Albendazole 40Mg/Ml Oral
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Study of the Partial and Exhaustive Intramolecular Dehydration of D-Sorbitol
Available online at http://journal-of-agroalimentary.ro Journal of Journal of Agroalimentary Processes and Agroalimentary Processes and Technologies 2013, 19(2), 259-270 Technologies Study of the partial and exhaustive intramolecular dehydration of D-sorbitol Ileana Cocan *, Monica Viorica Negrea, Ionel Vasile Jianu ”Dunărea de Jos” University of Galati, Faculty of Food Science and Engineering, Domnească Street, 47, RO-800008, Galati, Romania Received: 07 February 2013; Accepted: 09 March 2013 ______________________________________________________________________________________ Abstract Sweetener, humectants, sequestrant, texturizer, stabilizer, bulking agent, D-sorbitol represents a leading product in the processing of the polysorbate class. The scope of this work is to obtain details on the preparation and accession of D-sorbitol mono- and dianhydride as a method of chemical protection of primary and secondary hydroxyl functional groups (1:4) (3:6) as part of the strategy of controlled processing of polysorbates containing “homogeneous” polyoxyethylenic chains (n = 3, 6, 9, 18). This work is investigating the dependence of yield of internal dehydration (partial and/or exhaustive, direct of D-sorbitol) on temperature (100-160°C and 120-200°C , respectively ) and duration (5–35 minutes and 10–100 minutes , respectively ) for the processing of 1,4-sorbitan and 2,5-isosorbide. The evolution of the hydroxyl number (mg KOH/g D-sorbitol) (mg KOH/g 1,4-sorbitan) related to the theoretical (initial) value was followed and optimal values for the yield and dehydration parameters were established. Also in this work, the mathematical modeling of the dependence curves experimentally registered is realized. Keywords : izosorbide, D-sorbitol, D-glucitol, D-sorbit, 1,4-sorbitan ______________________________________________________________________________________ 1. -

Sorbitol Malabsorption 1/2
Internal Medicine and Gastroenterology Clinic Dres. Mares,M.Hanig,Mares, Hanig, Rambow, S.Blau, Blau, M.Seip, Hanig, Seip, A.Borchers,Blau,Kirchner, Hochstr. Hochstr. Hochstr. 43, 60313 43, 43, 60313 60313 Frankfurt/Main, Frankfurt/M., Frankfurt/M., www.gastroenterologie-ffm.de www.gastroenterologie-ffm.de www.gastroenterologie-ffm.de Nutrition hints for sorbitol malabsorption 1/2 Sorbitol intolerance Sorbitol incompatibility 1. What does this mean for nutrition? Avoid sorbitol as a sweetener and foods high in sorbitol. Small amount of sorbitol can often be tolerated (at most 10-10 g per day, sometimes less). In order to prevent general upper GI problems, easily digestible foods that do not cause gas are recommended. 2. Key points about foods – Avoid sorbitol and foods containing sorbitol – easily digestible food that does not cause gas 3. vegetables and fruits low in fibre (see 5) 4. Choice of foods The following foods are high in sorbitol and not suitable: – Sorbitol as sweetener: e.g. Sionon, Flarom, diabetic sweetener – Dietetic foods produced with sorbitol: for example, diabetic marmalades, diabetic sweets, diabetic baked goods – Types of fruit which have a naturally high sorbitol content: Apples, pears, cherries, prunes, plums, dates fruits with seeds, such as mirabelles, apricots, nectarines, and all fruit syrups made from these types of fruits – Types of fruit which have a naturally low sorbitol content: Berry fruits such as strawberries, raspberries, blackberries, blueberries, currants, gooseberries, citrus fruits, bananas, pineapples, kiwis – Sorbitol as a coating for: sultanas, raisins, and dried fruit or candied fruit – Sorbitol in sweets: chewing gum, jelly babies, jelly fruits, candies, chocolate bars, filled wafers, chocolate, etc. -

