Counteracting EMF Toxicity with Peptides (Forget Global Warming Emfs Are Killing Us)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The National Drugs List
^ ^ ^ ^ ^[ ^ The National Drugs List Of Syrian Arab Republic Sexth Edition 2006 ! " # "$ % &'() " # * +$, -. / & 0 /+12 3 4" 5 "$ . "$ 67"5,) 0 " /! !2 4? @ % 88 9 3: " # "$ ;+<=2 – G# H H2 I) – 6( – 65 : A B C "5 : , D )* . J!* HK"3 H"$ T ) 4 B K<) +$ LMA N O 3 4P<B &Q / RS ) H< C4VH /430 / 1988 V W* < C A GQ ") 4V / 1000 / C4VH /820 / 2001 V XX K<# C ,V /500 / 1992 V "!X V /946 / 2004 V Z < C V /914 / 2003 V ) < ] +$, [2 / ,) @# @ S%Q2 J"= [ &<\ @ +$ LMA 1 O \ . S X '( ^ & M_ `AB @ &' 3 4" + @ V= 4 )\ " : N " # "$ 6 ) G" 3Q + a C G /<"B d3: C K7 e , fM 4 Q b"$ " < $\ c"7: 5) G . HHH3Q J # Hg ' V"h 6< G* H5 !" # $%" & $' ,* ( )* + 2 ا اوا ادو +% 5 j 2 i1 6 B J' 6<X " 6"[ i2 "$ "< * i3 10 6 i4 11 6! ^ i5 13 6<X "!# * i6 15 7 G!, 6 - k 24"$d dl ?K V *4V h 63[46 ' i8 19 Adl 20 "( 2 i9 20 G Q) 6 i10 20 a 6 m[, 6 i11 21 ?K V $n i12 21 "% * i13 23 b+ 6 i14 23 oe C * i15 24 !, 2 6\ i16 25 C V pq * i17 26 ( S 6) 1, ++ &"r i19 3 +% 27 G 6 ""% i19 28 ^ Ks 2 i20 31 % Ks 2 i21 32 s * i22 35 " " * i23 37 "$ * i24 38 6" i25 39 V t h Gu* v!* 2 i26 39 ( 2 i27 40 B w< Ks 2 i28 40 d C &"r i29 42 "' 6 i30 42 " * i31 42 ":< * i32 5 ./ 0" -33 4 : ANAESTHETICS $ 1 2 -1 :GENERAL ANAESTHETICS AND OXYGEN 4 $1 2 2- ATRACURIUM BESYLATE DROPERIDOL ETHER FENTANYL HALOTHANE ISOFLURANE KETAMINE HCL NITROUS OXIDE OXYGEN PROPOFOL REMIFENTANIL SEVOFLURANE SUFENTANIL THIOPENTAL :LOCAL ANAESTHETICS !67$1 2 -5 AMYLEINE HCL=AMYLOCAINE ARTICAINE BENZOCAINE BUPIVACAINE CINCHOCAINE LIDOCAINE MEPIVACAINE OXETHAZAINE PRAMOXINE PRILOCAINE PREOPERATIVE MEDICATION & SEDATION FOR 9*: ;< " 2 -8 : : SHORT -TERM PROCEDURES ATROPINE DIAZEPAM INJ. -

First Reversals of Cognitive Decline in Alzheimer's Disease and Its
1/12/2017 First Reversals of Cognitive Decline in Alzheimer’sSystems Therapeutics, Disease and its PresidentPrecursors, Obama, MCI and and the End ofSCI Alzheimer’s Disease Dale E. Bredesen, M.D. Augustus Rose Professor Easton Laboratories for Neurodegenerative Disease Research UCLA Founding President, Buck Institute “There is nothing that will prevent, reverse, or slow the progress of Alzheimer’s disease.” “Everyone knows someone who is a cancer survivor; no one knows an Alzheimer’s survivor.” 1 1/12/2017 30,000,000 patients in 2012 3rd leading cause (James, B. D. et al. Contribution of Alzheimer disease to mortality in the United States. Neurology 82, 1045-1050, doi:10.1212/WNL. 0240 (2014) Pres. Obama and NAPA, 2011 160,000,000 patients in 2050 2 1/12/2017 Women at the epicenter of the epidemic •65% of patients •60% of caregivers •More common than breast cancer 0 Cures 3 1/12/2017 Alzheimer’s Disease (AD) Therapeutic Landscape A production APPROVED A aggregation Donepezil (Aricept) A clearance Rivastigmine (Exelon) Tau aggregation/phosph Galantamine (Razadyne) Cholinergic drugs Tacrine (Cognex) Others Memantine (Namenda) PHASE 3 PHASE 2 PHASE 1 Solanezumab ELND005 PBT2 AL-108 GSK933776 NIC5-15 PF04360365 AF102B MABT5102A Bapineuzmab Valproate Bryostatin-1 Nicotinamide Talsaclidine EHT-0202 NP12 ACC001 UB311 Alzemed Antioxidant BMS708163 Lithium AN1792 R1450 Begacestat Semagacestat Statins ABT089 NGF CAD106 V950 PF3084014 AZD3480 SB742457 Flurizan Dimebon E2012 Huperzine-A PRX03140 CTS21166 Rosiglitazone EGCg EVP6124 PUFA MK0752 MEM3454 TTP448 Phenserine CHF5074 PF-04447943 Clinical Trial in AD terminated “Game of Throwns” (243/244) R.I.P. R.I.P. R.I.P. -

Mode of Action
