Vancomycin Plus Piperacillin/Tazobactam and Acute Kidney Injury in Adults: a Systematic Review and Meta-Analysis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cefoxitin Versus Piperacillin– Tazobactam As Surgical Antibiotic Prophylaxis in Patients Undergoing Pancreatoduodenectomy: Protocol for a Randomised Controlled Trial
Open access Protocol BMJ Open: first published as 10.1136/bmjopen-2020-048398 on 4 March 2021. Downloaded from Cefoxitin versus piperacillin– tazobactam as surgical antibiotic prophylaxis in patients undergoing pancreatoduodenectomy: protocol for a randomised controlled trial Nicole M Nevarez ,1 Brian C Brajcich,2 Jason Liu,2,3 Ryan Ellis,2 Clifford Y Ko,2 Henry A Pitt,4 Michael I D'Angelica,5 Adam C Yopp1 To cite: Nevarez NM, ABSTRACT Strengths and limitations of this study Brajcich BC, Liu J, et al. Introduction Although antibiotic prophylaxis is Cefoxitin versus piperacillin– established in reducing postoperative surgical site tazobactam as surgical ► A major strength of this study is the multi- infections (SSIs), the optimal antibiotic for prophylaxis in antibiotic prophylaxis institutional, double- arm, randomised controlled in patients undergoing pancreatoduodenectomy (PD) remains unclear. The study trial design. objective is to evaluate if administration of piperacillin– pancreatoduodenectomy: ► A limitation of this study is that all perioperative care protocol for a randomised tazobactam as antibiotic prophylaxis results in decreased is at the discretion of the operating surgeon and is controlled trial. BMJ Open 30- day SSI rate compared with cefoxitin in patients not standardised. 2021;11:e048398. doi:10.1136/ undergoing elective PD. ► All data will be collected through the American bmjopen-2020-048398 Methods and analysis This study will be a multi- College of Surgeons National Surgical Quality ► Prepublication history for institution, double- arm, non- blinded randomised controlled Improvement Program, which is a strength for its this paper is available online. superiority trial. Adults ≥18 years consented to undergo PD ease of use but a limitation due to the variety of data To view these files, please visit for all indications who present to institutions participating included. -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -
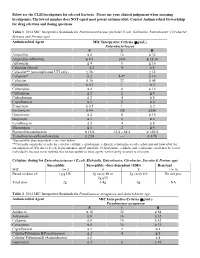
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp. -

Fosfomycin Resistance in Escherichia Coli Isolates from South Korea and in Vitro Activity of Fosfomycin Alone and in Combination with Other Antibiotics
antibiotics Article Fosfomycin Resistance in Escherichia coli Isolates from South Korea and in vitro Activity of Fosfomycin Alone and in Combination with Other Antibiotics Hyeri Seok 1 , Ji Young Choi 2, Yu Mi Wi 3, Dae Won Park 1, Kyong Ran Peck 4 and Kwan Soo Ko 2,* 1 Division of Infectious Diseases, Department of Medicine, Korea University Ansan Hospital, Korea University College of Medicine, Ansan 15355, Korea; [email protected] (H.S.); [email protected] (D.W.P.) 2 Department of Microbiology, Sungkyunkwan University School of Medicine, Suwon 16419, Korea; [email protected] 3 Division of Infectious Diseases, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, Changwon 51353, Korea; [email protected] 4 Division of Infectious Diseases, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul 06351, Korea; [email protected] * Correspondence: [email protected]; Tel.: +82-31-299-6223 Received: 17 February 2020; Accepted: 3 March 2020; Published: 6 March 2020 Abstract: We investigated fosfomycin susceptibility in Escherichia coli clinical isolates from South Korea, including community-onset, hospital-onset, and long-term care facility (LTCF)-onset isolates. The resistance mechanisms and genotypes of fosfomycin-resistant isolates were also identified. Finally, the in vitro efficacy of combinations of fosfomycin with other antibiotics were examined in susceptible or extended spectrum β-lactamase (ESBL)-producing E. coli isolates. The fosfomycin resistance rate was 6.7% and was significantly higher in LTCF-onset isolates than community-onset and hospital-onset isolates. Twenty-one sequence types (STs) were identified among 19 fosfomycin-resistant E. coli isolates, showing diverse genotypes. fosA3 was found in only two isolates, and diverse genetic variations were identified in three genes associated with fosfomycin resistance, namely, GlpT, UhpT, and MurA. -