Sodium Benzoate Inhibits Growth of Or Inactivates Listeria Monocytogenes
525 Journal of Food Protection, Vol. 51, No. 7, Pages 525-530 (July 1988) Copyright© International Association of Milk, Food and Environmental Sanitarians Sodium Benzoate Inhibits Growth of or Inactivates Listeria monocytogenes MOUSTAFA A. EL-SHENAWY and ELMER H. MARTH* Department of Food Science and The Food Research Institute, University of Wisconsin-Madison, Madison, Wisconsin 53706 (Received for publication February 26, 1988) Downloaded from http://meridian.allenpress.com/jfp/article-pdf/51/7/525/1658063/0362-028x-51_7_525.pdf by guest on 28 September 2021 ABSTRACT Recognition of Listeria monocytogenes as an agent of foodborne disease has increased in the last few years. The The ability of Listeria monocytogenes to grow or survive was pathogen'can cause abortion in pregnant women as well as determined using tryptose broth at pH 5.6 or 5.0, supplemented meningitis in newborn infants and immunocompromised with 0, 0.05. 0.1, 0.15. 0.2. 0.25 or 0.3% sodium benzoate, and adults (17,26,31). Also, this bacterium is pathogenic for incubated at 4,13,21 or 35°C. The bacterium grew in benzoate- animals and can cause abortion (33) and mastitis (15). L. free controls under all conditions except at 4°C and pH 5.0. At pH 5.6 and 4°C, after 60 d, L. monocytogenes (initial population ca. monocytogenes can be transmitted from infected animals to 103/ml) was inactivated by 0.2, 0.25 and 0.3% sodium benzoate. humans (16,21,24,25) and also can be transmitted to hu Other concentrations of benzoate permitted slight growth during mans through consumption of some foods of animal origin. -

Copyrighted Material
Index Abbreviations receptor sites 202, 211 weak 122 amino acids, Table 500–1 muscarinic 413 weak, pH calculation 147–8 nucleic acids 551 nicotinic 413 Acid–base nucleotides, Table 551 as quaternary ammonium salt 202 catalysis, enzymes 516 peptides and proteins 504 Acetylcholinesterase equilibria 121 phosphates and diphosphates 277 enzyme mechanism 519–21 interactions, predicting 155–7 structural, Table 14 hydrolysis of acetylcholine 279 Acidic reagents, Table 157 ACE (angiotensin-converting enzyme) inhibitors 279 Acidity enzyme action 532 Acetyl-CoA carboxylase, in fatty acid acidity constant 122 inhibitors 532 biosynthesis 595 bond energy effects 125 Acetal Acetyl-CoA (acetyl coenzyme A) definition 121 in etoposide 233 as acylating agent 262 electronegativity effects 125 formation 229 carboxylation to malonyl-CoA 595, hybridization effects 128 polysaccharides as polyacetals 232 609 inductive effects 125–7 as protecting group 230 Claisen reaction 381 influence of electronic and structural Acetal and ketal enolate anions 373 features 125–34 cyclic, as protecting groups 481 in Krebs cycle 585 and leaving groups, Table 189 groups in sucrose 231 from β-oxidation of fatty acids 388 pKa values 122–5 Acetaldehyde, basicity 139 as thioester 262, 373 resonance / delocalization effects 129–34 Acetamide, basicity 139 Acetylene, bonding molecular orbitals 31 Acidity (compounds) Acetaminophen, see paracetamol Acetylenes, acidity 128 acetone 130 Acetoacetyl-CoA, biosynthesis from N-Acetylgalactosamine, in blood group acetonitrile 365 acetyl-CoA 392 -

Polyols Have a Variety of Functional Properties That Make Them Useful Alternatives to Sugars in Applications Including Baked Goods
Polyols have a variety of functional properties that make them useful alternatives to sugars in applications including baked goods. Photo © iStockphoto.com/Synergee pg 22 09.12 • www.ift.org BY LYN NABORS and THERESA HEDRICK SUGAR REDUCTION WITH Polyols Polyols are in a unique position to assist with reduced-sugar or sugar-free reformulations since they can reduce calories and complement sugar’s functionality. ugar reduction will be an important goal over the of the product’s original characteristics may still be main- next few years as consumers, government, and in- tained with the replacement of those sugars by polyols. Sdustry alike have expressed interest in lower-calorie In addition, excellent, good-tasting sugar-free products and lower-sugar foods. The 2010 Dietary Guidelines for can be developed by using polyols. Polyols are in a unique Americans put a strong emphasis on consuming fewer position to assist with reduced-sugar or sugar-free refor- calories and reducing intake of added sugars. The In- mulations; since they are only partially digested and ab- stitute of Medicine (IOM) held a public workshop in sorbed, they can reduce calories and complement sugar’s November 2010 to discuss ways the food industry can functionality. Polyols provide the same bulk as sugars and use contemporary and innovative food processing tech- other carbohydrates. Additionally, polyols have a clean, nologies to reduce calorie intake in an effort to reduce sweet taste, which is important since consumers are not and prevent obesity, and in October 2011 recommended likely to sacrifice taste for perceived health benefits. Poly- front-of-package labeling that includes rating the product ols have a host of other functional properties that make based on added sugars content. -

