Report 27 Quantitative Determination of Pesticide
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Historical Perspectives on Apple Production: Fruit Tree Pest Management, Regulation and New Insecticidal Chemistries
Historical Perspectives on Apple Production: Fruit Tree Pest Management, Regulation and New Insecticidal Chemistries. Peter Jentsch Extension Associate Department of Entomology Cornell University's Hudson Valley Lab 3357 Rt. 9W; PO box 727 Highland, NY 12528 email: [email protected] Phone 845-691-7151 Mobile: 845-417-7465 http://www.nysaes.cornell.edu/ent/faculty/jentsch/ 2 Historical Perspectives on Fruit Production: Fruit Tree Pest Management, Regulation and New Chemistries. by Peter Jentsch I. Historical Use of Pesticides in Apple Production Overview of Apple Production and Pest Management Prior to 1940 Synthetic Pesticide Development and Use II. Influences Changing the Pest Management Profile in Apple Production Chemical Residues in Early Insect Management Historical Chemical Regulation Recent Regulation Developments Changing Pest Management Food Quality Protection Act of 1996 The Science Behind The Methodology Pesticide Revisions – Requirements For New Registrations III. Resistance of Insect Pests to Insecticides Resistance Pest Management Strategies IV. Reduced Risk Chemistries: New Modes of Action and the Insecticide Treadmill Fermentation Microbial Products Bt’s, Abamectins, Spinosads Juvenile Hormone Analogs Formamidines, Juvenile Hormone Analogs And Mimics Insect Growth Regulators Azadirachtin, Thiadiazine Neonicotinyls Major Reduced Risk Materials: Carboxamides, Carboxylic Acid Esters, Granulosis Viruses, Diphenyloxazolines, Insecticidal Soaps, Benzoyl Urea Growth Regulators, Tetronic Acids, Oxadiazenes , Particle Films, Phenoxypyrazoles, Pyridazinones, Spinosads, Tetrazines , Organotins, Quinolines. 3 I Historical Use of Pesticides in Apple Production Overview of Apple Production and Pest Management Prior to 1940 The apple has a rather ominous origin. Its inception is framed in the biblical text regarding the genesis of mankind. The backdrop appears to be the turbulent setting of what many scholars believe to be present day Iraq. -

Genetically Modified Baculoviruses for Pest
INSECT CONTROL BIOLOGICAL AND SYNTHETIC AGENTS This page intentionally left blank INSECT CONTROL BIOLOGICAL AND SYNTHETIC AGENTS EDITED BY LAWRENCE I. GILBERT SARJEET S. GILL Amsterdam • Boston • Heidelberg • London • New York • Oxford Paris • San Diego • San Francisco • Singapore • Sydney • Tokyo Academic Press is an imprint of Elsevier Academic Press, 32 Jamestown Road, London, NW1 7BU, UK 30 Corporate Drive, Suite 400, Burlington, MA 01803, USA 525 B Street, Suite 1800, San Diego, CA 92101-4495, USA ª 2010 Elsevier B.V. All rights reserved The chapters first appeared in Comprehensive Molecular Insect Science, edited by Lawrence I. Gilbert, Kostas Iatrou, and Sarjeet S. Gill (Elsevier, B.V. 2005). All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopy, recording, or any information storage and retrieval system, without permission in writing from the publishers. Permissions may be sought directly from Elsevier’s Rights Department in Oxford, UK: phone (þ44) 1865 843830, fax (þ44) 1865 853333, e-mail [email protected]. Requests may also be completed on-line via the homepage (http://www.elsevier.com/locate/permissions). Library of Congress Cataloging-in-Publication Data Insect control : biological and synthetic agents / editors-in-chief: Lawrence I. Gilbert, Sarjeet S. Gill. – 1st ed. p. cm. Includes bibliographical references and index. ISBN 978-0-12-381449-4 (alk. paper) 1. Insect pests–Control. 2. Insecticides. I. Gilbert, Lawrence I. (Lawrence Irwin), 1929- II. Gill, Sarjeet S. SB931.I42 2010 632’.7–dc22 2010010547 A catalogue record for this book is available from the British Library ISBN 978-0-12-381449-4 Cover Images: (Top Left) Important pest insect targeted by neonicotinoid insecticides: Sweet-potato whitefly, Bemisia tabaci; (Top Right) Control (bottom) and tebufenozide intoxicated by ingestion (top) larvae of the white tussock moth, from Chapter 4; (Bottom) Mode of action of Cry1A toxins, from Addendum A7. -

