FGD2 (V-15): Sc-11114
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Regulation of Cdc42 and Its Effectors in Epithelial Morphogenesis Franck Pichaud1,2,*, Rhian F
© 2019. Published by The Company of Biologists Ltd | Journal of Cell Science (2019) 132, jcs217869. doi:10.1242/jcs.217869 REVIEW SUBJECT COLLECTION: ADHESION Regulation of Cdc42 and its effectors in epithelial morphogenesis Franck Pichaud1,2,*, Rhian F. Walther1 and Francisca Nunes de Almeida1 ABSTRACT An overview of Cdc42 Cdc42 – a member of the small Rho GTPase family – regulates cell Cdc42 was discovered in yeast and belongs to a large family of small – polarity across organisms from yeast to humans. It is an essential (20 30 kDa) GTP-binding proteins (Adams et al., 1990; Johnson regulator of polarized morphogenesis in epithelial cells, through and Pringle, 1990). It is part of the Ras-homologous Rho subfamily coordination of apical membrane morphogenesis, lumen formation and of GTPases, of which there are 20 members in humans, including junction maturation. In parallel, work in yeast and Caenorhabditis elegans the RhoA and Rac GTPases, (Hall, 2012). Rho, Rac and Cdc42 has provided important clues as to how this molecular switch can homologues are found in all eukaryotes, except for plants, which do generate and regulate polarity through localized activation or inhibition, not have a clear homologue for Cdc42. Together, the function of and cytoskeleton regulation. Recent studies have revealed how Rho GTPases influences most, if not all, cellular processes. important and complex these regulations can be during epithelial In the early 1990s, seminal work from Alan Hall and his morphogenesis. This complexity is mirrored by the fact that Cdc42 can collaborators identified Rho, Rac and Cdc42 as main regulators of exert its function through many effector proteins. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

High Throughput Strategies Aimed at Closing the GAP in Our Knowledge of Rho Gtpase Signaling
cells Review High Throughput strategies Aimed at Closing the GAP in Our Knowledge of Rho GTPase Signaling Manel Dahmene 1, Laura Quirion 2 and Mélanie Laurin 1,3,* 1 Oncology Division, CHU de Québec–Université Laval Research Center, Québec, QC G1V 4G2, Canada; [email protected] 2 Montréal Clinical Research Institute (IRCM), Montréal, QC H2W 1R7, Canada; [email protected] 3 Université Laval Cancer Research Center, Québec, QC G1R 3S3, Canada * Correspondence: [email protected] Received: 21 May 2020; Accepted: 7 June 2020; Published: 9 June 2020 Abstract: Since their discovery, Rho GTPases have emerged as key regulators of cytoskeletal dynamics. In humans, there are 20 Rho GTPases and more than 150 regulators that belong to the RhoGEF, RhoGAP, and RhoGDI families. Throughout development, Rho GTPases choregraph a plethora of cellular processes essential for cellular migration, cell–cell junctions, and cell polarity assembly. Rho GTPases are also significant mediators of cancer cell invasion. Nevertheless, to date only a few molecules from these intricate signaling networks have been studied in depth, which has prevented appreciation for the full scope of Rho GTPases’ biological functions. Given the large complexity involved, system level studies are required to fully grasp the extent of their biological roles and regulation. Recently, several groups have tackled this challenge by using proteomic approaches to map the full repertoire of Rho GTPases and Rho regulators protein interactions. These studies have provided in-depth understanding of Rho regulators specificity and have contributed to expand Rho GTPases’ effector portfolio. Additionally, new roles for understudied family members were unraveled using high throughput screening strategies using cell culture models and mouse embryos. -

