temperatures can replace conventional fuels in cryogenic heat hr/kg. Lead- acid batteries take hours to recharge & the major engines used as a propellant. The ambient temperature of the drawback of the batteries in all the cases is their replacement surrounding vaporizes the liquid form of N2 under pressure & periodically. This directly/indirectly increases the operating cost leads to the formation of compressed N2 gas. This gas actuates a when studied carefully & thereby not 100% acceptable. pneumatic motor. A combination of multiple reheat open Recent studies make it clear that the vehicles using liquid Rankine cycle & closed Brayton cycle are involved in the nitrogen as their means provide an excellent alternative before process to make use of liquid N2 as a non-polluting fuel. -

Capacity Enhancement of Indigenous Expansion Engine Based Helium Liquefier
IOP Conference Series: Materials Science and Engineering PAPER • OPEN ACCESS Capacity enhancement of indigenous expansion engine based helium liquefier To cite this article: R S Doohan et al 2017 IOP Conf. Ser.: Mater. Sci. Eng. 171 012011 View the article online for updates and enhancements. This content was downloaded from IP address 170.106.202.8 on 27/09/2021 at 00:16 ICECICMC IOP Publishing IOP Conf. Series: Materials Science and Engineering 171 (2017) 012011 doi:10.1088/1757-899X/171/1/012011 International Conference on Recent Trends in Physics 2016 (ICRTP2016) IOP Publishing Journal of Physics: Conference Series 755 (2016) 011001 doi:10.1088/1742-6596/755/1/011001 Capacity enhancement of indigenous expansion engine based helium liquefier R S Doohan1, P K Kush1, G Maheshwari2 1Raja Ramanna Centre for Advanced Technology, Indore, Madhya Pradesh India. 2Institute of Engineering and Technology, DAVV, Indore, Madhya Pradesh, India. E-mail: [email protected] Abstract. Development of technology and understanding for large capacity helium refrigeration and liquefaction at helium temperature is indispensable for coming-up projects. A new version of helium liquefier designed and built to provide approximately 35 liters of liquid helium per hour. The refrigeration capacity of this reciprocating type expansion engine machine has been increased from its predecessor version with continuous improvement and deficiency debugging. The helium liquefier has been built using components by local industries including cryogenic Aluminum plate fin heat exchangers. Two compressors with nearly identical capacity have been deployed for the operation of system. Together they consume about 110 kW of electric power. The system employs liquid Nitrogen precooling to enhance liquid Helium yield. -
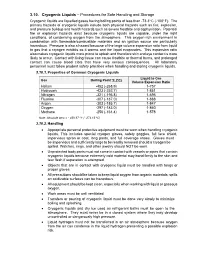
3.10. Cryogenic Liquids – Procedures for Safe Handling and Storage Cryogenic Liquids Are Liquefied Gases Having Boiling Points of Less Than -73.3O C (-100O F)
3.10. Cryogenic Liquids – Procedures for Safe Handling and Storage Cryogenic liquids are liquefied gases having boiling points of less than -73.3o C (-100o F). The primary hazards of cryogenic liquids include both physical hazards such as fire, explosion, and pressure buildup and health hazards such as severe frostbite and asphyxiation. Potential fire or explosion hazards exist because cryogenic liquids are capable, under the right conditions, of condensing oxygen from the atmosphere. This oxygen-rich environment in combination with flammable/combustible materials and an ignition source are particularly hazardous. Pressure is also a hazard because of the large volume expansion ratio from liquid to gas that a cryogen exhibits as it warms and the liquid evaporates. This expansion ratio also makes cryogenic liquids more prone to splash and therefore skin and eye contact is more likely to occur. Contact with living tissue can cause frostbite or thermal burns, and prolonged contact can cause blood clots that have very serious consequences. All laboratory personnel must follow prudent safety practices when handling and storing cryogenic liquids. 3.10.1. Properties of Common Cryogenic Liquids Liquid to Gas Gas Boiling Point oF (oC) Volume Expansion Ratio Helium -452 (-268.9) 1-757 Hydrogen -423 (-252.7) 1-851 Nitrogen -321 (-195.8) 1-696 Fluorine -307 (-187.0) 1-888 Argon -303 (-185.7) 1-847 Oxygen -297 (-183.0) 1-860 Methane -256 (-161.4) 1-578 Note: Absolute zero = - 459.67 oF (- 273.15 oC) 3.10.2. Handling Appropriate personal protective equipment must be worn when handling cryogenic liquids. -