US5264213.Pdf
||||||||||||||| US005264213A United States Patent (19) 11) Patent Number: 5,264,213 Shibahara et al. 45 Date of Patent: Nov. 23, 1993 (54) PROCESS FOR PREPARING HIGHLY 57-145,861, published Sep. 9, 1982 (Derwent Abstract ACTIVE WATER-DESPERSIBLE PESTC DES 82-8883i E/42). (75) Inventors: Tetsuya Shibahara, Hatano; Naohiko Shimazaki, et al., Japanese Patent Application Kondo, Atsugi; Jun Kato, Susono, all 59-212,462, published Dec. 1, 1984 (Derwent Abstract of Japan 85-015809/03). Haga, et al., Japanese Patent Application 60-193,960, 73) Assignee: Dowelanco, Indianapolis, Ind. published Oct. 2, 1985 (Derwent Abstract (21) Appl. No.: 377,026 85-285507/46). Haga, et al., Japanese Patent Application 62-155,248, (22 Filed: Jul. 7, 1989 published Jul. 10, 1987 (Derwent Abstract (30) Foreign Application Priority Data 87-231613/33). Jul. 8, 1988 (JP) Japan ................................ 63-168672 Haga, et al., Japanese Patent Application 62-155,249, published Jul. 10, 1987 (Derwent Abstract 51) Int. Cl........................ CO7C 127/22; A01N 9/20 87-231614/33). 52 U.S.C. .................................... 424/409; 514/351; Nagasaki, et al., Japanese Patent Application 514/522; 514/594; 546/300; 564/44 58) Field of Search .......................... 546/300; 564/44; 62-195,365, published Aug. 28, 1989 (Derwent Abstract 514/351, 522,554; 424/409 87-280996/40). (56) References Cited Primary Examiner-Lester L. Lee U.S. PATENT DOCUMENTS Attorney, Agent, or Firm-D. Wendell Osborne 3,989,842 11/1976 Wellinger et al. 3,989,942 11/1976 Wellinga et al..................... 424/322 (57) ABSTRACT 4,139,636 2/1979 Sirrenberg et al. -
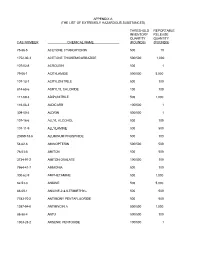
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

744 Hydrolysis of Chiral Organophosphorus Compounds By
[Frontiers in Bioscience, Landmark, 26, 744-770, Jan 1, 2021] Hydrolysis of chiral organophosphorus compounds by phosphotriesterases and mammalian paraoxonase-1 Antonio Monroy-Noyola1, Damianys Almenares-Lopez2, Eugenio Vilanova Gisbert3 1Laboratorio de Neuroproteccion, Facultad de Farmacia, Universidad Autonoma del Estado de Morelos, Morelos, Mexico, 2Division de Ciencias Basicas e Ingenierias, Universidad Popular de la Chontalpa, H. Cardenas, Tabasco, Mexico, 3Instituto de Bioingenieria, Universidad Miguel Hernandez, Elche, Alicante, Spain TABLE OF CONTENTS 1. Abstract 2. Introduction 2.1. Organophosphorus compounds (OPs) and their toxicity 2.2. Metabolism and treatment of OP intoxication 2.3. Chiral OPs 3. Stereoselective hydrolysis 3.1. Stereoselective hydrolysis determines the toxicity of chiral compounds 3.2. Hydrolysis of nerve agents by PTEs 3.2.1. Hydrolysis of V-type agents 3.3. PON1, a protein restricted in its ability to hydrolyze chiral OPs 3.4. Toxicity and stereoselective hydrolysis of OPs in animal tissues 3.4.1. The calcium-dependent stereoselective activity of OPs associated with PON1 3.4.2. Stereoselective hydrolysis commercial OPs pesticides by alloforms of PON1 Q192R 3.4.3. PON1, an enzyme that stereoselectively hydrolyzes OP nerve agents 3.4.4. PON1 recombinants and stereoselective hydrolysis of OP nerve agents 3.5. The activity of PTEs in birds 4. Conclusions 5. Acknowledgments 6. References 1. ABSTRACT Some organophosphorus compounds interaction of the racemic OPs with these B- (OPs), which are used in the manufacturing of esterases (AChE and NTE) and such interactions insecticides and nerve agents, are racemic mixtures have been studied in vivo, ex vivo and in vitro, using with at least one chiral center with a phosphorus stereoselective hydrolysis by A-esterases or atom. -