Supplementary Table 1
Supplementary Table 1. 492 genes are unique to 0 h post-heat timepoint. The name, p-value, fold change, location and family of each gene are indicated. Genes were filtered for an absolute value log2 ration 1.5 and a significance value of p ≤ 0.05. Symbol p-value Log Gene Name Location Family Ratio ABCA13 1.87E-02 3.292 ATP-binding cassette, sub-family unknown transporter A (ABC1), member 13 ABCB1 1.93E-02 −1.819 ATP-binding cassette, sub-family Plasma transporter B (MDR/TAP), member 1 Membrane ABCC3 2.83E-02 2.016 ATP-binding cassette, sub-family Plasma transporter C (CFTR/MRP), member 3 Membrane ABHD6 7.79E-03 −2.717 abhydrolase domain containing 6 Cytoplasm enzyme ACAT1 4.10E-02 3.009 acetyl-CoA acetyltransferase 1 Cytoplasm enzyme ACBD4 2.66E-03 1.722 acyl-CoA binding domain unknown other containing 4 ACSL5 1.86E-02 −2.876 acyl-CoA synthetase long-chain Cytoplasm enzyme family member 5 ADAM23 3.33E-02 −3.008 ADAM metallopeptidase domain Plasma peptidase 23 Membrane ADAM29 5.58E-03 3.463 ADAM metallopeptidase domain Plasma peptidase 29 Membrane ADAMTS17 2.67E-04 3.051 ADAM metallopeptidase with Extracellular other thrombospondin type 1 motif, 17 Space ADCYAP1R1 1.20E-02 1.848 adenylate cyclase activating Plasma G-protein polypeptide 1 (pituitary) receptor Membrane coupled type I receptor ADH6 (includes 4.02E-02 −1.845 alcohol dehydrogenase 6 (class Cytoplasm enzyme EG:130) V) AHSA2 1.54E-04 −1.6 AHA1, activator of heat shock unknown other 90kDa protein ATPase homolog 2 (yeast) AK5 3.32E-02 1.658 adenylate kinase 5 Cytoplasm kinase AK7 -

1 Fibrillar Αβ Triggers Microglial Proteome Alterations and Dysfunction in Alzheimer Mouse 1 Models 2 3 4 Laura Sebastian
bioRxiv preprint doi: https://doi.org/10.1101/861146; this version posted December 2, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 1 Fibrillar triggers microglial proteome alterations and dysfunction in Alzheimer mouse 2 models 3 4 5 Laura Sebastian Monasor1,10*, Stephan A. Müller1*, Alessio Colombo1, Jasmin König1,2, Stefan 6 Roth3, Arthur Liesz3,4, Anna Berghofer5, Takashi Saito6,7, Takaomi C. Saido6, Jochen Herms1,4,8, 7 Michael Willem9, Christian Haass1,4,9, Stefan F. Lichtenthaler 1,4,5# & Sabina Tahirovic1# 8 9 1 German Center for Neurodegenerative Diseases (DZNE) Munich, 81377 Munich, Germany 10 2 Faculty of Chemistry, Technical University of Munich, Garching, Germany 11 3 Institute for Stroke and Dementia Research (ISD), Ludwig-Maximilians Universität München, 12 81377 Munich, Germany 13 4 Munich Cluster for Systems Neurology (SyNergy), Munich, Germany 14 5 Neuroproteomics, School of Medicine, Klinikum Rechts der Isar, Technical University of Munich, 15 Munich, Germany 16 6 Laboratory for Proteolytic Neuroscience, RIKEN Center for Brain Science Institute, Wako, 17 Saitama 351-0198, Japan 18 7 Department of Neurocognitive Science, Nagoya City University Graduate School of Medical 19 Science, Nagoya, Aichi 467-8601, Japan 20 8 Center for Neuropathology and Prion Research, Ludwig-Maximilians-Universität München, 81377 21 Munich, Germany 22 9 Biomedical Center (BMC), Ludwig-Maximilians Universität München, 81377 Munich, Germany 23 10 Graduate School of Systemic Neuroscience, Ludwig-Maximilians-University Munich, Munich, 24 Germany. 25 *Contributed equally 26 #Correspondence: [email protected] and [email protected] 27 28 Running title: 29 Microglial proteomic signatures of AD 30 Keywords: Alzheimer’s disease / microglia / proteomic signatures / neuroinflammation / 31 phagocytosis 32 1 bioRxiv preprint doi: https://doi.org/10.1101/861146; this version posted December 2, 2019. -

