Liquid Helium
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Liquid Helium Variable Temperature Research Dewars
CRYO Variable Temperature Liquid Helium Research Dewars CRYO Variable Temperature Liquid Helium Research Dewars Cryo Industries Variable Temperature Liquid Helium Research Dewars (CN Series) provide soluitions for an extensive variety of low temperature optical and non-optical requirements. There are numerous designs available, including Sample in Flowing Vapor, Sample in Vacuum and Sample in Exchange Gas. Our most popular models features Sample in Flowing Vapor (dynamic exchange gas), where the sample is cooled by insertion into flowing helium gas exiting from the vaporizer (also known as the diffuser or heat exchanger). The samples are top loading and can be quickly changed while operating. The temperature of the sample can be varied from typically less than 1.4 K to room temperature. Liquid helium flows from the reservoir through the adjustable flow valve down to the vaporizer located at the bottom of the sample tube. Applying heat, vaporizes the liquid and raises the gas temperature. This gas enters the sample zone to cool the sample to your selected temperature. Pumping on the sample zone will provide temperatures below 2 K with either sample in vapor or immersed in liquid. No inefficient liquid helium reservoir pumping is required. The system uses enthalpy (heat capacity) of the helium vapor which results in very high power handling, fast temperature change, ultra stable temperatures, ease of use an much more - Super Variable Temperature. Optical ‘cold’ windows are normally epoxy sealed, strain relief mounted into indium sealed mounts or direct indium mounted. Window seals are reliable and fully guaranteed. For experiments where flowing vapor may be undesirable (such as mossbauer or infrared detectors), static exchange gas cooling and sample in vacuum inserts are available. -

Liquid Argon
Safetygram 8 Liquid argon Liquid argon is tasteless, colorless, odorless, noncorrosive, nonflammable, and extremely cold. Belonging to the family of rare gases, argon is the most plentiful, making up approximately 1% of the earth’s atmosphere. It is monatomic and extremely inert, forming no known chemical compounds. Since argon is inert, special materials of construction are not required. However, materials of construction must be selected to withstand the low temperature of liquid argon. Vessels and piping should be designed to American Society of Mechanical Engineers (ASME) specifications or the Department of Transportation (DOT) codes for the pressures and temperatures involved. Although used more commonly in the gaseous state, argon is commonly stored and transported as a liquid, affording a more cost-effective way of providing product supply. Liquid argon is a cryogenic liquid. Cryogenic liquids are liquefied gases that have a normal boiling point below –130°F (–90°C). Liquid argon has a boiling point of –303°F (–186°C). The temperature difference between the product and the surrounding environment, even in winter, is substantial. Keeping this surrounding heat from the product requires special equipment to store and handle cryogenic liquids. A typical system consists of the following components: a cryogenic storage tank, one or more vaporizers, a pressure control system, and all of the piping required for fill, vaporization. The cryogenic tank is constructed, in principle, like a vacuum bottle. It is designed to keep heat away from the liquid that is contained in the inner vessel. Vaporizers convert the liquid argon to its gaseous state. A pressure control manifold controls the pressure at which the gas is fed to the process. -

Cryogenic Liquid Nitrogen Vehicles (ZEV's)
International Journal of Scientific and Research Publications, Volume 6, Issue 9, September 2016 562 ISSN 2250-3153 Cryogenic Liquid Nitrogen Vehicles (ZEV’S) K J Yogesh Department Of Mechanical Engineering, Jain Engineering College, Belagavi Abstract- As a result of widely increasing air pollution available zero emission vehicle (ZEV) meeting it's standards are throughout the world & vehicle emissions having a major the electrically recharged ones, however these vehicles are also contribution towards the same, it makes its very essential to not a great success in the society due to its own limitations like engineer or design an alternative to the present traditional initial cost, slow recharge, speeds etc. Lead acid & Ni-Cd gasoline vehicles. Liquid nitrogen fueled vehicles can act as an batteries are the past of major technologies in the electric excellent alternative for the same. Liquefied N2 at cryogenic vehicles. They exhibit specific energy in the range of 30-40 W- temperatures can replace conventional fuels in cryogenic heat hr/kg. Lead- acid batteries take hours to recharge & the major engines used as a propellant. The ambient temperature of the drawback of the batteries in all the cases is their replacement surrounding vaporizes the liquid form of N2 under pressure & periodically. This directly/indirectly increases the operating cost leads to the formation of compressed N2 gas. This gas actuates a when studied carefully & thereby not 100% acceptable. pneumatic motor. A combination of multiple reheat open Recent studies make it clear that the vehicles using liquid Rankine cycle & closed Brayton cycle are involved in the nitrogen as their means provide an excellent alternative before process to make use of liquid N2 as a non-polluting fuel. -