New Naphthalene Whole-Cell Bioreporter for Measuring and Assessing Naphthalene in Polycyclic Aromatic Hydrocarbons Contaminated Site
Accepted Manuscript New naphthalene whole-cell bioreporter for measuring and assessing naphthalene in polycyclic aromatic hydrocarbons contaminated site Yujiao Sun, Xiaohui Zhao, Dayi Zhang, Aizhong Ding, Cheng Chen, Wei E. Huang, Huichun Zhang PII: S0045-6535(17)31248-1 DOI: 10.1016/j.chemosphere.2017.08.027 Reference: CHEM 19725 To appear in: ECSN Received Date: 17 April 2017 Revised Date: 22 July 2017 Accepted Date: 7 August 2017 Please cite this article as: Sun, Y., Zhao, X., Zhang, D., Ding, A., Chen, C., Huang, W.E., Zhang, H., New naphthalene whole-cell bioreporter for measuring and assessing naphthalene in polycyclic aromatic hydrocarbons contaminated site, Chemosphere (2017), doi: 10.1016/j.chemosphere.2017.08.027. This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain. ACCEPTED MANUSCRIPT 1 New naphthalene whole-cell bioreporter for measuring and assessing 2 naphthalene in polycyclic aromatic hydrocarbons contaminated site 3 Yujiao Sun a, Xiaohui Zhao a,b* , Dayi Zhang c, Aizhong Ding a, Cheng Chen a, Wei E. 4 Huang d, Huichun Zhang a. 5 a College of Water Sciences, Beijing Normal University, Beijing, 100875, PR China 6 b Department of Water Environment, China Institute of Water Resources and 7 Hydropower Research, Beijing, 100038, China 8 c Lancaster Environment Centre, Lancaster University, Lancaster, LA1 4YQ, UK 9 d Kroto Research Institute, University of Sheffield, Sheffield, S3 7HQ, United 10 Kingdom 11 12 Corresponding author 13 Dr Xiaohui Zhao 14 a College of Water Sciences, Beijing Normal UniversiMANUSCRIPTty, Beijing 100875, P. -

CHM 224 Test 2 Chapters 13, 14, Organometallics, 20 NAME
CHM 224 NAME: Test 2 Chapters 13, 14, organometallics, 20 1. Answer the following 3 questions: A. Brandy is 60% alcohol. What is its proof? B. This alcohol can be created by heating wood chips: C. This alcohol is referred to as "rubbing alcohol": 2. The three compounds below have nearly identical molecular weights. Arrange them according to their expected boiling points from highest >>> lowest. OH OH O A B C 3. Match the pKa values with the compounds provided: pKa's = 7.2, 8.0, 10.3 OH OH OH NO2 CH3 CN 4. What is the expected major product of the following reaction? 1. 1 equivalent BH3•THF 2. NaOH, H2O2 5. Which of the following compounds is expected to fail to react with KMnO4 (may be more than one)? A. 1-methylcyclopentanol B. 2-methyl-3-hexanol C. 4-ethyl-4-heptanol D. 3-bromo-1-butanol 6. Provide the IUPAC name for the following compound: OH Br 7. Which ONE of the following statements is true? A. ethers are generally water soluble, flammable, and reactive with strong bases B. ethers are generally water insoluble, not flammable, and reactive with strong acids C. ethers are generally water soluble, flammable, and reactive with strong bases D. ethers are generally water insoluble, flammable, and reactive with strong acids 8. Provide a synthetic route for the synthesis of tert-butyl isopropyl ether using the Williamson ether synthesis from an appropriate starting alcohol and alkyl halide. 9. Which one of the following alkenes will form the epoxide below upon treatment with a peroxyacid? O H CH2CH3 H3C CH3 A B C D 10. -