Cryogenic Liquid Safety
Cryogenic Liquid Safety Hazards and Safe Handling of Cryogenic Liquids Cryogenic liquids have boiling points less than -73ºC (-100ºF). Liquid nitrogen, liquid oxygen and carbon dioxide are the most common cryogenic materials used in the laboratory. Hazards may include fire, explosion, embrittlement, pressure buildup, frostbite and asphyxiation. Many of the safety precautions observed for compressed gases also apply to cryogenic liquids. There are also additional hazards due to the unique properties of cryogenic liquids: • Extremely Low Temperatures: The cold boil-off vapor of cryogenic liquids rapidly freezes human tissue. Most metals become stronger upon exposure to cold temperatures, but materials, such as; carbon steel, plastics and rubber become brittle or fracture under stress at these temperatures. Proper material selection is important. Cold burns and frostbite caused by cryogenic liquids can result in extensive tissue damage. • Vaporization: All cryogenic liquids produce large volumes of gas when they vaporize. Liquid nitrogen will expand 696 times as it vaporizes. The expansion ratio of argon is 847:1, hydrogen is 851:1 and oxygen is 862:1. If these liquids vaporize in a sealed container, they can produce enormous pressures that could rupture the vessel. For this reason, pressurized cryogenic containers are usually protected with multiple pressure relief devices. • Vaporization of cryogenic liquids (except oxygen) in an enclosed area can cause asphyxiation! • Vaporization of liquid O2 can produce an oxygen-rich atmosphere, supporting and accelerating combustion of other materials. Vaporization of liquid hydrogen can form an extremely flammable mixture with air. Most cryogenic liquids are odorless, colorless, and tasteless when vaporized. When cryogenic liquids are exposed to the atmosphere, cold boil-off gases condense the moisture in the air, creating a highly visible fog. -

Liquid Helium
Safetygram 22 Liquid helium Liquid helium is inert, colorless, odorless, noncorrosive, extremely cold, and nonflammable. Helium will not react with other elements or compounds under ordinary conditions. Since helium is noncorrosive, special materials of construction are not required. However, materials must be suitable for use at the extremely low temperatures of liquid helium. Vessels and piping must be selected and designed to withstand the pressure and temperatures involved and comply with applicable codes for transport and use. Manufacture Most of commercial helium is recovered from natural gas through a cryogenic separation process. Normally, helium is present in less than 1% by volume in natural gas. Helium is recovered, refined, and liquefied. Liquid helium is typically shipped from production sources to storage and transfill facilities. Tankers, ranging in size from 5,000 to 11,000 gallons, contain an annular space insulated with vacuum, nitrogen shielding, and multilayer insulation. This de- sign reduces heat leak and vaporization of liquid helium during transportation. Uses The extremely low temperature of liquid helium is utilized to maintain the superconducting properties of magnets in applications such as MRI, NMR spectroscopy, and particle physics research. The main application for gas- eous helium is for inert shielding gas in metal arc and laser welding. Helium provides a protective atmosphere in the production of reactive metals, such as titanium and zirconium. Gaseous helium is used as a coolant during the draw- ing of optical fibers, as a carrier gas for chromatography, and as a leak detection gas in a variety of industries. Being both lighter than air and nonflammable, helium is used to inflate balloons and airships. -