STROKE TBI DEMENTIA Mode of Action Reconnecting Neurons. Empowering for Life. NEW MULTI-MODAL THERAPIES FOR NEUROLOGICAL DISORDERS Sonic hedgehog (Shh) The Sonic Hedgehog (Shh) signalling pathway regulates the development Smoothened of organs including the organization of the brain. For example, the Shh (Smo) Patched (Ptch) activates the Gli complex, which is responsible for the expression of develop- mental genes underpinning neurorecovery and leading to an amplification of natural recovery. Gli Complex Gli Repressor Gli Activator Cerebrolysin activates the Shh pathway1 Cerebrolysin has a promoting effect on neurogenesis and oligodendrogenesis via stimulating the expression of the Shh signalling pathway. Cerebrolysin increases mRNA modulation of Shh and its receptors ‘Patched’ (Ptch) and ‘Smoothened’ (Smo). Shh PTCH Smo 5 * * 3 * 3 4 (n=9) (n=9) *p<0.05 (n=9) *p<0.05 *p<0.05 3 2 2 2 1 1 Fold changes (mean±SE) changes Fold (mean±SE) changes Fold 1 (mean±SE) changes Fold 0 0 0 0 20 0 20 0 20 CERE (µl/ml) CERE (µl/ml) CERE (µl/ml) Figure 1: Cerebrolysin stimulates the expression of the sonic hedgehog signalling pathway components in neural progenitor cells. Graphs show mRNA levels in an in vitro experiment. Studies confirm the important role of the sonic hedgehog pathway in post-stroke brain repair and functional recovery, and suggests the Shh pathway to be a possible target for prolongation of the therapeutic window after stroke.2 MAINTENANCE AND RECOVERY OF THE NEURONAL NETWORK Neurotrophic factors (NTFs) are signaling molecules that maintain, protect, and restore the neuronal network and ensure proper functioning of the brain. -

Workbook Psychiatry and Narcology
Kharkiv National Medical University Department of Psychiatry, Narcology and Medical Psychology WORKBOOK MANUAL FOR INDIVIDUAL WORK FOR MEDICAL STUDENTS PSYCHIATRY AND NARCOLOGY (Part 2) Student ___________________________________________________________ Faculty _________________________________________________________ Course _________________ Group _____________________________________ Kharkiv 2019 Затверджено вченою радою ХНМУ Протокол №5 від 23.05.2019 р. Psychiatry (Part 2) : workbook manual for individual work of students / I. Strelnikova, G. Samardacova, К. Zelenska – Kharkiv, 2019. – 103 p. Копіювання для розповсюдження в будь-якому вигляді частин або повністю можливо тільки з дозволу авторів навчального посібника. CLASS 7. NEUROTIC DISORDERS. CLINICAL FORMS. TREATMENT AND REHABILITATION. POSTTRAUMATIC STRESS DISORDER. TREATMENT AND REHABILITATION. Psychogenic diseases are a large and clinically varied group of diseases resulting from an effect of acute or long-term psychic traumas, which manifest themselves by both mental and somatoneurological disorders and, as a rule, are reversible. Psychogenic diseases are caused by a psychic trauma, i.e. some events which affect significant aspects of existence of the human being and result in deep psychological feelings. These may be subjectively significant events, i.e. those which are pathogenic for the majority of people. Besides, the psyche may be traumatized by conventionally pathogenic events, which cause feelings in an individual because of his peculiar hierarchy of values. Unfavorable psychogenic effects on the human being cause stress in him, i.e. a nonspecific reaction at the physiological, psychological and behavioural levels. Stress may exert some positive, mobilizing influence, but may result in disorganization of the organism activity. The stress, which exerts a negative influence and causes various disturbances and even diseases, is termed distress. Classification of neurotic disorders I. -

The Ndac (Neurology and Psychiatry) Held on 17.12.2011
