Modeling of Growth and Yield of Some Wheat Cultivars Under Water Shortage and Expected Climate Change
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

BEGR Ü NDUNG Bebauungsplan Mit Integriertem
Begründung zum Bebauungsplan und Flächennutzungsplan mit integriertem Grünordnungsplan B 41 „Lärchenweg” für ein allgemeines Wohngebiet im OT Reundorf der Stadt Lichtenfels, Lkr. Lichtenfels B E G R Ü N D U N G Bebauungsplan mit integriertem Grünordnungsplan mit Änderung des Flächennutzungsplanes B 41 “Lärchenweg” im OT Reundorf Begründung zum Bebauungsplan und Flächennutzungsplan mit integriertem Grünordnungsplan B 41 „Lärchenweg” für ein allgemeines Wohngebiet im OT Reundorf der Stadt Lichtenfels, Lkr. Lichtenfels Vorhabensträger: Stadt Lichtenfels Marktplatz 1+ 5, 96215 Lichtenfels Ansprechpartner: Stadtbauamt Lichtenfels Datum: 26.10.2019 Entwurfsverfasser: Stadt Lichtenfels Marktplatz 1 + 5, 96215 Lichtenfels Ansprechpartner: Stadtbauamt Lichtenfels Begründung zum Bebauungsplan und Flächennutzungsplan mit integriertem Grünordnungsplan B 41 „Lärchenweg” für ein allgemeines Wohngebiet im OT Reundorf der Stadt Lichtenfels, Lkr. Lichtenfels I N H A L T S V E R Z E I C H N I S RECHTSGRUNDLAGEN 1. ANLASS FÜR DIE AUFSTELLUNG DES BEBAUUNGSPLANES 1.1 Flächennutzungsplan 2. PLANUNGSRECHTLICHE GRUNDLAGEN 2.1 Vorhabensträger 2.2 Stadt Lichtenfels 2.3 Planungs- und Verfahrensstand 3. ZIEL DER PLANUNG 4. ABGRENZUNG UND BESCHREIBUNG DES PLANUNGSGEBIETES 4.1 Beschreibung des Gebietes 4.2 Räumlicher Geltungsbereich 4.3 Baugrund 4.4 Denkmalschutz 4.5 Schutzgebiete und schützenswerte Landschaftsteile 4.6 Entwicklung aus dem Flächennutzungsplan 4.7 Immissionsschutz 5. BESCHAFFENHEIT DES PLANUNGSGEBIETES 5.1 Topographie 5.2 Bodenbeschaffenheit 6. BODENORDNENDE -

Neumarkt <> Regensburg <> Plattling
Fahrplanänderung mit Schienenersatzverkehr Freitag, 16.03.18 (23 Uhr) bis Montag, Neumarkt <> Regensburg <> Plattling 19.03.18 (5 Uhr) ZvF 70389 Neumarkt - Regensburg - Obertraubling - Plattling ZvFnur 17.,18.3. 70389 Neumarkt - Regensburg - Obertraubling - Plattling nur 17.,18.3. ag ag ag Bus ag ag RE ag Bus ag as ag RE ag Bus ag ag RE ag Bus ag ag RE ag Bus ag ag RE ag Bus ag ag RE ag Bus ag ag RE ag Bus ag RE ag ag RE Bus ag84321 ag84323 ag93423 Bus323 ag84185 ag84187 RE4855 ag93187 Bus187 ag84189 as86781 ag84191 RE4857 ag93191 Bus191 ag84193 ag84195 RE4859 ag93195 Bus195 ag84331 ag84199 RE4861 ag93199 Bus199 ag84201 ag84203 RE4863 ag93203 Bus203 ag84205 ag84207 RE4865 ag93207 Bus207 ag84209 ag84211 RE4867 ag93211 Bus211 ag84213 RE4869 ag84215 ag84217 RE59499 Bus59499 84321 84323 9342317. + 32317. + 84185 84187 4855 9318717. + 18717. + 84189 86781 84191 4857 9319117. + 19117. + 84193 84195 4859 9319517. + 19517. + 84331 84199 4861 9319917. + 19917. + 84201 84203 4863 9320317. + 20317. + 84205 84207 4865 9320717. + 20717. + 84209 84211 4867 9321117. + 21117. + 84213 4869 84215 84217 5949916.- 5949916.- sa sa+so täglich täglich täglich sa+so sa+so täglich täglich täglich täglich täglich sa+so täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.17. + 18.03.16.- 18.03.16.- sa sa+so täglich täglich täglich sa+so sa+so täglich täglich täglich täglich täglich sa+so täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich täglich Ersatzhaltestelle von 18.03. -