Capacity Enhancement of Indigenous Expansion Engine Based Helium Liquefier
IOP Conference Series: Materials Science and Engineering PAPER • OPEN ACCESS Capacity enhancement of indigenous expansion engine based helium liquefier To cite this article: R S Doohan et al 2017 IOP Conf. Ser.: Mater. Sci. Eng. 171 012011 View the article online for updates and enhancements. This content was downloaded from IP address 170.106.202.8 on 27/09/2021 at 00:16 ICECICMC IOP Publishing IOP Conf. Series: Materials Science and Engineering 171 (2017) 012011 doi:10.1088/1757-899X/171/1/012011 International Conference on Recent Trends in Physics 2016 (ICRTP2016) IOP Publishing Journal of Physics: Conference Series 755 (2016) 011001 doi:10.1088/1742-6596/755/1/011001 Capacity enhancement of indigenous expansion engine based helium liquefier R S Doohan1, P K Kush1, G Maheshwari2 1Raja Ramanna Centre for Advanced Technology, Indore, Madhya Pradesh India. 2Institute of Engineering and Technology, DAVV, Indore, Madhya Pradesh, India. E-mail: [email protected] Abstract. Development of technology and understanding for large capacity helium refrigeration and liquefaction at helium temperature is indispensable for coming-up projects. A new version of helium liquefier designed and built to provide approximately 35 liters of liquid helium per hour. The refrigeration capacity of this reciprocating type expansion engine machine has been increased from its predecessor version with continuous improvement and deficiency debugging. The helium liquefier has been built using components by local industries including cryogenic Aluminum plate fin heat exchangers. Two compressors with nearly identical capacity have been deployed for the operation of system. Together they consume about 110 kW of electric power. The system employs liquid Nitrogen precooling to enhance liquid Helium yield. -
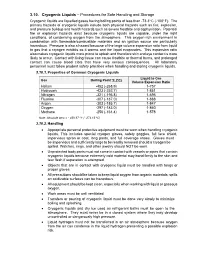
3.10. Cryogenic Liquids – Procedures for Safe Handling and Storage Cryogenic Liquids Are Liquefied Gases Having Boiling Points of Less Than -73.3O C (-100O F)
3.10. Cryogenic Liquids – Procedures for Safe Handling and Storage Cryogenic liquids are liquefied gases having boiling points of less than -73.3o C (-100o F). The primary hazards of cryogenic liquids include both physical hazards such as fire, explosion, and pressure buildup and health hazards such as severe frostbite and asphyxiation. Potential fire or explosion hazards exist because cryogenic liquids are capable, under the right conditions, of condensing oxygen from the atmosphere. This oxygen-rich environment in combination with flammable/combustible materials and an ignition source are particularly hazardous. Pressure is also a hazard because of the large volume expansion ratio from liquid to gas that a cryogen exhibits as it warms and the liquid evaporates. This expansion ratio also makes cryogenic liquids more prone to splash and therefore skin and eye contact is more likely to occur. Contact with living tissue can cause frostbite or thermal burns, and prolonged contact can cause blood clots that have very serious consequences. All laboratory personnel must follow prudent safety practices when handling and storing cryogenic liquids. 3.10.1. Properties of Common Cryogenic Liquids Liquid to Gas Gas Boiling Point oF (oC) Volume Expansion Ratio Helium -452 (-268.9) 1-757 Hydrogen -423 (-252.7) 1-851 Nitrogen -321 (-195.8) 1-696 Fluorine -307 (-187.0) 1-888 Argon -303 (-185.7) 1-847 Oxygen -297 (-183.0) 1-860 Methane -256 (-161.4) 1-578 Note: Absolute zero = - 459.67 oF (- 273.15 oC) 3.10.2. Handling Appropriate personal protective equipment must be worn when handling cryogenic liquids. -