1. RECOMMENDATIONS OF THE NDAC (NEUROLOGY AND PSYCHIATRY) HELD ON 17.12.2011:- The NDAC (Neurology and Psychiatry) deliberated the proposals on 17.12.2011and recommended the following:- AGENDA DRUG NAME RECOMMENDATIONS NO. Global Clinical Trials The drug has not yet been studied / approved in the country in adults. The tools to assess the cognitive affects are also not included in the 1 Rufinamide protocol except CBCL (child behaviours check list) which is not adequate. In view of above, the proposal to conduct the study in children 1 to 4 years of age was not recommended. DEFFREED Recommended for granting permission for the conduct of proposed study subject to following conditions: That subject from extension study 01050256 2 Lurasidone HCl should only be rolled over to the proposed study. Patients aged 18 to 65 years only should be included in the study. Since the study involves use of placebo without rescue medication in one arm in patients with 3 OPC-34712 acute schizophrenia, the proposal was not recommended for grant of permission. Since the study involves use of placebo without rescue medication in one arm in patients with 4 OPC-34712 acute schizophrenia, the proposal was not recommended for grant of permission. Recommended for granting permission for the 5 BMS-820836 conduct of proposed study. Patients aged 18 to 65 years only should be included in the study. Recommended for granting permission for the 6 BMS-820836 conduct of proposed study. Patients aged 18 to 65 years only should be included in the study. There were earlier studies conducted in USA which had failed as there was no statistically significant effect of the study drug at dose level of 5, 10 mg per day over placebo. -
Cphi & P-MEC China Exhibition List展商名单version版本20180116
CPhI & P-MEC China Exhibition List展商名单 Version版本 20180116 Booth/ Company Name/公司中英文名 Product/产品 展位号 Carbosynth Ltd E1A01 Toronto Research Chemicals Inc E1A08 SiliCycle Inc. E1A10 SA TOURNAIRE E1A11 Indena SpA E1A17 Trifarma E1A21 LLC Velpharma E1A25 Anuh Pharma E1A31 Chemclone Industries E1A51 Hetero Labs Limited E1B09 Concord Biotech Limited E1B10 ScinoPharm Taiwan Ltd E1B11 Dongkook Pharmaceutical Co., Ltd. E1B19 Shenzhen Salubris Pharmaceuticals Co., Ltd E1B22 GfM mbH E1B25 Leawell International Ltd E1B28 DCS Pharma AG E1B31 Agno Pharma E1B32 Newchem Spa E1B35 APEX HEALTHCARE LIMITED E1B51 AMRI E1C21 Aarti Drugs Limited E1C25 Espee Group Innovators E1C31 Ruland Chemical Co., Ltd. E1C32 Merck Chemicals (Shanghai) Co., Ltd. E1C51 Mediking Pharmaceutical Group Ltd E1C57 珠海联邦制药股份有限公司/The United E1D01 Laboratories International Holdings Ltd. FMC Corporation E1D02 Kingchem (Liaoning) Chemical Co., Ltd E1D10 Doosan Corporation E1D22 Sunasia Co., Ltd. E1D25 Bolon Pharmachem Co., Ltd. E1D26 Savior Lifetec Corporation E1D27 Alchem International Pvt Ltd E1D31 Polish Investment and Trade Agency E1D57 Fischer Chemicals AG E1E01 NGL Fine Chem Limited E1E24 常州艾柯轧辊有限公司/ECCO Roller E1E25 Linnea SA E1E26 Everlight Chemical Industrial Corporation E1E27 HARMAN FINOCHEM E1E28 Zhechem Co Ltd E1F01 Midas Pharma GmbH Shanghai Representativ E1F03 Supriya Lifescience Ltd E1F10 KOA Shoji Co Ltd E1F22 NOF Corporation E1F24 上海贺利氏工业技术材料有限公司/Heraeus E1F26 Materials Technology Shanghai Ltd. Novacyl Asia Pacific Ltd E1F28 PharmSol Europe Limited E1F32 Bachem AG E1F35 Louston International Inc. E1F51 High Science Co Ltd E1F55 Chemsphere Technology Inc. E1F57a PharmaCore Biotech Co., Ltd. E1F57b Rockwood Lithium GmbH E1G51 Sarv Bio Labs Pvt Ltd E1G57 抗病毒类、抗肿瘤类、抗感染类和甾体类中间体、原料药和药物制剂及医药合约研发和加工服务 上海创诺医药集团有限公司/Shanghai Desano APIs and Finished products of ARV, Oncology, Anti-infection and Hormone drugs and E1H01 Pharmaceuticals Co., Ltd. -