Whole Genome Sequencing Reveals Multiple Linked Genetic Variants On
G C A T T A C G G C A T genes Article Whole Genome Sequencing Reveals Multiple Linked Genetic Variants on Canine Chromosome 12 Associated with Risk for Symmetrical Lupoid Onychodystrophy (SLO) in the Bearded Collie Liza C. Gershony 1 , Janelle M. Belanger 1 , Marjo K. Hytönen 2,3,4 , Hannes Lohi 2,3,4 and Anita M. Oberbauer 1,* 1 Department on Animal Science, University of California, Davis, CA 95616, USA; [email protected] (L.C.G.); [email protected] (J.M.B.) 2 Department of Medical and Clinical Genetics, University of Helsinki, 00014 Helsinki, Finland; marjo.hytonen@helsinki.fi (M.K.H.); hannes.lohi@helsinki.fi (H.L.) 3 Department of Veterinary Biosciences, University of Helsinki, 00014 Helsinki, Finland 4 Folkhälsan Research Center, 00290 Helsinki, Finland * Correspondence: [email protected] Abstract: In dogs, symmetrical lupoid onychodystrophy (SLO) results in nail loss and an abnormal regrowth of the claws. In Bearded Collies, an autoimmune nature has been suggested because certain dog leukocyte antigen (DLA) class II haplotypes are associated with the condition. A genome-wide Citation: Gershony, L.C.; Belanger, association study of the Bearded Collie revealed two regions of association that conferred risk for J.M.; Hytönen, M.K.; Lohi, H.; disease: one on canine chromosome (CFA) 12 that encompasses the DLA genes, and one on CFA17. Oberbauer, A.M. Whole Genome Case-control association was employed on whole genome sequencing data to uncover putative Sequencing Reveals Multiple Linked causative variants in SLO within the CFA12 and CFA17 associated regions. Genotype imputation was Genetic Variants on Canine then employed to refine variants of interest. -

The Evolutionary Landscape of Dbl-Like Rhogef Families: Adapting Eukaryotic Cells to Environmental Signals
GBE The Evolutionary Landscape of Dbl-Like RhoGEF Families: Adapting Eukaryotic Cells to Environmental Signals Philippe Fort1,2,* and Anne Blangy1,2 1CRBM, Universite´ of Montpellier, France 2CNRS, UMR5237, Montpellier, France *Corresponding author: E-mail: [email protected]. Accepted: May 24, 2017 Abstract Thedynamicsofcellmorphologyineukaryotesislargely controlledbysmallGTPasesoftheRhofamily.RhoGTPasesareactivatedby guanine nucleotide exchange factors (RhoGEFs), of which diffuse B-cell lymphoma (Dbl)-like members form the largest family. Here, we surveyed Dbl-like sequences from 175 eukaryotic genomes and illuminate how the Dbl family evolved in all eukaryotic supergroups. By combining probabilistic phylogenetic approaches and functional domain analysis, we show that the human Dbl- like family is made of 71 members, structured into 20 subfamilies. The 71 members were already present in ancestral jawed vertebrates, but several membersweresubsequentlylost in specific clades,up to 12% in birds.The jawed vertebrate repertoirewas established from two rounds of duplications that occurred between tunicates, cyclostomes, and jawed vertebrates. Duplicated members showed distinct tissue distributions, conserved at least in Amniotes. All 20 subfamilies have members in Deuterostomes and Protostomes. Nineteen subfamilies are present in Porifera, the first phylum that diverged in Metazoa, 14 in Choanoflagellida and Filasterea, single- celled organisms closely related to Metazoa and three in Fungi, the sister clade to Metazoa. Other eukaryotic supergroups show an extraordinary variability of Dbl-like repertoires as a result of repeated and independent gain and loss events. Last, we observed that in Metazoa, the number of Dbl-like RhoGEFs varies in proportion of cell signaling complexity. Overall, our analysis supports the conclusion that Dbl-like RhoGEFs were present at the origin of eukaryotes and evolved as highly adaptive cell signaling mediators. -