Use Date Issued: September 2006 ______
United States Environmental Protection Agency Office of Prevention, Pesticides and Toxic Substances (7505P) _______________________________________________________ Pesticide Fact Sheet Name of Chemical: Metofluthrin Reason for Issuance: New Chemical Nonfood Use Date Issued: September 2006 _______________________________________________________ Description of Chemical IUPAC name: 2,3,5,6-tetrafluoro-4-(methoxymethyl)benzyl (EZ)- (1RS,3RS;1RS,3SR)-2,2-dimethyl-3-prop-1- enylcyclopropanecarboxylate CAS name: [2,3,5,6-tetrafluoro-4-(methoxymethyl)phenyl]methyl 2,2-dimethyl-3-(1-propenyl)cyclopropanecarboxylate Common Name: Metofluthrin Empirical Formula: C18H20F4O3 EPA Chemical Code: 109709 Chemical Abstracts Service (CAS) Number: 240494-70-6 Chemical Class: Pyrethroid ester Registration Status: New Chemical, nonfood use Pesticide Type: Insecticide repellent not applied to human skin U.S.Technical Registrant : Sumitomo Chemical Company, LTD. 1330 Dillon Hghts. Ave. Baltimore, MD 21228 Use Pattern and Formulations Currently there are two end use products being proposed for metofluthrin. DeckMate ™ Mosquito Repellent Strip is an impregnated paper strip (~3,528 cm2) containing 1.82 percent metofluthrin as the active ingredient. The product also contains Bitrex ™ to discourage oral exposure to children or animals. The product is for use on patios, campsites, decks, cabanas, and other outdoor areas. One strip is applied per 10 ft × 10 ft outdoor area. Indoors the application rate is two strips per 50 m3. There are approximately 200 mg of metofluthrin initially in the strip. The strips can provide up to one week of protection Metofluthrin evaporates readily and therefore requires no external heat. Norm 1- is a personal outdoor insect repellent product consisting of a holder containing a replaceable cartridge insert coated with up to 50 mg of metofluthrin. -

Pesticide Resistance in Bed Bugs Everywhere!!!!!
2/24/2018 Pesticide Resistance in Bed bugs were virtually eradicated from the U.S. in Bed Bugs the post WWII era due to DDT and other powerful Shujuan (Lucy) Li insecticides. University of Arizona Alvaro Romero New Mexico State University 2 By the 1960s, bed bugs had developed resistance Public housing Apartments to DDT, methoxychlor and analogues, BHC, Schools dieldrin and analogues , and pyrethrins ( Busvine 1958, Hospitals Nursing homes Cwilich & Mer 1957, Mallis and Miller 1964 ) . Homes Transportation Child care Medical facilities Hotels & motels Health care facilities Airports Movie theaters Department stores Products, vendors, or commercial services mentioned or pictured in this seminar are for Everywhere!!!!! illustrative purposes only and are not meant to be endorsements. 3 4 University of Arizona; Arizona Pest Management Center 1 2/24/2018 Possible reasons for treatment failure? Missed some Clutter Reintroduction Have you seen these after treatments? 5 6 Dose - response assays for field - collected strains Bed bugs survived direct insecticide sprays 99 deltamethrin 90 Ft. Dix F1 50 ) e l a c 10 s t CIN1 i b o 1.0 r p ( y t i l a t r 99 - cyhalothrin o m e 90 g a t n Resistance ratio (RR) at least 6,000 !!! e c Ft. Dix r 50 e P 10 CIN1 Suspend® ( Deltamethrin ) 1.0 10 -7 10 -6 10 -5 10 -4 10 -3 10 -2 10 -1 10 0 10 1 10 2 10 3 10 4 Treatment (mg active ingredient/cm 2 ) Products, vendors, or commercial services mentioned or pictured in this seminar are for illustrative purposes only and are not meant Romero et al. -