Alzheimer's Disease: Overview and Treatment Options
Alzheimer’s Disease: Overview and Treatment Options Jillian Blair Thomas, Pharm.D. Candidate 2013 Michael W. Frye, Pharm.D. Candidate 2013 Bernie R. Olin, Pharm.D., Associate Clinical Professor and Director Drug Information Center Auburn University, Harrison School of Pharmacy Universal Activity #: 0178-0000-12-114-H04-P | 1.5 credit hours (.15 CEUs) | Expires Dec. 12, 2015 Alabama Pharmacy Association | 334.271.4222 | www.aparx.org | [email protected] Introduction toxicity. The rate of progression of dementia may be slowed by controlling hypertension. Diabetes Alzheimer’s disease (AD) is the most may increase the risk of dementia through common cause of dementia in the United States multiple mechanisms. Glucose metabolites may affecting an estimated 5.4 million people, with have a toxic effect on the brain, specifically over 60% of those diagnosed being women. structures of the hippocampus, and vasculature. Worldwide, an estimated 24.3 million, possibly Insulin itself may increase the risk of AD due to as many as 35 million, people have been disturbances in insulin signaling pathways 1-6 diagnosed with Alzheimer’s disease. It is peripherally and in the brain.2,5 Insulin may also estimated that by the year 2050 between 13 and have a role in the regulation of metabolism of Aβ 2, 3 16 million Americans will be affected by AD. and tau protein.2 Alzheimer’s disease is more prevalent in African Dominantly inherited forms of AD 1 Americans and Hispanics than in whites. The account for only a small percentage of cases; majority of patients with AD are 65 years or however, there is a definite genetic link.1,2,5 older at the time of diagnosis, but about 5% of There is a high familial occurrence of AD linked patients diagnosed with AD are under the age of to autosomal dominant traits on chromosomes 65. -

(12) Patent Application Publication (10) Pub. No.: US 2014/0051633 A1 Harding Et Al
US 20140051633A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2014/0051633 A1 Harding et al. (43) Pub. Date: Feb. 20, 2014 (54) HEPATOCYTE GROWTH FACTOR MIMICS (60) Provisional application No. 61/706,567, filed on Sep. AS THERAPEUTICAGENTS 27, 2012, provisional application No. 61/706,437, filed on Sep. 27, 2012. (71) Applicant: Washington State University, Pullman, WA (US) Publication Classification (72) Inventors: Joseph W. Harding, Pullman, WA (US); John W. Wright, Pullman, WA (US); (51) Int. C. Caroline C. Benoist, Nashville, TN C07K5/065 (2006.01) (US); Leen H. Kawas, Pullman, WA (52) U.S. C. (US); Gary A. Wayman, Pullman, WA CPC .................................. C07K5/06078 (2013.01) (US) USPC ........................................................... S14/95 (73) Assignee: Washington State University, Pullman, WA (US) (57) ABSTRACT (21) Appl. No.: 14/038,973 (22) Filed: Sep. 27, 2013 Small molecule, peptidic hepatocyte growth factors mimics, which act as both mimetics and antagonists, have been gen Related U.S. Application Data erated. These molecules have been shown or predicted to have (63) Continuation-in-part of application No. PCT/US2012/ therapeutic potential for numerous pathologies including 031815, filed on Apr. 2, 2012. dementia (e.g. Alzheimer's) and Parkinson's disease. Patent Application Publication Feb. 20, 2014 Sheet 1 of 40 US 2014/0051633 A1 a --- --- - - -s ------- CS -8- Scopolarine s -k- Oii exa-ow " -: N. Sks s i. Y s re. Dihexa-high as: ^ N.-- st-scs:- -------------------------------------------------------------------------------------------. Acquisition (days) Figure 1A -8-CSF -- -8- Scopoanine i. --- - -k- Dihexa-ow -- hexa-high reta-or 2 3 4 5 6 7 8 Acquisition (days) Figure 1B -8- vici-treated -- hexa Acquisition (days) Figure 1C Patent Application Publication Feb. -