Original Article Integrative Analysis of Somatic Mutations and Differential Expression Profiles in Glioblastoma Based on Aging Acceleration
Int J Clin Exp Pathol 2021;14(5):582-595 www.ijcep.com /ISSN:1936-2625/IJCEP0121428 Original Article Integrative analysis of somatic mutations and differential expression profiles in glioblastoma based on aging acceleration Huize Wang1,2, Shiyan Li3, Hongxin Liu3, Shiyu Bian4, Wanjiang Huang5, Chengzhong Xing2, Yin Wang2,3 1Department of Nursing, First Affiliated Hospital of China Medical University, 155# North Nanjing Street, Shenyang 110001, Liaoning, China; 2Tumor Etiology and Screening Department of Cancer Institute and General Surgery, The First Affiliated Hospital of China Medical University, 155# North Nanjing Street, Heping District, Shenyang 110001, Liaoning Province, China; 3Department of Biomedical Engineering, School of Fundamental Sciences, China Medical University, Shenyang 110122, Liaoning Province, China; 4China Medical University - The Queen’s University of Belfast Joint College, China Medical University, Shenyang 110122, Liaoning Province, China; 5No. 10 Middle School, Xiangyang 441021, Hubei Province, China Received August 31, 2020; Accepted February 7, 2021; Epub May 15, 2021; Published May 30, 2021 Abstract: Background: Glioblastoma (GBM) is an aggressive brain tumor and the mechanisms of progression are very complex. Accelerated aging is a driving factor of GBM. However, there has not been thorough research about the mechanisms of GBM progression based on aging acceleration. Methods: The aging predictor was modeled based on normal brain samples. Then an aging acceleration background network was constructed to explore GBM mechanisms. Results: The accelerated aging-related mechanisms provided an innovative way to study GBM, where- in integrative analysis of somatic mutations and differential expression revealed key pathologic characteristics. Moreover, the influence of the immune system, the nervous system and other critical factors on GBM were identi- fied. -
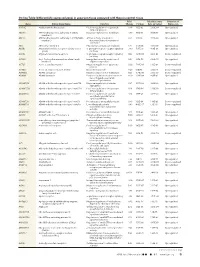
Differentially Expressed Genes in Aneurysm Tissue Compared With
On-line Table: Differentially expressed genes in aneurysm tissue compared with those in control tissue Fold False Discovery Direction of Gene Entrez Gene Name Function Change P Value Rate (q Value) Expression AADAC Arylacetamide deacetylase Positive regulation of triglyceride 4.46 1.33E-05 2.60E-04 Up-regulated catabolic process ABCA6 ATP-binding cassette, subfamily A (ABC1), Integral component of membrane 3.79 9.15E-14 8.88E-12 Up-regulated member 6 ABCC3 ATP-binding cassette, subfamily C (CFTR/MRP), ATPase activity, coupled to 6.63 1.21E-10 7.33E-09 Up-regulated member 3 transmembrane movement of substances ABI3 ABI family, member 3 Peptidyl-tyrosine phosphorylation 6.47 2.47E-05 4.56E-04 Up-regulated ACKR1 Atypical chemokine receptor 1 (Duffy blood G-protein–coupled receptor signaling 3.80 7.95E-10 4.18E-08 Up-regulated group) pathway ACKR2 Atypical chemokine receptor 2 G-protein–coupled receptor signaling 0.42 3.29E-04 4.41E-03 Down-regulated pathway ACSM1 Acyl-CoA synthetase medium-chain family Energy derivation by oxidation of 9.87 1.70E-08 6.52E-07 Up-regulated member 1 organic compounds ACTC1 Actin, ␣, cardiac muscle 1 Negative regulation of apoptotic 0.30 7.96E-06 1.65E-04 Down-regulated process ACTG2 Actin, ␥2, smooth muscle, enteric Blood microparticle 0.29 1.61E-16 2.36E-14 Down-regulated ADAM33 ADAM domain 33 Integral component of membrane 0.23 9.74E-09 3.95E-07 Down-regulated ADAM8 ADAM domain 8 Positive regulation of tumor necrosis 4.69 2.93E-04 4.01E-03 Up-regulated factor (ligand) superfamily member 11 production ADAMTS18 -

Fibrosis Accumulation in Idiopathic
Downloaded from http://www.jimmunol.org/ by guest on September 26, 2021 D. is online at: average * The Journal of Immunology published online 1 June 2011 from submission to initial decision 4 weeks from acceptance to publication Groshong, Gregory P. Cosgrove, Elizabeth F. Redente, Alison Bamberg, Kevin K. Brown, Nichole Reisdorph, Rebecca C. Keith, Stephen K. Frankel and David W. H. Riches http://www.jimmunol.org/content/early/2011/05/29/jimmun ol.1100447 Increased Cell Surface Fas Expression Is Necessary and Sufficient To Sensitize Lung Fibroblasts to Fas Ligation-Induced Apoptosis: Implications for Fibroblast Accumulation in Idiopathic Pulmonary Fibrosis Murry W. Wynes, Benjamin L. Edelman, Amanda G. Kostyk, Michael G. Edwards, Christopher Coldren, Steve J Immunol Submit online. Every submission reviewed by practicing scientists ? is published twice each month by http://jimmunol.org/subscription Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts http://www.jimmunol.org/content/suppl/2011/06/01/jimmunol.110044 7.DC1 Information about subscribing to The JI No Triage! Fast Publication! Rapid Reviews! 30 days* Why • • • Material Permissions Email Alerts Subscription Supplementary The Journal of Immunology The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2011 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. This information is current as of September 26, 2021. Published June 1, 2011, doi:10.4049/jimmunol.1100447 The Journal of Immunology Increased Cell Surface Fas Expression Is Necessary and Sufficient To Sensitize Lung Fibroblasts to Fas Ligation-Induced Apoptosis: Implications for Fibroblast Accumulation in Idiopathic Pulmonary Fibrosis Murry W. -