Naturpark Altmühltal (Südliche Frankenalb)“ Vom 14
Verordnung über den „Naturpark Altmühltal (Südliche Frankenalb)“ Vom 14. September 1995 (GVBl. S. 692) BayRS 791-5-15-U (§§ 1–14) Verordnung über den „Naturpark Altmühltal (Südliche Frankenalb)“ Vom 14. September 1995 (GVBl. S. 692) BayRS 791-5-15-U Vollzitat nach RedR: Verordnung über den „Naturpark Altmühltal (Südliche Frankenalb)“ vom 14. September 1995 (GVBl. S. 692, BayRS 791-5-15-U) Auf Grund von Art. 11, 45 Abs. 1 Nr. 2, Art. 55 Abs. 1 Satz 2 in Verbindung mit Art. 45 Abs. 2 Satz 3 Halbsatz 2 und Art. 37 Abs. 2 Nr. 1 des Bayerischen Naturschutzgesetzes – BayNatSchG – (BayRS 791-1- U), zuletzt geändert durch Gesetz vom 28. April 1994 (GVBl S. 299), erläßt das Bayerische Staatsministerium für Landesentwicklung und Umweltfragen folgende Verordnung: § 1 Schutzgegenstand (1) 1Teilgebiete der Naturräume „Südliche Frankenalb“ und „Vorland der Südlichen Frankenalb“ in der kreisfreien Stadt Ingolstadt und in den Landkreisen Eichstätt, Neuburg-Schrobenhausen, Kelheim, Regensburg, Neumarkt i. d. OPf., Roth, Weißenburg-Gunzenhausen und Donau-Ries werden in den in § 2 näher bezeichneten Grenzen als Naturpark festgesetzt. 2Das Gebiet hat eine Größe von ca. 296 240 Hektar. (2) Der Naturpark erhält die Bezeichnung „Naturpark Altmühltal (Südliche Frankenalb)“. (3) Träger des Naturparks ist der „Verein Naturpark Altmühltal (Südliche Frankenalb) e. V.“ mit Sitz in Weißenburg i. Bay. § 2 Naturparkgrenzen (1) Die Grenzen des Naturparks sind in einer Karte M = 1:100 000, die als Anlage 1 Bestandteil dieser Verordnung ist, grob dargestellt. (2) 1Die genauen Grenzen des Naturparks sind in einer Karte M = 1:25 000 eingetragen, die beim Staatsministerium für Landesentwicklung und Umweltfragen als oberster Naturschutzbehörde niedergelegt ist und auf die Bezug genommen wird. -

Flyer Öko-Modellregion Landkreis Neumarkt I.D.Opf
DIE BAYERISCHEN ÖKO-MODELLREGIONEN UNSERE ANSPRECHPARTNERIN Die Öko-Modellregionen sind als Baustein des Landespro- Sandra Foistner gramms BioRegio Bayern 2020 des Bayerischen Staatsminis- Projektmanagerin Öko-Modellregion teriums für Ernährung, Landwirtschaft und Forsten gestartet +49 (0)9181 50 929 14 und werden in BioRegio 2030 fortgeführt. Ziel des Landespro- [email protected] gramms ist ein Anteil von 30 % ökologisch bewirtschafteter Fläche in Bayern bis zum Jahr 2030. In den Öko-Modellregionen wird eine große Bandbreite an Im Auftrag des Landkreises Projekten umgesetzt, angefangen von der Erzeugung und Ver- arbeitung über die Vermarktung und Gemeinschaftsverpfle- REGINA GmbH gung bis hin zur Bildung. Im Fokus steht aber nicht nur die Dr.