Dementia Summary
Agency for Healthcare Research and Quality Evidence Report/Technology Assessment Number 97 Pharmacological Treatment of Dementia Summary Introduction 1. Does pharmacotherapy for dementia syndromes improve cognitive symptoms and The focus of this review is the pharmacological outcomes? treatment of dementia. Pharmacotherapy is often 2. Does pharmacotherapy delay cognitive the central intervention used to improve deterioration or delay disease onset of symptoms or delay the progression of dementia dementia syndromes? syndromes. The available agents vary with respect 3. Are certain drugs, including alternative to their therapeutic actions, and are supported by medicines (non-pharmaceutical), more varying levels of evidence for efficacy. This report effective than others? is a systematic evaluation of the evidence for 4. Do certain patient populations benefit more pharmacological interventions for the treatment from pharmacotherapy than others? of dementia in the domains of cognition, global 5. What is the evidence base for the treatment function, behavior/mood, quality of life/activities of ischemic vascular dementia (VaD)? of daily living (ADL) and caregiver burden. This review considers different types of Many medications have been studied in dementia populations (not just Alzheimer’s dementia patients. These agents can be classified Disease [AD]) in subjects from both community into three broad categories: and institutional settings. The studies eligible in 1. Cholinergic neurotransmitter modifying this systematic review were restricted to parallel agents, such as acetylcholinesterase inhibitors. RCTs of high methodological quality. 2. Non-cholinergic neurotransmitters/ neuropeptide modifying agents. Methods 3. Other pharmacological agents. A team of content specialists was assembled Although only five agents have been approved from both international and local experts. -
Pure Pricing List
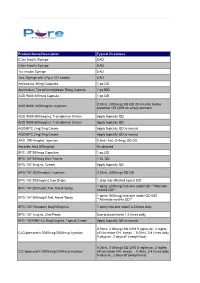
Product Name/Description Typical Directions 0.3cc Insulin Syringe UAD 0.5cc Insulin Syringe UAD 1cc Insulin Syringe UAD 1mL Syringe with 27g x 1/2" needle UAD Amlexanox 40mg Capsules 1 po QD Ammonium Tetrathiomolybdate 50mg Capsule 1 po BID AOD 9604 600mcg Capsule 1 po QD 0.25mL (300mcg) SQ QD 30 minutes before AOD 9604 1200mcg/mL Injection breakfast OR QHS on empty stomach AOD 9604 600mcg/mL Transdermal Cream Apply topically QD AOD 9604 600mcg/mL Transdermal Cream Apply topically QD AOD/BPC 2mg/2mg Cream Apply topically QD to wound AOD/BPC 2mg/2mg Cream Apply topically QD to wound ARA 290 4mg/mL Injection 0.5mL-1mL (2-4mg) SQ QD Ascorbic Acid 500mg/mL As directed BPC-157 500mcg Capsules 1 po QD BPC-157 500mcg Mini-Troche 1 SL QD BPC-157 2mg/mL Cream Apply topically QD BPC-157 2000mcg/mL Injection 0.25mL (500mcg) SQ QD BPC-157 200mcg/mL Eye Drops 1 drop into affected eye(s) BID 1 spray (200mcg) into one nostril QD **Alternate BPC-157 200mcg/0.1mL Nasal Spray nostrils QD** 1 spray (500mcg) into one nostril QD-BID BPC-157 500mcg/0.1mL Nasal Spray **Alternate nostrils QD** BPC-157/ Synapsin 5mg/50mg/mL 1 spray into one nostril 2-3 times daily BPC-157 1mg/mL Oral Paste Use to brush teeth 1-2 times daily BPC-157/GHK-Cu 2mg/2mg/mL Topical Cream Apply topically QD to wound 0.05mL (100mcg) SQ QHS 5 nights on, 2 nights CJC/Ipamorelin 2000mcg/2000mcg Injection off (Increase GH, sleep)… 0.05mL 3-4 times daily 5 days on, 2 days off (weight loss) 0.05mL (100mcg) SQ QHS 5 nights on, 2 nights CJC/Ipamorelin 2000mcg/2000mcg Injection off (Increase GH, -