Recommended Classification of Pesticides by Hazard and Guidelines to Classification 2019 Theinternational Programme on Chemical Safety (IPCS) Was Established in 1980
The WHO Recommended Classi cation of Pesticides by Hazard and Guidelines to Classi cation 2019 cation Hazard of Pesticides by and Guidelines to Classi The WHO Recommended Classi The WHO Recommended Classi cation of Pesticides by Hazard and Guidelines to Classi cation 2019 The WHO Recommended Classification of Pesticides by Hazard and Guidelines to Classification 2019 TheInternational Programme on Chemical Safety (IPCS) was established in 1980. The overall objectives of the IPCS are to establish the scientific basis for assessment of the risk to human health and the environment from exposure to chemicals, through international peer review processes, as a prerequisite for the promotion of chemical safety, and to provide technical assistance in strengthening national capacities for the sound management of chemicals. This publication was developed in the IOMC context. The contents do not necessarily reflect the views or stated policies of individual IOMC Participating Organizations. The Inter-Organization Programme for the Sound Management of Chemicals (IOMC) was established in 1995 following recommendations made by the 1992 UN Conference on Environment and Development to strengthen cooperation and increase international coordination in the field of chemical safety. The Participating Organizations are: FAO, ILO, UNDP, UNEP, UNIDO, UNITAR, WHO, World Bank and OECD. The purpose of the IOMC is to promote coordination of the policies and activities pursued by the Participating Organizations, jointly or separately, to achieve the sound management of chemicals in relation to human health and the environment. WHO recommended classification of pesticides by hazard and guidelines to classification, 2019 edition ISBN 978-92-4-000566-2 (electronic version) ISBN 978-92-4-000567-9 (print version) ISSN 1684-1042 © World Health Organization 2020 Some rights reserved. -

Application of ACQUITY TQD for the Analysis of Pesticide Residues
[ application application note note ] ] APPLICATION OF ACQUIT Y TQD FOR THE ANALYSIS OF PESTICIDE RESIduES IN Baby FOOD James Morphet and Peter Hancock Waters Corporation, Manchester, UK OVERVIEW This application note assesses the suitability of the Waters® ACQUITY™ TQD for tandem quadrupole-based analysis of pesticide residue in baby food. Polarity switching, differing dwell times and ion ratio robustness will also be assessed. INTRODUCTION The European Union residue monitoring program, 2005-2007, establishes the need to cover 55 active ingredients in various foods, Figure 1. The Waters ACQUITY TQD with the TQ Detector. including baby foods1, 2. Twenty of these pesticides are suitable for multi-residue LC/MS analysis; only one has a negative polarity in electrospray mode, normally requiring two injections (one in each EXPERIMENTAL polarity ion mode). Consequently compounds with negative polarity 3 are often excluded from monitoring programs. Ideally, these should The sample extraction method has been previously reported . be determined in a single analysis with polarity switching. Extracts of blank baby food matrix in acetonitrile were provided along with mixtures of the compounds in acetonitrile by the Central Chemists analyzing pesticide residues are under increasing pres- Science Laboratory (CSL), York, UK. Extracts for injection were sure to broaden the range of pesticides determined in a single prepared by spiking the compounds into the baby food matrix. The analysis: to improve limits of detection, precision and quantitation; supernatant was analyzed on the ACQUITY TQD following dilution to increase confidence in the validity of residue data; to provide with water (1:9) v/v. faster methods and to reduce usage of hazardous solvents while maintaining or reducing costs. -

Fate of Pyriproxyfen in Soils and Plants
toxics Review Fate of Pyriproxyfen in Soils and Plants James Devillers CTIS, 3 Chemin de la Gravière, 69140 Rillieux-La-Pape, France; [email protected] Received: 17 February 2020; Accepted: 10 March 2020; Published: 13 March 2020 Abstract: Since the 1990s, the insect growth regulator pyriproxyfen has been widely used worldwide as a larvicide in vector control and in agriculture to fight a very large number of pests. Due to its widespread use it is of first importance to know how pyriproxyfen behaves in the terrestrial ecosystems. This was the goal of this work to establish the fate profile of pyriproxyfen in soils and plants. Thus, in soil, pyriproxyfen photodegrades slowly but its aerobic degradation is fast. The insecticide presents a high tendency to adsorb onto soils and it is not subject to leaching into groundwater. On the contrary its two main metabolites (40-OH-Pyr and PYPAC) show a different fate in soil. When sprayed to plants, pyriproxyfen behaves as a translaminar insecticide. Its half-life in plants ranges from less than one week to about three weeks. The review ends by showing how the fate profile of pyriproxyfen in soils and plants influences the adverse effects of the molecule on non-target organisms. Keywords: pyriproxyfen; soil; plant; metabolites; insect growth regulator; endocrine disruptor; terrestrial ecosystems 1. Introduction The behavior of pesticides in soils is under the dependence of physical, chemical, and biological processes including volatilization, sorption–desorption, aerobic and anaerobic degradation, uptake and release by plants, run-off, and leaching. These various complex processes control the transport of pesticides within the soil and their transfer from the soil to the other environmental compartments [1–3]. -