Piperacillin/Tazobactam Drug Class1 Antibiotic – Penicillin with Β-Lactamase Inhibitor
Monographs for Commonly Administered Intravenous Medications in Home and Community Care Piperacillin/Tazobactam Drug Class1 Antibiotic – penicillin with β-lactamase inhibitor Spectrum1 Refer to product monograph for complete spectrum For β-lactamase producing bacteria strains (e.g., Haemophilus influenza, Escherichia coli, and Staphylococcus aureus). Also to susceptible Acinetobacter species, Klebsiella pneumonia, Pseudomonas aeruginosa often combined with aminoglycoside). Cross Sensitivities / Allergies1 Cross sensitivities with penicillin and possibly cephalosporin and/or beta-lactam inhibitors Indications1,2 Intra-abdominal Respiratory tract Skin and skin structure Septicemia Gynecological Urinary tract Other conditions based on culture and sensitivity results Outpatient Considerations1 For patients with a documented allergy to penicillin, cephalosporin or beta-lactam inhibitor, the first dose should be administered in a hospital or clinic setting. Must be able to access laboratory monitoring (either at outpatient laboratory or by arranging in-home lab) if using an interacting oral medication (see Potential Drug Interactions section) Prescribing Considerations At time of ordering please provide the following to the pharmacist: and Dosage in Adults1,2 Height, weight Most recent serum creatinine with date obtained Indication (infection being treated) Usually dosed every 6 to 8 hours. Available as 2 g/0.25 g, 3 g/0.375 g, 4 g/0.5 g piperacillin/tazobactam (dose selected based on indication) Maximum 18 g/ 2.25 g piperacillin/tazobactam daily Dose and administration interval require adjustment for renal impairment Errors have occurred due to unusual ordering as combined dose of piperacillin and tazobactam. Order may be expressed only as piperacillin component or as a total of piperacillin + tazobactam (8:1 ratio). -

Eml-2017-Antibacterials-Eng.Pdf
Consideration of antibacterial medicines as part of the revisions to 2017 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) Section 6.2 Antibacterials including Access, Watch and Reserve Lists of antibiotics This summary has been prepared by the Health Technologies and Pharmaceuticals (HTP) programme at the WHO Regional Office for Europe. It is intended to communicate changes to the 2017 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) to national counterparts involved in the evidence-based selection of medicines for inclusion in national essential medicines lists (NEMLs), lists of medicines for inclusion in reimbursement programs, and medicine formularies for use in primary, secondary and tertiary care. This document does not replace the full report of the WHO Expert Committee, 2017 and this summary should be read in conjunction with the full report (WHO Technical Report Series, No. 1006; http://apps.who.int/iris/bitstream/10665/259481/1/9789241210157-eng.pdf?ua=1). The revised lists of essential medicines (in English) are available as follows: 2017 WHO Model List of Essential Medicines for adults (EML) http://www.who.int/medicines/publications/essentialmedicines/20th_EML2017_FINAL_amend edAug2017.pdf?ua=1 2017 Model List of Essential Medicines for children (EMLc) http://www.who.int/medicines/publications/essentialmedicines/6th_EMLc2017_FINAL_amend edAug2017.pdf?ua=1 Summary of changes to Section 6.2 Antibacterials: Section 6 of the EML covers anti-infective medicines. Disease-specific subsections within Section 6, such as those covering medicines for tuberculosis, HIV, hepatitis and malaria, have been regularly reviewed and updated, taking into consideration relevant WHO treatment guidelines. -