Cryogenic Liquid Safety
Cryogenic Liquid Safety Hazards and Safe Handling of Cryogenic Liquids Cryogenic liquids have boiling points less than -73ºC (-100ºF). Liquid nitrogen, liquid oxygen and carbon dioxide are the most common cryogenic materials used in the laboratory. Hazards may include fire, explosion, embrittlement, pressure buildup, frostbite and asphyxiation. Many of the safety precautions observed for compressed gases also apply to cryogenic liquids. There are also additional hazards due to the unique properties of cryogenic liquids: • Extremely Low Temperatures: The cold boil-off vapor of cryogenic liquids rapidly freezes human tissue. Most metals become stronger upon exposure to cold temperatures, but materials, such as; carbon steel, plastics and rubber become brittle or fracture under stress at these temperatures. Proper material selection is important. Cold burns and frostbite caused by cryogenic liquids can result in extensive tissue damage. • Vaporization: All cryogenic liquids produce large volumes of gas when they vaporize. Liquid nitrogen will expand 696 times as it vaporizes. The expansion ratio of argon is 847:1, hydrogen is 851:1 and oxygen is 862:1. If these liquids vaporize in a sealed container, they can produce enormous pressures that could rupture the vessel. For this reason, pressurized cryogenic containers are usually protected with multiple pressure relief devices. • Vaporization of cryogenic liquids (except oxygen) in an enclosed area can cause asphyxiation! • Vaporization of liquid O2 can produce an oxygen-rich atmosphere, supporting and accelerating combustion of other materials. Vaporization of liquid hydrogen can form an extremely flammable mixture with air. Most cryogenic liquids are odorless, colorless, and tasteless when vaporized. When cryogenic liquids are exposed to the atmosphere, cold boil-off gases condense the moisture in the air, creating a highly visible fog. -

Helium, Refrigerated Liquid
Helium, Refrigerated Liquid Safety Data Sheet P-4600 This SDS conforms to U.S. Code of Federal Regulations 29 CFR 1910.1200, Hazard Communication. Issue date: 01/01/1979 Revision date: 01/31/2021 Supersedes: 09/08/2020 Version: 2.0 SECTION: 1. Product and company identification 1.1. Product identifier Product form : Substance Trade name : Liquid Helium CAS-No. : 7440-59-7 Formula : He Other means of identification : Helium, Refrigerated Liquid; Helium-4; Refrigerant Gas R-704 1.2. Relevant identified uses of the substance or mixture and uses advised against Use of the substance/mixture : Industrial use; Use as directed. Diving Gas (Underwater Breathing) 1.3. Details of the supplier of the safety data sheet Linde Inc. 10 Riverview Drive Danbury, CT 06810-6268 - USA 1.4. Emergency telephone number Emergency number : Onsite Emergency: 1-800-645-4633 CHEMTREC, 24hr/day 7days/week — Within USA: 1-800-424-9300, Outside USA: 001-703-527-3887 (collect calls accepted, Contract 17729) SECTION 2: Hazard identification 2.1. Classification of the substance or mixture GHS US classification Simple asphyxiant SIAS Press. Gas (Ref. Liq.) H281 2.2. Label elements GHS US labeling Hazard pictograms (GHS US) : GHS04 Signal word (GHS US) : Warning Hazard statements (GHS US) : H281 - CONTAINS REFRIGERATED GAS; MAY CAUSE CRYOGENIC BURNS OR INJURY OSHA-H01 - MAY DISPLACE OXYGEN AND CAUSE RAPID SUFFOCATION. Precautionary statements (GHS US) : P202 - Do not handle until all safety precautions have been read and understood. P271+P403 - Use and store only outdoors or in a well-ventilated place. P282 - Wear cold insulating gloves/face shield/eye protection. -