-Grundler-Str. 1 Steigerung der ökologischen Anbaufläche, sondern auch die 92318 Neumarkt i.d.OPf. Verbindung von Regionalität und ökologischer Erzeugung mit +49 (0)918 150 929 0 naturverträglichen, nachhaltigen und regionalen Projekten. [email protected] www.reginagmbh.de Es geht vor allem darum, die in den Regionen vorhandenen Po- tenziale zu erschließen und gemeinsam mit engagierten Akteu- ren vorhandene Strukturen zu beleben oder neue aufzubauen. In jeder Region gibt es aktive, unternehmerische Menschen, die 11 25 etwas bewegen wollen, die ihre Region und den ökologischen 27 10 22 Landbau voranbringen möchten. Die Öko-Modellregionen bie- 3 12 26 ten jedem Engagierten Unterstützung und Begleitung, um die 23 nächsten Schritte zu gehen. Nur in der Zusammenarbeit wird es 4 6 gelingen, tragfähige, über die Förderung hinausgehende Struk- 2 turen aufzubauen. 24 7 21 17 14 18 13 1 19 5 16 20 15 8 9 N E N Ö O K I O G E M R O D E L L Fotos: Christian Amthor, BIregO, Daniel Delang, Anne Fröhlich, Marion Lang 1 Mühldorfer Land 15 Ostallgäu 2 Neumarkt i.d. -

Kommunale Partnerschaften Der Europäischen Metropolregion Nürnberg
Stadt Nürnberg Amt für Internationale Beziehungen Partnerkommunen von Städten, Gemeinden und Landkreisen in der Europäischen Metropolregion Nürnberg Stadt / Gemeinde Landkreis Partnerkommune Land Landkreis Adelsdorf Erlangen-Höchstadt, Uggiate Trevano Italien MFr Adelsdorf Erlangen-Höchstadt, Feldbach Österreich MFr Ahorn Coburg, OFr Irdning Österreich Ahorn Coburg, OFr Eisfeld Thüringen Allersberg Roth, MFr Saint-Céré Frankreich Altdorf b. Nürnberg Nürnberger Land, MFr Sehma Sachsen Altdorf b. Nürnberg Nürnberger Land, MFr Dunaharaszti Ungarn Altdorf b. Nürnberg Nürnberger Land, MFr Pfitsch Italien Altdorf b. Nürnberg Nürnberger Land, MFr Colbitz Sachsen-Anhalt Amberg kreisfrei, OPf Perigueux Frankreich Amberg kreisfrei, OPf Trikala Griechenland Amberg kreisfrei, OPf Desenzano del Garda Italien Amberg kreisfrei, OPf Bystrzyca Klodzka Polen Amberg kreisfrei, OPf Kranj Slowenien Amberg kreisfrei, OPf Usti nad Orilici Tschechien Amberg-Sulzbach Landkreis, OPf Canton Maintenon Frankreich Amberg-Sulzbach Landkreis, OPf Grafschaft Argyll Großbritannien Amberg-Sulzbach Landkreis, OPf Lkr. Sächsische Sachsen Schweiz Ammerndorf Fürth, MFr Dulliken Schweiz Ammerthal Amberg-Sulzbach, OPf Modiim Israel Ansbach kreisfrei, MFr Jingjiang China Ansbach Landkreis, MFr Jingjiang China Ansbach kreisfrei, MFr Anglet Frankreich Ansbach kreisfrei, MFr Fermo Italien Ansbach Landkreis, MFr Erzgebirgskreis Sachsen Ansbach Landkreis, MFr Mudanya Türkei Ansbach kreisfrei, MFr Bay City USA Arzberg Wunsiedel, Ofr Arzberg Österreich Arzberg Wunsiedel, Ofr Horní Slavkov -
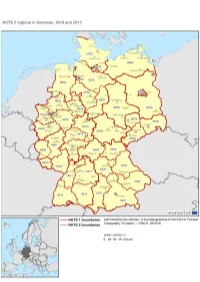
Nuts-Map-DE.Pdf
GERMANY NUTS 2013 Code NUTS 1 NUTS 2 NUTS 3 DE1 BADEN-WÜRTTEMBERG DE11 Stuttgart DE111 Stuttgart, Stadtkreis DE112 Böblingen DE113 Esslingen DE114 Göppingen DE115 Ludwigsburg DE116 Rems-Murr-Kreis DE117 Heilbronn, Stadtkreis DE118 Heilbronn, Landkreis DE119 Hohenlohekreis DE11A Schwäbisch Hall DE11B Main-Tauber-Kreis DE11C Heidenheim DE11D Ostalbkreis DE12 Karlsruhe DE121 Baden-Baden, Stadtkreis DE122 Karlsruhe, Stadtkreis DE123 Karlsruhe, Landkreis DE124 Rastatt DE125 Heidelberg, Stadtkreis DE126 Mannheim, Stadtkreis