Clinical Guidelines for the Antibiotic Treatment for Community-Acquired
Special Article Infection & https://doi.org/10.3947/ic.2017.49.4.301 Infect Chemother 2017;49(4):301-325 Chemotherapy ISSN 2093-2340 (Print) · ISSN 2092-6448 (Online) Clinical Guidelines for the Antibiotic Treatment for Community-Acquired Skin and Soft Tissue Infection Yee Gyung Kwak1,2*, Seong-Ho Choi3,4*, Tark Kim1,5*, Seong Yeon Park3,6*, Soo-Hong Seo7,8, Min Bom Kim9,10, Sang-Ho Choi2,11 1The Korean Society of Infectious Diseaes; 2Department of Internal Medicine, Inje University Ilsan Paik Hospital, Goyang; 3The Korean Society for Chemotherapy; 4Department of Internal Medicine, Chung Ang University College of Medicine, Seoul; 5Department of Internal Medicine, SoonChunHyang University Bucheon Hospital, Bucheon; 6Department of Internal Medicine, Dongguk University College of Medicine, Goyang; 7The Korean Dermatological Association; 8Department of Dermatology, Korea University College of Medicine, Seoul; 9The Korean Orthopaedic Association; 10Department of Orthopedic Surgery, Seoul National University College of Medicine, Seoul; 11Department of Infectious Diseases, Ulsan University College of Medicine, Seoul, Korea Skin and soft tissue infection (SSTI) is common and important infectious disease. This work represents an update to 2012 Korean guideline for SSTI. The present guideline was developed by the adaptation method. This clinical guideline provides recommen- dations for the diagnosis and management of SSTI, including impetigo/ecthyma, purulent skin and soft tissue infection, erysipe- las and cellulitis, necrotizing fasciitis, pyomyositis, clostridial myonecrosis, and human/animal bite. This guideline targets com- munity-acquired skin and soft tissue infection occurring among adult patients aged 16 years and older. Diabetic foot infection, surgery-related infection, and infections in immunocompromised patients were not included in this guideline. -

Ceftaroline in Complicated Skin and Skin-Structure Infections
Infection and Drug Resistance Dovepress open access to scientific and medical research Open Access Full Text Article REVIEW Ceftaroline in complicated skin and skin-structure infections Paul O Hernandez1 Abstract: Ceftaroline is an advanced-generation cephalosporin antibiotic recently approved by Sergio Lema2 the US Food and Drug Administration for the treatment of complicated skin and skin-structure Stephen K Tyring3 infections (cSSSIs). This intravenous broad-spectrum antibiotic exerts potent bactericidal activity Natalia Mendoza2,4 by inhibiting bacterial cell wall synthesis. A high affinity for the penicillin-binding protein 2a (PBP2a) of methicillin-resistant Staphylococcus aureus (MRSA) makes the drug especially 1University of Texas School of Medicine at San Antonio, beneficial to patients with MRSA cSSSIs. Ceftaroline has proved in multiple well-conducted San Antonio, TX, 2Woodhull clinical trials to have an excellent safety and efficacy profile. In adjusted doses it is also recom- Medical and Mental Health Center, mended for patients with renal or hepatic impairment. Furthermore, the clinical effectiveness Brooklyn, NY, 3Department of Dermatology, University of Texas and high cure rate demonstrated by ceftaroline in cSSSIs, including those caused by MRSA Health Science Center at Houston, and other multidrug-resistant strains, warrants its consideration as a first-line treatment option 4 Houston, TX, USA; Department of for cSSSIs. This article reviews ceftaroline and its pharmacology, efficacy, and safety data to Dermatology, El -