Liquid Nitrogen Safety & Handling
Liquid Nitrogen Safety & Handling 1.0 Liquid Nitrogen & Cryogenic Liquids a. Liquid nitrogen, LN2, is colorless, odorless and tasteless. It is non-flammable, physiologically inert and non-toxic. It is an extremely cold liquid with a boiling point of - 196ºC = -320ºF. b. Cryogenic liquids are liquefied gases that are kept in their liquid state at extremely low temperatures. Cryogenic liquids have boiling points below -150°C (-238°F) (Carbon dioxide and nitrous oxide, which have slightly higher boiling points are sometimes included in this category). All cryogenic liquids are gases at normal temperatures and pressures. All Cryogenic liquids produce large amounts of gas when they vaporize. c. There a many applications of LN2 in a biomedical research facility, one of the most common being cryopreservation, the storage of cells at temperatures below -130 ºC, which is essential in order to preserve biological material unaltered. 2.0 Safety Hazards Associated with Handling LN2 a. The extremely low temperature of the liquid can cause severe frostbite or eye damage upon contact. Symptoms of frostbite include change in skin color to white or grayish yellow and the pain after contact with LN2 may quickly subside. Items in contact with liquid nitrogen become extremely cold. Touching these items may result in torn flesh. Many substances become brittle upon contact with liquid nitrogen and may shatter when cold (such as common glass and large solid plastics), sending pieces of the material flying. b. Upon vaporization it expands by a factor of 700; one liter of liquid nitrogen becomes 24.6 cubic feet of nitrogen gas. -

Visualization of Two-Fluid Flows of Superfluid Helium-4 SPECIAL FEATURE
Visualization of two-fluid flows of superfluid helium-4 SPECIAL FEATURE Wei Guoa,b, Marco La Mantiac, Daniel P. Lathropd, and Steven W. Van Scivera,b,1 aMechanical Engineering Department, Florida State University, Tallahassee, FL 32303; bNational High Magnetic Field Laboratory, Florida State University, Tallahassee, FL 32310; cDepartment of Low Temperature Physics, Faculty of Mathematics and Physics, Charles University, 180 00 Prague, Czech Republic; and dDepartments of Physics and Geology, Institute for Research in Electronics and Applied Physics, and Institute for Physics Science and Technology, University of Maryland, College Park, MD 20742 Edited by Katepalli R. Sreenivasan, New York University, New York, NY, and approved December 13, 2013 (received for review July 17, 2013) Cryogenic flow visualization techniques have been proved in not kept pace, in part due to the extremely low temperature and recent years to be a very powerful experimental method to study low density of the fluid. A number of early efforts were devoted superfluid turbulence. Micron-sized solid particles and metastable to producing macroscopic particles for qualitative investigations helium molecules are specifically being used to investigate in (10–12) and the challenge of producing neutrally buoyant par- detail the dynamics of quantum flows. These studies belong to ticles that faithfully follow the complex flow fields has been the a well-established, interdisciplinary line of inquiry that focuses on main impediment to quantitative advancement. In addition, sev- the deeper understanding of turbulence, one of the open problem eral attempts have been made to visualize fluid dynamics in su- of modern physics, relevant to many research fields, ranging from perfluid helium with microscopic tracers, which include neutron fluid mechanics to cosmology. -