Molecular Mechanisms of Oncogenesis & Precision Medicine
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 2016 Molecular Mechanisms of Oncogenesis & Precision Medicine Approaches for Pediatric Low-Grade Gliomas Payal Jain University of Pennsylvania, [email protected] Follow this and additional works at: https://repository.upenn.edu/edissertations Part of the Cell Biology Commons, Medicine and Health Sciences Commons, and the Molecular Biology Commons Recommended Citation Jain, Payal, "Molecular Mechanisms of Oncogenesis & Precision Medicine Approaches for Pediatric Low- Grade Gliomas" (2016). Publicly Accessible Penn Dissertations. 1782. https://repository.upenn.edu/edissertations/1782 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/edissertations/1782 For more information, please contact [email protected]. Molecular Mechanisms of Oncogenesis & Precision Medicine Approaches for Pediatric Low-Grade Gliomas Abstract Pediatric low-grade gliomas (PLGGs) are a heterogeneous group of tumors that collectively represent the most common childhood brain cancer. Despite favorable outcomes with surgical and adjuvant therapies, majority of patients suffer from long-term treatment-related morbidities and recurrent/inoperable disease. This necessitates a deeper understanding of PLGG biology to aid development of molecular diagnostics and low-toxicity targeted therapeutics. Hitherto, PLGGs have been defined by activating mutations that dysregulate the MAPK signaling pathway, leading to clinical testing of RAF/MAPK inhibitors for PLGGs. -

Table S1. 103 Ferroptosis-Related Genes Retrieved from the Genecards
Table S1. 103 ferroptosis-related genes retrieved from the GeneCards. Gene Symbol Description Category GPX4 Glutathione Peroxidase 4 Protein Coding AIFM2 Apoptosis Inducing Factor Mitochondria Associated 2 Protein Coding TP53 Tumor Protein P53 Protein Coding ACSL4 Acyl-CoA Synthetase Long Chain Family Member 4 Protein Coding SLC7A11 Solute Carrier Family 7 Member 11 Protein Coding VDAC2 Voltage Dependent Anion Channel 2 Protein Coding VDAC3 Voltage Dependent Anion Channel 3 Protein Coding ATG5 Autophagy Related 5 Protein Coding ATG7 Autophagy Related 7 Protein Coding NCOA4 Nuclear Receptor Coactivator 4 Protein Coding HMOX1 Heme Oxygenase 1 Protein Coding SLC3A2 Solute Carrier Family 3 Member 2 Protein Coding ALOX15 Arachidonate 15-Lipoxygenase Protein Coding BECN1 Beclin 1 Protein Coding PRKAA1 Protein Kinase AMP-Activated Catalytic Subunit Alpha 1 Protein Coding SAT1 Spermidine/Spermine N1-Acetyltransferase 1 Protein Coding NF2 Neurofibromin 2 Protein Coding YAP1 Yes1 Associated Transcriptional Regulator Protein Coding FTH1 Ferritin Heavy Chain 1 Protein Coding TF Transferrin Protein Coding TFRC Transferrin Receptor Protein Coding FTL Ferritin Light Chain Protein Coding CYBB Cytochrome B-245 Beta Chain Protein Coding GSS Glutathione Synthetase Protein Coding CP Ceruloplasmin Protein Coding PRNP Prion Protein Protein Coding SLC11A2 Solute Carrier Family 11 Member 2 Protein Coding SLC40A1 Solute Carrier Family 40 Member 1 Protein Coding STEAP3 STEAP3 Metalloreductase Protein Coding ACSL1 Acyl-CoA Synthetase Long Chain Family Member 1 Protein