Optimizing Intravenous Fosfomycin Dosing in Combination With
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector International Journal of Infectious Diseases 50 (2016) 23–29 Contents lists available at ScienceDirect International Journal of Infectious Diseases jou rnal homepage: www.elsevier.com/locate/ijid Optimizing intravenous fosfomycin dosing in combination with carbapenems for treatment of Pseudomonas aeruginosa infections in critically ill patients based on pharmacokinetic/pharmacodynamic (PK/PD) simulation a b b c a, O. Asuphon , P. Montakantikul , J. Houngsaitong , P. Kiratisin , P. Sonthisombat * a Department of Pharmacy Practice, Faculty of Pharmaceutical Sciences, Naresuan University, Phitsanulok, Thailand b Department of Pharmacy, Faculty of Pharmacy, Mahidol University, Bangkok, Thailand c Department of Microbiology, Faculty of Medicine, Siriraj Hospital, Mahidol University, Bangkok, Thailand A R T I C L E I N F O A B S T R A C T Article history: Objective: The purpose of the study was to determine the optimal dosing regimen of intravenous Received 29 March 2016 fosfomycin for the treatment of Pseudomonas aeruginosa (PA) based on PK/PD targets. Received in revised form 11 June 2016 Method: A total of 120 PA isolates were recovered from various clinical specimens at university hospital Accepted 15 June 2016 in Thailand. Minimum Inhibitory Concentrations (MICs) of all the isolates were determined by the E-test Corresponding Editor: Eskild Petersen, method. PK parameters were obtained from a published study. Monte Carlo simulation was performed to Aarhus, Denmark calculate the percentage of target attainment (PTA) and cumulative fraction of response (CFR). Results: MIC90 of fosfomycin alone, fosfomycin in combination with carbapenem, carbapenems alone Keywords: and carbapenems in combination with fosfomycin were >1,024, 1,024, >32 and 32 mg/ml, for multidrug fosfomycin resistant (MDR)-PA and 512, 128, 8 and 3 mg/ml respectively, for non-MDR PA. -

Journal Club: “Effect of Piperacillin-Tazobactam Vs
Infectious Disease Update 02/25/19 Steven T. Park Division of Infectious Diseases Objectives • Zosyn versus Meropenem for ESBL bacteremia • 7 versus 14 days for gram negative bacteremia • Oral versus IV for osteomyelitis “Effect of Piperacillin-Tazobactam vs Meropenem on 30-Day Mortality for Patients With E coli or Klebsiella pneumoniae Bloodstream Infection and Ceftriaxone Resistance” (MERINO trial) Harris PNA et al. JAMA 2018. Sep 11; 320:984. Introduction • Treatment of choice for ESBL organisms are thought to be carbapenems • Reports of failure of non-carbapenems in the past even when microbiologic data showed susceptibility • Most reported before CLSI lowered the breakpoints for Enterobacteriaceae • Recent reviews looking at retrospective data conclude that you can use non-carbapenems (Zosyn, Cefepime) for urine infections • However no randomized controlled trial testing this hypothesis • First randomized controlled trial testing whether Zosyn is equivalent to meropenem for ESBL bacteremia Design, Setting, Participants • Non-inferiority, parallel group, randomized clinical trial • Hospitalized patients enrolled from 26 sites in 9 countries (mostly from Singapore, Australia, Italy, Saudi Arabia, and Turkey) from February 2014 to July 2017 • Patients over the age of 21 was eligible if he/she had at least 1 positive blood culture with E coli or Klebsiella spp that was non-susceptible to ceftriaxone but susceptible to pip-tazo and meropenem • Of 1646 patients screened, 391 were included in the study. • Meropenem 1g IV q8h (n = 191) or piperacillin/tazobactam 4.5g IV q6h (not extended infusion, n = 188) was given for a minimum of 4 days and up to 14 days. The duration of therapy was determined by the treating clinician. -