Liquid Nitrogen Safety & Handling
Liquid Nitrogen Safety & Handling 1.0 Liquid Nitrogen & Cryogenic Liquids a. Liquid nitrogen, LN2, is colorless, odorless and tasteless. It is non-flammable, physiologically inert and non-toxic. It is an extremely cold liquid with a boiling point of - 196ºC = -320ºF. b. Cryogenic liquids are liquefied gases that are kept in their liquid state at extremely low temperatures. Cryogenic liquids have boiling points below -150°C (-238°F) (Carbon dioxide and nitrous oxide, which have slightly higher boiling points are sometimes included in this category). All cryogenic liquids are gases at normal temperatures and pressures. All Cryogenic liquids produce large amounts of gas when they vaporize. c. There a many applications of LN2 in a biomedical research facility, one of the most common being cryopreservation, the storage of cells at temperatures below -130 ºC, which is essential in order to preserve biological material unaltered. 2.0 Safety Hazards Associated with Handling LN2 a. The extremely low temperature of the liquid can cause severe frostbite or eye damage upon contact. Symptoms of frostbite include change in skin color to white or grayish yellow and the pain after contact with LN2 may quickly subside. Items in contact with liquid nitrogen become extremely cold. Touching these items may result in torn flesh. Many substances become brittle upon contact with liquid nitrogen and may shatter when cold (such as common glass and large solid plastics), sending pieces of the material flying. b. Upon vaporization it expands by a factor of 700; one liter of liquid nitrogen becomes 24.6 cubic feet of nitrogen gas. -

Liquid Oxygen
Safetygram 6 Liquid oxygen Oxygen is the second largest component of the atmosphere, comprising 20.8% by volume. Liquid oxygen is pale blue and extremely cold. Although nonflammable, oxygen is a strong oxidizer. Oxygen is necessary to support life. Oxygen will react with nearly all organic materials and metals, usually forming an oxide. Materials that burn in air will burn more vigorously in oxygen. Equipment used in oxygen service must meet stringent cleaning requirements, and systems must be constructed of materials that have high ignition temperatures and that are nonreactive with oxygen under the service conditions. Vessels should be manufactured to American Society of Mechanical Engineers (ASME) codes and designed to withstand the process temperatures and pressures. Liquid oxygen is a cryogenic liquid. Cryogenic liquids are liquefied gases that have a normal boiling point below –130°F (–90°C). Liquid oxygen has a boiling point of –297°F (–183°C). Because the temperature difference between the product and the surrounding environment is substantial—even in the winter—keeping liquid oxygen insulated from the surrounding heat is essential. The product also requires special equipment for handling and storage. Oxygen is often stored as a liquid, although it is used primarily as a gas. Liquid storage is less bulky and less costly than the equivalent capacity of high-pressure gaseous storage. A typical storage system consists of a cryogenic storage tank, one or more vaporizers and a pressure control system. The cryogenic tank is constructed, in principle, like a vacuum bottle. There is an inner vessel surrounded by an outer vessel. Between the vessels is an annular space that contains an insulating medium from which all the air has been removed. -

Cryogenic Liquid Containers
Safetygram 27 Cryogenic liquid containers Cryogenic liquid containers, also referred to as liquid cylinders, are double-walled vacuum vessels with multilayer insulation in the annular space. They are designed for the reliable and economic transportation and storage of liquefied gases at cryogenic temperatures, typically colder than –130°F (–90°C). There are two primary advantages of a liquid container. The first is that it contains a large volume of gas at a relatively low pressure compared to a compressed gas cylinder. The second is that it provides a source of cryogenic liquids which can be easily handled. Cryogenic liquid containers are often incorrectly referred to as dewars. Dewars are open, nonpressurized vessels for holding cryogenic liquids. The cryogenic products normally found in liquid containers are liquid nitrogen (LIN), liquid argon (LAR), liquid oxygen (LOX), and liquid helium (LHE). Carbon dioxide and nitrous oxide are also available as refrigerated liquids in similar containers. Although these containers are well insulated, heat will continuously leak into the product, due to the extremely large temperature difference between the cryogenic liquid and the ambient environment. The heat leak will cause some vaporization to occur. Vaporized product, if not used, will collect in the vapor space above the liquid and build pressure. This is referred to as head pressure. The head pressure will build in the container and periodically vent via the pressure relief valve. Vaporization rates will vary and may be as low as 0.4% or as high as 3% of the container’s volume per day. This is a normal and safe function of the container. -