DE127 Neckar-Odenwald-Kreis DE128 Rhein-Neckar-Kreis DE129 Pforzheim, Stadtkreis DE12A Calw DE12B Enzkreis DE12C Freudenstadt DE13 Freiburg DE131 Freiburg im Breisgau, Stadtkreis DE132 Breisgau-Hochschwarzwald DE133 Emmendingen DE134 Ortenaukreis DE135 Rottweil DE136 Schwarzwald-Baar-Kreis DE137 Tuttlingen DE138 Konstanz DE139 Lörrach DE13A Waldshut DE14 Tübingen DE141 Reutlingen DE142 Tübingen, Landkreis DE143 Zollernalbkreis DE144 Ulm, Stadtkreis DE145 Alb-Donau-Kreis DE146 Biberach DE147 Bodenseekreis DE148 Ravensburg DE149 Sigmaringen DE2 BAYERN DE21 Oberbayern DE211 Ingolstadt, Kreisfreie Stadt DE212 München, Kreisfreie Stadt DE213 Rosenheim, Kreisfreie Stadt DE214 Altötting DE215 Berchtesgadener Land DE216 Bad Tölz-Wolfratshausen DE217 Dachau DE218 Ebersberg DE219 Eichstätt DE21A Erding DE21B Freising DE21C Fürstenfeldbruck DE21D Garmisch-Partenkirchen DE21E Landsberg am Lech DE21F Miesbach DE21G Mühldorf a. Inn DE21H München, Landkreis DE21I Neuburg-Schrobenhausen DE21J Pfaffenhofen a. d. Ilm DE21K Rosenheim, Landkreis DE21L Starnberg DE21M Traunstein DE21N Weilheim-Schongau DE22 Niederbayern DE221 Landshut, Kreisfreie Stadt DE222 Passau, Kreisfreie Stadt DE223 Straubing, Kreisfreie Stadt DE224 Deggendorf DE225 Freyung-Grafenau DE226 Kelheim DE227 Landshut, Landkreis DE228 Passau, Landkreis DE229 Regen DE22A Rottal-Inn DE22B Straubing-Bogen DE22C Dingolfing-Landau DE23 Oberpfalz DE231 Amberg, Kreisfreie Stadt DE232 Regensburg, Kreisfreie Stadt DE233 Weiden i. -
![Arxiv:2007.11896V2 [Stat.AP] 3 Aug 2020](https://docslib.b-cdn.net/cover/9807/arxiv-2007-11896v2-stat-ap-3-aug-2020-1429807.webp)
Arxiv:2007.11896V2 [Stat.AP] 3 Aug 2020
Causal analysis of Covid-19 spread in Germany Atalanti A. Mastakouri Department of Empirical Inference Max Planck Institute for Intelligent Systems Tübingen, Germany [email protected] Bernhard Schölkopf Department of Empirical Inference Max Planck Institute for Intelligent Systems Tübingen, Germany [email protected] Abstract In this work, we study the causal relations among German regions in terms of the spread of Covid-19 since the beginning of the pandemic, taking into account the restriction policies that were applied by the different federal states. We propose and prove a new theorem for a causal feature selection method for time series data, robust to latent confounders, which we subsequently apply on Covid-19 case numbers. We present findings about the spread of the virus in Germany and the causal impact of restriction measures, discussing the role of various policies in containing the spread. Since our results are based on rather limited target time series (only the numbers of reported cases), care should be exercised in interpreting them. However, it is encouraging that already such limited data seems to contain causal signals. This suggests that as more data becomes available, our causal approach may contribute towards meaningful causal analysis of political interventions on the development of Covid-19, and thus also towards the development of rational and data-driven methodologies for choosing interventions. 1 Introduction arXiv:2007.11896v2 [stat.AP] 3 Aug 2020 The ongoing outbreak of the Covid-19 pandemic has rendered the tracking of the virus spread a problem of major importance, in order to better understand the role of the demographics and of political measures taken to contain the virus. -