Antimicrobials As Single and Combination Therapy for Colistin-Resistant Pseudomonas Aeruginosa at a University Hospital in Thailand
antibiotics Article Antimicrobials as Single and Combination Therapy for Colistin-Resistant Pseudomonas aeruginosa at a University Hospital in Thailand Supanun Pungcharoenkijkul 1,2, Jantima Traipattanakul 3, Sudaluck Thunyaharn 4 and Wichai Santimaleeworagun 5,6,* 1 College of Pharmacotherapy Thailand, Nontaburi 11000, Thailand; [email protected] 2 Department of Pharmacist, Nopparat Rajathanee Hospital, Bangkok 10230, Thailand 3 Division of Infectious Disease, Department of Medicine, Phramongkutklao Hospital, Bangkok 10400, Thailand; [email protected] 4 Faculty of Medical Technology, Nakhonratchasima College, Nakhon Ratchasima 30000, Thailand; [email protected] 5 Department of Pharmacy, Faculty of Pharmacy, Silpakorn University, Nakorn Pathom 73000, Thailand 6 Pharmaceutical Initiative for Resistant Bacteria and Infectious Disease Working Group (PIRBIG), Nakorn Pathom 73000, Thailand * Correspondence: [email protected]; Tel.: +66-3425-580-0 Received: 29 June 2020; Accepted: 31 July 2020; Published: 3 August 2020 Abstract: Global infections with colistin-resistant Pseudomonas aeruginosa (CoR-PA) are increasing; there are currently very few studies focused on the antimicrobial susceptibility of CoR-PA isolates, and none from Thailand. Here, we investigated the impact of various antimicrobials, alone and in combination, via the in vitro testing of CoR-PA clinical isolates. Eighteen CoR-PA isolates were obtained from patients treated at Phramongkutklao Hospital from January 2010 through June 2019; these were classified into six different clonal types by using the enterobacterial repetitive intergenic consensus (ERIC)-PCR method, with a high prevalence of Group A (27.8%). The antimicrobial susceptibility was determined as the minimal inhibitory concentrations (MICs) using the epsilometer-test (E-test) method. The synergistic activities of six antimicrobial combinations were reported via the fractional-inhibitory-concentration index. -

Antibiotic Resistance Patterns of Uropathogens Causing Urinary Tract Infections in Children with Congenital Anomalies of Kidney and Urinary Tract
children Article Antibiotic Resistance Patterns of Uropathogens Causing Urinary Tract Infections in Children with Congenital Anomalies of Kidney and Urinary Tract Raluca Isac 1,2 , Diana-Georgiana Basaca 2 , Ioana-Cristina Olariu 1,2, Ramona F. Stroescu 2,3, Andrada-Mara Ardelean 1,2 , Ruxandra M. Steflea 1,2, Mihai Gafencu 1,2, Adela Chirita-Emandi 2,4 , Iulia Cristina Bagiu 5,* , Florin George Horhat 5,* , Dan-Dumitru Vulcanescu 2, Dan Ionescu 6 and Gabriela Doros 1,2 1 IIIrd Pediatric Clinic, Department of Pediatrics, “Victor Babes, ” University of Medicine and Pharmacy Timis, oara, 300041 Timis, oara, Romania; [email protected] (R.I.); [email protected] (I.-C.O.); [email protected] (A.-M.A.); stefl[email protected] (R.M.S.); [email protected] (M.G.); [email protected] (G.D.) 2 Emergency Hospital for Children “Louis Turcanu”, 300011 Timis, oara, Romania; [email protected] (D.-G.B.); [email protected] (R.F.S.); [email protected] (A.C.-E.); [email protected] (D.-D.V.) 3 Ist Pediatric Clinic, Department of Pediatrics, “Victor Babes, ” University of Medicine and Pharmacy Timis, oara, 300041 Timis, oara, Romania 4 Department of Microscopic Morphology, Genetics Discipline, Center of Genomic Medicine, “Victor Babes” University of Medicine and Pharmacy, 300041 Timis, oara, Romania 5 Citation: Isac, R.; Basaca, D.-G.; Multidisciplinary Research Center on Antimicrobial Resistance (MULTI-REZ), Microbiology Department, Olariu, I.-C.; Stroescu, R.F.; Ardelean, “Victor Babes, ” University of Medicine and Pharmacy, 300041 Timis, oara, Romania 6 Physical Education and Sports Department, Polytechnic University, 300223 Timisoara, Romania; A.-M.; Steflea, R.M.; Gafencu, M.; , [email protected] Chirita-Emandi, A.; Bagiu, I.C.; * Correspondence: [email protected] (I.C.B.); horhat.fl[email protected] (F.G.H.) Horhat, F.G.; et al.