Environmental and Disposal Liabilities Identification, Documentation and Reporting Handbook V 3.0
Environmental and Disposal Liabilities Identification, Documentation And Reporting Handbook v 3.0 DECEMBER 2011 Environmental and Disposal Liabilities Identification, Documentation and Reporting Handbook v3.0 TABLE OF CONTENTS Page 1.0 INTRODUCTION .............................................................................................................. 1 1.1 Applicable Standards and Guidance ............................................................................... 1 1.2 Definitions ...................................................................................................................... 3 1.3 Responsibilities ............................................................................................................... 6 2.0 EDL IDENTIFICATION .................................................................................................... 7 2.1 Environmental Location of Concern............................................................................... 8 2.2 Deconstruction and Renovation Activities ..................................................................... 9 2.3 Environmental and Disposal Liabilities ........................................................................ 10 3.0 LIABILITY STATUS ...................................................................................................... 12 3.1 Probable ........................................................................................................................ 12 3.2 Reasonably Possible .................................................................................................... -

The Nervous System – Target Organ Into the Twenty-First Century
The Nervous System – Target Organ into the Twenty-First Century R. Douglas Hamm MD, CCFP, FRCPC (Occ Med), FCBOM President, Canadian Board of Occupational Medicine CONTENTS 1. WHY NEURONS MAKE GOOD TARGETS FOR OCCUPATIONAL TOXICANTS 2. FROM CLASSICAL PLUMBISM TO BEHAVIORAL TOXICOLOGY 3. TOXICOKINETICS, COMPARTMENTS, AND PBPK MODELS 4. THE DIVERSE TOXICODYNAMICS OF THE NERVOUS SYSTEM 1. Peripheral Neurons (neuronopathy, axonopathy, myelinopathy) 2. Synaptic Neurotransmission (cholinergic pathways) 3. Special Senses (visual, auditory, olfactory) 4. Movement Disorders (parkinsonism, ataxia, tremor) 5. Neuroaffective and Neurocognitive Effects 5. NOTEWORTHY OCCUPATIONAL NEUROTOXICANTS 1. “Heavy metals” (Pb, Hg, Tl) and other elements (Mn, As, Al, Sb, Te) 2. Organic Solvents (toluene, xylene, styrene, C2HCl3, C2Cl4, CH3CCl3, CS2) 3. Gases (HCN, CO, H2S, ethylene oxide) 4. Pesticides (organophosphates, carbamates, organochlorines, pyrethroids, neonicotinoids) 6. CLINICAL NEUROTOXICOLOGY 1. Identification of Occupational Neurotoxic Disorders 2. Biomarkers of Exposure and Effect 3. Clinical Investigations of Neurotoxicity 1. WHY NEURONS MAKE GOOD 2. FROM CLASSICAL PLUMBISM TO TARGETS FOR OCCUPATIONAL BEHAVIORAL TOXICOLOGY TOXICANTS Hippocrates (c. 460-370 BC) has been cited as Neuroanatomical structures have large surface the first ancient author to describe a case of areas and receptor populations, e.g., the occupational neurotoxicity but this has been surface area of the brain’s 100 billion neurons shown to be erroneous (Osler, 1907; Waldron, totals hundreds of square metres. 1973, 1978; Skrabanek, 1986; Vance, 2007). The earliest report appears to be that of Neurons have high rates of metabolism, e.g., Nicander of Colophon (2nd cent. BC) who the brain at 2% body mass consumes 20% of observed that in “psimuthion” i.e.