Pichia Process Optimization by Methanol/Sorbitol Co-Feeding – Patrick Fickers - University of Liege
Short Communication Journal of Applied Microbiology and Biochemistry 2020 Vol.4 No.4 Applied Microbiology 2016- Pichia process optimization by methanol/sorbitol co-feeding – Patrick Fickers - University of Liege Patrick Fickers University of Liege Abstract Recombinant protein production driven by AOX1 promoter is at this stage. During the second step, so as to extend biomass challenged by a high oxygen demand and warmth production, production and de-repress the methanol metabolic machinery, a especially in large-scale bioreactor. A reliable solution depends glycerol-limited fed-batch procedure is initiated. This phase on a methanol/sorbitol co-feeding strategy during the induction leads to gradual de-repression of the enzymes necessary for the phase. during this work, transient continuous cultures were first dissimilation of methanol and reduces the time necessary for performed to quantitatively assess the advantages of a the cells to adapt to growth on methanol. Finally, recombinant methanol/sorbitol co-feeding process with a P. pastoris Mut+ protein expression is typically induced by feeding methanol strain bearing a pAOX1-lacZ construct served as a reporter because the sole carbon source. gene. Our results demonstrated that cell-specific oxygen consumption (qO2) might be reduced by decreasing the A fermentation guideline for both Mut+ and Muts strains of P. methanol fraction within the feeding media. Optimal pAOX1 pastoris is available from Invitrogen . Stratton et al. devised a induction was achieved and maintained within the range of protocol for P. pastoris high cell-density fermentation, during 0.45~0.75 C-mol/C-mol of methanol fraction. additionally, the which detailed procedures are provided. -

Study of Genes Relating to Degradation of Aromatic Compounds and Carbon Metabolism in Mycobacterium Sp. Strain KMS
Utah State University DigitalCommons@USU All Graduate Theses and Dissertations Graduate Studies 5-2013 Study of Genes Relating To Degradation of Aromatic Compounds and Carbon Metabolism in Mycobacterium Sp. Strain KMS Chun Zhang Utah State University Follow this and additional works at: https://digitalcommons.usu.edu/etd Part of the Biology Commons, Environmental Sciences Commons, and the Microbiology Commons Recommended Citation Zhang, Chun, "Study of Genes Relating To Degradation of Aromatic Compounds and Carbon Metabolism in Mycobacterium Sp. Strain KMS" (2013). All Graduate Theses and Dissertations. 1532. https://digitalcommons.usu.edu/etd/1532 This Dissertation is brought to you for free and open access by the Graduate Studies at DigitalCommons@USU. It has been accepted for inclusion in All Graduate Theses and Dissertations by an authorized administrator of DigitalCommons@USU. For more information, please contact [email protected]. STUDY OF GENES RELATING TO DEGRADATION OF AROMATIC COMPOUNDS AND CARBON METABOLISM IN MYCOBACTERIUM SP . STRAIN KMS by Chun Zhang A dissertation submitted in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY in Biology Approved: ____________________________ ____________________________ Anne J. Anderson, Ph.D. Dennis L. Welker, Ph.D. Major Professor Committee Member ____________________________ ____________________________ Jeanette M. Norton, Ph.D. Ronald C. Sims, Ph.D. Committee Member Committee Member ____________________________ ____________________________ Charles D. Miller, Ph.D. Mark R. McLellan, Ph.D. Committee Member Vice President for Research and Dean of the School of Graduate Studies UTAH STATE UNIVERSITY Logan, Utah 2013 ii Copyright © Chun Zhang 2013 All Rights Reserved iii ABSTRACT Study of Genes Relating to Degradation of Aromatic Compounds and Carbon Metabolism in Mycobacterium sp . -

Enhanced Protein Production by Sorbitol Co-Feeding with Methanol in Recombinant Pichia Pastoris Strains
Biotechnology and Bioprocess Engineering 22: 767-773 (2017) pISSN 1226-8372 DOI 10.1007/s12257-017-0011-9 eISSN 1976-3816 RESEARCH PAPER Enhanced Protein Production by Sorbitol Co-feeding with Methanol in Recombinant Pichia pastoris Strains Li Chen, Ali Mohsin, Ju Chu, Yingping Zhuang, Yamei Liu, and Meijin Guo Received: 9 January 2017 / Revised: 8 May 2017 / Accepted: 29 August 2017 © The Korean Society for Biotechnology and Bioengineering and Springer 2017 Abstract Pichia pastoris strains carrying 1, 6, 12, and 18 Keywords: sorbitol, co-feeding, multi-copy, Pichia pastoris, copies of the porcine insulin precursor (PIP) gene, were porcine insulin precursor employed to investigate the effects of sorbitol co-feeding with methanol on the physiology of the strains. Multicopy clones of the methylotrophic yeast were generated to vary 1. Introduction the PIP gene dosage and recombinant proteins. Elevated gene dosage increased levels of the recombinant PIP protein The methylotrophic yeast, Pichia pastoris, has been when methanol served as the sole carbon and energy developed to be a highly successful system for large-scale source i.e., an increase of 1.9% for a strain carrying 1 copy, production of functionally active recombinant proteins 42.6% for a strain carrying 6 copies, 34.7% for a strain [1,2]. As a yeast, it is a unique system which has many carrying 12 copies and 80.9% for a strain carrying 18 advantages e.g., its ability to produce foreign proteins at copies, respectively (using sorbitol co-feeding with methanol high levels using one of the strongest and highly regulated during the induction phase). -