Thermodynamic Analysis on an Integrated Liquefied Air Energy
energies Article Thermodynamic Analysis on an Integrated Liquefied Air Energy Storage and Electricity Generation System Yingbai Xie and Xiaodong Xue * Department of Power Engineering, North China Electric Power University, Baoding 071003, China; [email protected] * Correspondence: [email protected] Received: 22 August 2018; Accepted: 20 September 2018; Published: 23 September 2018 Abstract: For an integrated liquefied air energy storage and electricity generation system, mathematical models of the liquefied air energy storage and electricity generation process are established using a thermodynamic theory. The effects of the outlet pressure of the compressor unit, the outlet pressure of the cryogenic pump, the heat exchanger effectiveness, the initial air temperature and pressure before throttling on the performances of the integrated liquefied air energy storage, and the electricity generation system are investigated, using the cycle efficiency and liquid air yield ratio as the evaluation indexes. The results show that if the compressor outlet pressure is raised, both the compression work and the expansion work increase, but because the expansion work increases more slowly, the cycle efficiency of the system gradually decreases. Increasing the cryogenic pump outlet pressure and heat exchanger effectiveness can significantly increase the cycle efficiency of the system; the higher the air pressure and the lower the air temperature before throttling, the greater the liquid air yield after expansion, and the higher the cycle efficiency. The theoretical analysis models and research results can provide a reference for the development of an integrated system of liquefied air energy storage and electricity production, as well as for the development of medium-capacity energy storage technology. -

Cryogenic Liquid Containers
Cryogenic Liquid Containers Cryogenic liquid containers, also referred All cryogenic liquids produce large vol- If liquid oxygen is released, an oxygen- to as liquid cylinders, are designed for the umes of gas when they vaporize. The enriched atmosphere could result. An oxy- reliable and economic transportation and expansion ratio is the amount of gas gen- gen-enriched atmosphere is dangerous storage of liquefied gases at cryogenic erated from a given amount of liquid. Table due to the increased potential for a fire to temperatures, typically colder than – 130ºF 1 shows the liquid-to-gas expansion ratios occur. Although oxygen itself is nonflam- (-90ºC). The products found in liquid con- for the common cryogenic fluids. If a suffi- mable, ordinary combustible materials will tainers are nitrogen, argon, oxygen, helium, cient amount of liquid is vaporized within a burn more vigorously. Materials that nor- carbon dioxide and nitrous oxide. The con- closed container, it will produce enormous mally do not burn in air may burn in an tainers are double-walled, vacuum vessels pressures that could rupture the vessel. oxygen-enriched atmosphere. with multilayer insulation in the annular For this reason, cryogenic liquid containers space. The two primary advantages of a are protected with multiple pressure relief Elevator Transport liquid container are that it contains a large devices. Similarly, any system for the stor- Section to Be Modified please come back volume of gas at a relatively low pressure age and delivery of cryogenic liquids later. and it provides a source of cryogenic liq- should be carefully designed to avoid trap- uids which can be easily handled. -

Cryogenic Liquid / Solid Safety
SEPS Guidance Note (GN-CHEM07/2018) Cryogenic Liquid / Solid Safety General Introduction Cryogenic liquids and solids including liquid nitrogen, liquid helium, liquid carbon dioxide and dry ice (solid carbon dioxide) are commonly used in laboratories and other areas across the university as refrigerants, sources of dry gases and for many other reasons. Despite the common usage of cryogenic materials, the hazards associated with this substance are not always fully appreciated by everyone who might have cause to enter areas where cryogenic materials are in use. This guidance note is designed to inform staff and students of the physical properties of cryogenic substances, the hazards associated with storage and use and the basic safety procedures which can be applied to reduce the risk. This note focusses on the hazards associated with liquid nitrogen as one of the most common (and hazardous) cryogenic materials used across the institution but the principles within can be applied to most other cryogens and where safety procedures may differ for certain substances these will be highlighted in the text. Characteristics of Liquid Nitrogen Nitrogen gas forms the bulk of the atmosphere around us, comprising almost 80% of the air we breathe. It is colourless, odourless and tasteless and therefore under normal circumstances cannot be detected by human senses. It is non-combustible, non-toxic and inert to most chemicals. While nitrogen cannot be liquefied by compression alone at room temperature it can be condensed to a liquid if cooled below its normal boiling point of 196°C and will remain in the form of a colourless liquid so long as it is stored below this temperature.