The Noble Gases
The Noble Gases The Noble Gases (inert gases, Group 0, Group 18 or the helium group) are notoriously unreactive elements (‘noble’ means unreactive in chemistry) and in their elemental state they exist as monoatomic gases – gases whose ‘molecules’ are single atoms of the element, since the atoms are reluctant to react with anything, including one-another. This inertness is due to the fact that they have stable outer electron shells, with stable octets of electrons (full s and p subshells) except helium, which has a stable full inner shell. The electronic configurations are: Helium (He): 1s2 Neon (Ne): 1s2 2s2 2p6 Argon (Ar): [Ne] 3s2 3p6 Krypton (Kr): [Ar] 3d10 4s2 4p6 Xenon (Xe): [Kr] 4d10 5s2 5p6 Radon (Rn): [Xe] 5d10 6s2 6p6 Nevertheless, this group does have some interesting chemistry and also exhibit interesting physical properties. Reactivity increases down the group. Often helium is included as the first member of the group. Helium (He) Helium is chemically a highly unreactive element. It only forms transient species when electric discharges are passed through a mixture of helium gas and another gaseous element. for example, passing an electric discharge through a mixture of helium and hydrogen forms the transient molecule HHe, which has a very short half-life. HHeF is metastable. Neon (Ne) Neon is chemically the most unreactive element. It forms no true compounds, and no neutral molecules. Ionic molecules are known, e.g. (NeAr)+, (NeH)+, (HeNe)+ and Ne+. Argon (Ar) The unstable argon fluorohydride, HArF, is known. Ar also forms clathrates (see krypton) with water and highly unstable ArH+ and ArF are known. -

The Noble Gases
INTERCHAPTER K The Noble Gases When an electric discharge is passed through a noble gas, light is emitted as electronically excited noble-gas atoms decay to lower energy levels. The tubes contain helium, neon, argon, krypton, and xenon. University Science Books, ©2011. All rights reserved. www.uscibooks.com Title General Chemistry - 4th ed Author McQuarrie/Gallogy Artist George Kelvin Figure # fig. K2 (965) Date 09/02/09 Check if revision Approved K. THE NOBLE GASES K1 2 0 Nitrogen and He Air P Mg(ClO ) NaOH 4 4 2 noble gases 4.002602 1s2 O removal H O removal CO removal 10 0 2 2 2 Ne Figure K.1 A schematic illustration of the removal of O2(g), H2O(g), and CO2(g) from air. First the oxygen is removed by allowing the air to pass over phosphorus, P (s) + 5 O (g) → P O (s). 20.1797 4 2 4 10 2s22p6 The residual air is passed through anhydrous magnesium perchlorate to remove the water vapor, Mg(ClO ) (s) + 6 H O(g) → Mg(ClO ) ∙6 H O(s), and then through sodium hydroxide to remove 18 0 4 2 2 4 2 2 the carbon dioxide, NaOH(s) + CO2(g) → NaHCO3(s). The gas that remains is primarily nitrogen Ar with about 1% noble gases. 39.948 3s23p6 36 0 The Group 18 elements—helium, K-1. The Noble Gases Were Kr neon, argon, krypton, xenon, and Not Discovered until 1893 83.798 radon—are called the noble gases 2 6 4s 4p and are noteworthy for their rela- In 1893, the English physicist Lord Rayleigh noticed 54 0 tive lack of chemical reactivity. -

Liquid Oxygen
Safetygram 6 Liquid oxygen Oxygen is the second largest component of the atmosphere, comprising 20.8% by volume. Liquid oxygen is pale blue and extremely cold. Although nonflammable, oxygen is a strong oxidizer. Oxygen is necessary to support life. Oxygen will react with nearly all organic materials and metals, usually forming an oxide. Materials that burn in air will burn more vigorously in oxygen. Equipment used in oxygen service must meet stringent cleaning requirements, and systems must be constructed of materials that have high ignition temperatures and that are nonreactive with oxygen under the service conditions. Vessels should be manufactured to American Society of Mechanical Engineers (ASME) codes and designed to withstand the process temperatures and pressures. Liquid oxygen is a cryogenic liquid. Cryogenic liquids are liquefied gases that have a normal boiling point below –130°F (–90°C). Liquid oxygen has a boiling point of –297°F (–183°C). Because the temperature difference between the product and the surrounding environment is substantial—even in the winter—keeping liquid oxygen insulated from the surrounding heat is essential. The product also requires special equipment for handling and storage. Oxygen is often stored as a liquid, although it is used primarily as a gas. Liquid storage is less bulky and less costly than the equivalent capacity of high-pressure gaseous storage. A typical storage system consists of a cryogenic storage tank, one or more vaporizers and a pressure control system. The cryogenic tank is constructed, in principle, like a vacuum bottle. There is an inner vessel surrounded by an outer vessel. Between the vessels is an annular space that contains an insulating medium from which all the air has been removed.