Allergens 230118.Xlsx
Code Product Brand Owner Type Pack ABV% Size Ingredient List (Only if <1/2% ABV) 108660 Kopparberg Alcohol Free with mixed Fruit Cider Of Sweden Ltd Cider Packaged 0 500 ml Carbonated Water, Fermented Apples, Juice (Apple, Blackcurrant, Elderberry, Raspberry), Sugar, Acid (Citric Acid), Flavouring, Preservative (Potassium Sorbate), Antioxidant (E224/Sulphite). 109120 Kopparberg Alcohol Free with Strawberry & Lime Cider Of Sweden Ltd Cider Packaged 0 4 ltr Carbonated water, fermented pear or apple juice, sugar, flavouring, juice from other fruit and berries depending on the flavor of the cider, citric acid (E330), potassium sorbate (E202) and sulfite (E224). 109312Heineken 0.0 Heineken Uk Ltd Lager Packaged 0 330 ml Aater, malted barley, hop extract, natural flavourings 105355 Becks Blue Inbev Uk Lager Packaged 0 275 ml Brewing Water, Malted Barley, Hops 700633Coca Cola Zero Sugar Coca Cola Enterprises (Cce) Minerals Draught 0 7 ltr Carbonated Water, Caramel E150d, Phosphric Acid,Sweetners(Aspartame,Acesulfame K)Natural Flavourings inckudign Caffeine, Acidity Regulator,( Sodium Citrate) 700049 Coca Cola (B-I-B) Marstons Minerals Draught 0 7 ltr Carbonated Water, Sugar, Colour (Caramel E150D), Phosphoric Acid, Natural Flavourings Including Caffeine. 700050 Diet Coca Cola (B-I-B) Marstons Minerals Draught 0 7 ltr Carbonated Water, Colour (Caramel E150D), Sweeteners (Aspartame, Acesulfame K), Natural Flavourings Including Caffeine, Phosphoric Acid, Citric Acid. 700008Diet Pepsi (B-I-B) Marstons Minerals Draught 0 7 ltr Water, Colour (Caramel E150D), Acids (Phosphoric Acid, Citric Acid), Flavourings (Including Caffeine), Sweeteners (Aspartame, Acesulfame K), Acidity Regulator (Sodium Citrate), Preservative (Sodium Benzoate), Anti-Foaming Agent (E900). Contains A Source Of Phenylalanine 700009 Pepsi (B-I-B) Marstons Minerals Draught 0 7 ltr Sugar, Water, Colour (Caramel E150D), Acid (Phosphoric Acid), Flavourings (Including Caffeine). -
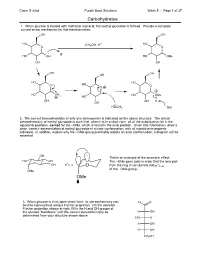
5.01 Carbohydrates Answers.Cdx
Chem S-20ab Purple Book Solutions Week 5 - Page 1 of 37 Carbohydrates 1. When glucose is treated with methanol and acid, the methyl glucoside is formed. Provide a complete curved-arrow mechanism for this transformation: OH OH HO CH OH, H+ HO O 3 O H+ HO OH HO OMe OH OH OH OH OH HO HO O HO O O HO OH HO OMe 2 HO OH OH H OH HOCH3 Sol 2. The correct stereochemistry of only one stereocenter is indicated on the above structure. The actual stereochemistry of methyl glucoside is such that, when it is in a chair form, all of the substituents fall in the equatorial positions, except for the –OMe, which is found in the axial position. Given this information, draw a clear, correct representation of methyl glucoside in a chair conformation, with all substituents properly indicated. In addition, explain why the –OMe group preferably adopts an axial conformation; a diagram will be essential. OH This is an example of the anomeric effect. HO OH The –OMe goes axial in order that the lone pair O * OH C–O O fromtheringOcandonateinto *C–O of the –OMe group. OMe OMe 3. When glucose is in its open-chain form, its stereochemistry can H O best be represented using a Fischer projection. On the skeleton Fischer projection shown at right, fill in the H and OH groups of the glucose "backbone" with the correct stereochemistry as H OH determined from your structure shown above. HO H H OH H OH CH2OH Chem S-20ab Purple Book Solutions Week 5 - Page 2 of 37 Carbohydrates: Mechanisms 1.