Eastern Bavaria
Basic text Eastern Bavaria Culture Eastern Bavaria is still home to more castles than anywhere else in Germany: Some medieval castles remain only as ruins, whilst other castles such as Falkenstein Castle have withstood decline and are open to visitors. The expansive spruce forests in Eastern Bavaria have given way to the Bavarian Glass Road, as they supplied the wood and quartz sand –the key raw materials – for the very first glass foundries. Spanning some 250 kilometres, it is one of the most picturesque holiday routes in Germany. Those choosing to travel along the route will learn all about the 700-year tradition of glass production and glass as a form of art. The route, which begins in Neustadt an der Waldnaab and leads to Passau, features glass foundries, galleries and museums, all packed to the brim with interesting facts about the traditional handicraft. Some Eastern Bavarian companies are keeping the tradition alive to this day and export to countries ranging from the United Arab Emirates to the United States of America. The largest towns in Eastern Bavaria include Regensburg, Landshut and Passau. The city of Regensburg, which was first founded by Roman Emperor Marcus Aurelius, has retained its medieval centre to this day. The Old Town of Regensburg together with Stadtamhof has been a UNESCO World Heritage Site since 2006. Landshut is the prototype of an old Bavarian town. Above all its town centre, which features gabled houses, decorative façades, oriels and arches, is one of the most beautiful squares to be found in the whole of Germany. The three-river town of Passau, which was built in the Italian baroque style, achieved early wealth thanks to its participation in the salt trade and was a place of border crossings due to its location on the border with Austria and just 30 kilometres from the Czech border. -

Lichtenfels/Ebern – Bamberg – Ebermannstadt Lichtenfels/Ebern
Lichtenfels/Ebern – Bamberg – Ebermannstadt gültig ab 09.12.2018 S RE RE S S ag RE ag RE S ag RE 4979 RE ag RB S ag ag RE RE RE S ag RE 59280 ag RE 4903/4923 RB Zugnummer 1 4921 4921 1 1 84431 4939 84437 4949 1 84433 /59393 4951 84439 4901 1 84441 84443 4608 4981 4941 1 84445 /59300 84447 /4943/4945 59331 Verkehrstag mo-fr mo-sa so mo-fr täglich mo-fr täglich mo-fr mo-fr täglich mo-fr täglich täglich täglich mo-fr täglich sa+so mo-fr mo-fr täglich mo-fr täglich täglich täglich täglich täglich täglich Probstzella/ Ludwigs- Hof/ Saalfeld/ von Coburg Sonneberg Coburg Sonneberg Kronach stadt Jena Bayreuth Sonneberg Lichtenfels ab 4:09 5:09 5:30 6:02 6:37 6:51 7:09 7:35 7:57 8:19 8:30 Bad Staffelstein ab 4:14 5:14 5:35 6:07 6:41 6:56 7:14 7:39 8:02 8:23 8:35 Ebensfeld ab 4:18 5:18 5:39 6:11 6:45 7:00 7:18 7:43 | | 8:39 Zapfendorf ab 4:22 5:22 5:43 6:15 6:49 7:04 7:22 7:47 | | 8:43 Ebing ab 4:25 5:25 5:46 6:17 | 7:07 7:24 7:50 | | 8:45 Ebern ab | | 5:18 | | 6:30 | | | | | 8:01 | | Rentweinsdorf ab | | x5:23 | | x6:35 | | | | | x8:06 | | Manndorf ab | | x5:28 | | x6:39 | | | | | x8:10 | | Reckendorf ab | | 5:32 | | 6:43 | | | | | 8:14 | | Baunach ab | | 5:37 | | 6:49 | | | | | 8:19 | | Breitengüßbach ab 4:29 5:28 5:43 5:49 6:21 6:55 | 7:11 7:28 7:54 | 8:25 8:32 8:48 Hallstadt (bei Bamberg) ab 4:32 5:32 5:47 5:54 6:25 6:59 | 7:15 7:32 7:58 | 8:28 | 8:52 Bamberg an 4:35 5:35 5:50 5:57 6:28 6:35 7:02 6:59 7:18 7:36 8:01 8:19 8:32 8:37 8:56 Bamberg ab 4:10 4:38 4:38 4:45 5:08 5:38 6:04 6:08 6:26 6:38 7:06 7:09 7:21 7:39 8:04 8:08 8:38 Strullendorf -

Regionalisierte Bevölkerungsvorausberechnung Für Bayern Bis 2039 X Demographisches Profil Für Den Xlandkreis Erlangen-Höchstadt
Beiträge zur Statistik Bayerns, Heft 553 Regionalisierte Bevölkerungsvorausberechnung für Bayern bis 2039 x Demographisches Profil für den xLandkreis Erlangen-Höchstadt Hrsg. im Dezember 2020 Bestellnr. A182AB 202000 www.statistik.bayern.de/demographie Zeichenerklärung Auf- und Abrunden 0 mehr als nichts, aber weniger als die Hälfte der kleins- Im Allgemeinen ist ohne Rücksicht auf die Endsummen ten in der Tabelle nachgewiesenen Einheit auf- bzw. abgerundet worden. Deshalb können sich bei der Sum mierung von Einzelangaben geringfügige Ab- – nichts vorhanden oder keine Veränderung weichun gen zu den ausgewiesenen Endsummen ergeben. / keine Angaben, da Zahlen nicht sicher genug Bei der Aufglie derung der Gesamtheit in Prozent kann die Summe der Einzel werte wegen Rundens vom Wert 100 % · Zahlenwert unbekannt, geheimzuhalten oder nicht abweichen. Eine Abstimmung auf 100 % erfolgt im Allge- rechenbar meinen nicht. ... Angabe fällt später an X Tabellenfach gesperrt, da Aussage nicht sinnvoll ( ) Nachweis unter dem Vorbehalt, dass der Zahlenwert erhebliche Fehler aufweisen kann p vorläufiges Ergebnis r berichtigtes Ergebnis s geschätztes Ergebnis D Durchschnitt ‡ entspricht Publikationsservice Das Bayerische Landesamt für Statistik veröffentlicht jährlich über 400 Publikationen. Das aktuelle Veröffentlichungsverzeich- nis ist im Internet als Datei verfügbar, kann aber auch als Druckversion kostenlos zugesandt werden. Kostenlos Publikationsservice ist der Download der meisten Veröffentlichungen, z.B. von Alle Veröffentlichungen sind im Internet -

23.-27.01.09 Nürnberg
Kursbuchstrecken 805 / 891.1 820 / 891.2 Nürnberg – Fürth – Neustadt (A) / Nürnberg – Fürth – Forchheim Gültig von Freitag, 23.01. – Montag, 26.01.2009 Schienenersatzverkehr zwischen Nürnberg Hbf und Fürth Hbf Zugausfälle zwischen Nürnberg Hbf und Fürth Hbf Ersatz durch U-Bahn Umleitung von Fernverkehrszügen über Würzburg – Ansbach – Nürnberg 01_70207 Sehr geehrte Fahrgäste, wegen Weichenarbeiten fallen Regionalzüge (RB + RE) zwischen Nürnberg Hbf und Fürth Hbf aus, Ersatz durch U-Bahn (U1). Auch werden einzelne Züge zwischen Nürnberg Hbf – Fürth Hbf durch Busse ersetzt (siehe Fahrplantabellen).Aufgrund