PEPTIDE INFORMATION Contact Kitt Burkley for Pricing 855-762-4878
PEPTIDE INFORMATION Contact Kitt Burkley for pricing 855-762-4878 AOD 9604 AOD 9604 works by mimicking the way natural HGH regulates fat metabolism but without the adverse effects on blood sugar or growth that is seen with unmodified HGH. Studies have suggested that AOD 9604 has an ability to stimulate lipolysis and inhibit lipogenesis. In addition AOD 9604 possesses other regenerative properties associated with growth hormone. Trials have been undertaken to determine the possible application of AOD 9604 in osteoarthritis and hypercholesterolemia, as well as for bone and cartilage repair. Results of oral glucose tolerance testing have demonstrated that AOD 9604 has no negative effect on carbohydrate metabolism, and it does not affect serum IGF-1 levels. AOD 9604 has an excellent safety profile and is generally well tolerated. It has attained Human GRAS status in the US. AOD 9604 600mcg/ml Cream – 30g AOD 9604* 0.5mg Troche BPC-157 (Body Protection Compound) BPC-157 promotes muscle and tendon healing, increases angiogenesis and decreases inflammatory response. Produces more type 1 collagen and has excellent potential for diabetic wound healing. BPC- 157 is highly stable, resistant to both hydrolysis and enzyme digestion. It can be given orally as well as administered subcutaneously or intramuscularly – no carrier molecule is required. This peptide has significant anti-inflammatory modulating effects. BPC-157 promotes tissue healing through signaling pathways – it provides specific response to particular injury thru cell signaling. BPC- 157 can be used continuously as body protective, or for specific period to treat body injury. BCP-157 is in clinical trials for IBD. -

EVIDENCE-BASED REVIEW of STROKE REHABILITATION (18Th
EVIDENCE-BASED REVIEW OF STROKE REHABILITATION (18th Edition) Robert Teasell MD1, Andreea Cotoi MSc1, Jeffrey Chow MSc1, Joshua Wiener BHSc1, Alice Iliescu BSc1, Norhayati Hussein MBBS, Norine Foley MSc, Katherine Salter PhD (cand.) 1Collaboration of Rehabilitation Research Institute, Parkwood Institute Research, Lawson Health Research Institute, St. Joseph’s Health Care London, London, Ontario Executive Summary The Stroke Rehabilitation Evidence-Based Review (SREBR) reviews techniques, therapies, devices, procedures and medications associated with stroke rehabilitation. The purpose of the Evidence-Based Review of Stroke Rehabilitation was to fulfil the 12th recommendation of The Stroke Rehabilitation Consensus Panel Report that supported the continuing review of stroke rehabilitation research with the “purpose of maintaining timely and accurate information on effective stroke rehabilitation, identifying ideas for further research, supporting continuous peer-review and encouraging improved evidence-based practice.” The aim of the SREBR was to: • Be an up-to-date review of the current evidence in stroke rehabilitation. • Provide a comprehensive and accessible review to facilitate best-practice. • Provide specific conclusion based on evidence that could be used to help direct stroke care at the bedside and at home. Since its original publication in April 2002, the SREBR has undergone eighteen major revisions and now includes articles published up to December 2016. To date, we have included over 2,300 randomized controlled trials (RCTs). Methods For the first edition of the SREBR a literature search using multiple databases (MEDLINE, EBASE, MANTIS, PASCAL, and Sci Search) was conducted to identify all potential trials published from 1970- 2001, regardless of study design. The search was restricted to the English language and excluded animal studies.