der Baumaßnahmen kommt es leider auch zu Ver- spätungen. Bitte berücksichtigen Sie die längeren Fahrzeiten bereits bei Ihrer Reiseplanung und wäh- len Sie ggf. eine frühere Verbindung. Hier nicht dargestellte Züge verkehren nach regulärem, bzw. verspätetem Fahrplan. Wir bitten um Verständnis für die notwendigen Arbeiten und die dadurch auftretenden Behinderungen und Unannehmlichkeiten. Ihre DB Regio Schienenersatzverkehr Reisegepäck, Faltrollstühle und Kinderwagen können im Bus mitgenommen werden. Fahrradmitnahme ist im Bus nicht möglich. Hinweise zur Beförderung finden Sie ggf. im Fahrplan. Zustieg nur mit gültigem Fahrschein, da im Bus kein Verkauf stattfindet. Fahrscheine erwerben Sie bitte im Voraus an den betreffenden Stationen durch Kauf am Schalter bzw. Automaten oder online unter www.bahn.de. Im Bus gelten die gleichen Tarife wie im Zug. Bitte beachten Sie die evtl. vom Bahnhof abweichenden Bus-Haltestellen (siehe Fahrplan). Erläuterung zum SEV-Symbol Bei Schienenersatzverkehr soll Ihnen dieses (lilafarbene) Symbol auf Bussen, Haltestellen und Aushängen durch seine Wiedererkennung bei der Orientierung behilflich sein. Umleitung von Fernverkehrszügen und Verspätungen im Regional-und Fernverkehr Bitte beachten Sie, dass aufgrund der Bautätigkeiten folgende Fernverkehrszüge über Würzburg– Ansbach– Nürnberg umgeleitet werden: IC 1687, IC 1886, IC 1887, ICE 920, IC 2029. -

OECD Territorial Grids
BETTER POLICIES FOR BETTER LIVES DES POLITIQUES MEILLEURES POUR UNE VIE MEILLEURE OECD Territorial grids August 2021 OECD Centre for Entrepreneurship, SMEs, Regions and Cities Contact: [email protected] 1 TABLE OF CONTENTS Introduction .................................................................................................................................................. 3 Territorial level classification ...................................................................................................................... 3 Map sources ................................................................................................................................................. 3 Map symbols ................................................................................................................................................ 4 Disclaimers .................................................................................................................................................. 4 Australia / Australie ..................................................................................................................................... 6 Austria / Autriche ......................................................................................................................................... 7 Belgium / Belgique ...................................................................................................................................... 9 Canada ......................................................................................................................................................