Developing of a Thermodynamic Model for the Performance Analysis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Recording and Evaluating the Pv Diagram with CASSY
LD Heat Physics Thermodynamic cycle Leaflets P2.6.2.4 Hot-air engine: quantitative experiments The hot-air engine as a heat engine: Recording and evaluating the pV diagram with CASSY Objects of the experiment Recording the pV diagram for different heating voltages. Determining the mechanical work per revolution from the enclosed area. Principles The cycle of a heat engine is frequently represented as a closed curve in a pV diagram (p: pressure, V: volume). Here the mechanical work taken from the system is given by the en- closed area: W = − ͛ p ⋅ dV (I) The cycle of the hot-air engine is often described in an idealised form as a Stirling cycle (see Fig. 1), i.e., a succession of isochoric heating (a), isothermal expansion (b), isochoric cooling (c) and isothermal compression (d). This description, however, is a rough approximation because the working piston moves sinusoidally and therefore an isochoric change of state cannot be expected. In this experiment, the pV diagram is recorded with the computer-assisted data acquisition system CASSY for comparison with the real behaviour of the hot-air engine. A pressure sensor measures the pressure p in the cylinder and a displacement sensor measures the position s of the working piston, from which the volume V is calculated. The measured values are immediately displayed on the monitor in a pV diagram. Fig. 1 pV diagram of the Stirling cycle 0210-Wei 1 P2.6.2.4 LD Physics Leaflets Setup Apparatus The experimental setup is illustrated in Fig. 2. 1 hot-air engine . 388 182 1 U-core with yoke . -

Novel Hot Air Engine and Its Mathematical Model – Experimental Measurements and Numerical Analysis
POLLACK PERIODICA An International Journal for Engineering and Information Sciences DOI: 10.1556/606.2019.14.1.5 Vol. 14, No. 1, pp. 47–58 (2019) www.akademiai.com NOVEL HOT AIR ENGINE AND ITS MATHEMATICAL MODEL – EXPERIMENTAL MEASUREMENTS AND NUMERICAL ANALYSIS 1 Gyula KRAMER, 2 Gabor SZEPESI *, 3 Zoltán SIMÉNFALVI 1,2,3 Department of Chemical Machinery, Institute of Energy and Chemical Machinery University of Miskolc, Miskolc-Egyetemváros 3515, Hungary e-mail: [email protected], [email protected], [email protected] Received 11 December 2017; accepted 25 June 2018 Abstract: In the relevant literature there are many types of heat engines. One of those is the group of the so called hot air engines. This paper introduces their world, also introduces the new kind of machine that was developed and built at Department of Chemical Machinery, Institute of Energy and Chemical Machinery, University of Miskolc. Emphasizing the novelty of construction and the working principle are explained. Also the mathematical model of this new engine was prepared and compared to the real model of engine. Keywords: Hot, Air, Engine, Mathematical model 1. Introduction There are three types of volumetric heat engines: the internal combustion engines; steam engines; and hot air engines. The first one is well known, because it is on zenith nowadays. The steam machines are also well known, because their time has just passed, even the elder ones could see those in use. But the hot air engines are forgotten. Our aim is to consider that one. The history of hot air engines is 200 years old. -

Boilers Leture Notes VUM
Steam Boilers Lecture Notes V Uma Maheshwar 11-11-11 Steam Boilers/Steam Generators Introduction: The function of a boiler is to evaporate water into steam at a pressure higher than the atmospheric pressure. Water free from impurities such as dissolved salts , gases and non soluble solids should be supplied to boilers. This is done by suitable water treatment . Steam is useful for running steam turbines in electrical power stations , ships and steam engines in railway locomotives. Boiler furnace fuel can use either solid , liquid or gaseous fuel: Wood, Charcoal, Coal, Coke, Oil, Municipal waste, industrial solid waste , refinery gas, rice husk, Paper sludge, Electric power Steam Boilers/Steam Generators Boilers are mainly classified as fire tube boilers and water tube boilers. Fire tube boilers :- hot gases from the furnace pass through the tubes which are surrounded by water. Water tube boilers :- water circulates inside the tubes which are surrounded by the hot gases from the furnace. Steam Boilers/Steam Generators Other Classifications : Based on Pressure : =>80 bar : High Pressure Boilers Babcock & Wilcox, Lamont, Benson, Velox <80 bar : Low Pressure Boilers Locomotive, Lancashire, Cornish, Cochran Steam Boilers/Steam Generators Other Classifications : Based on Pressure : =>80 bar : High Pressure Boilers Babcock & Wilcox, Lamont, Benson, Velox <80 bar : Low Pressure Boilers Locomotive, Lancashire, Cornish, Cochran Steam Boilers/Steam Generators Other Classifications : Based on Type of Circulation of water : Natural Circulation : Natural -

Comparison of ORC Turbine and Stirling Engine to Produce Electricity from Gasified Poultry Waste
Sustainability 2014, 6, 5714-5729; doi:10.3390/su6095714 OPEN ACCESS sustainability ISSN 2071-1050 www.mdpi.com/journal/sustainability Article Comparison of ORC Turbine and Stirling Engine to Produce Electricity from Gasified Poultry Waste Franco Cotana 1,†, Antonio Messineo 2,†, Alessandro Petrozzi 1,†,*, Valentina Coccia 1, Gianluca Cavalaglio 1 and Andrea Aquino 1 1 CRB, Centro di Ricerca sulle Biomasse, Via Duranti sn, 06125 Perugia, Italy; E-Mails: [email protected] (F.C.); [email protected] (V.C.); [email protected] (G.C.); [email protected] (A.A.) 2 Università degli Studi di Enna “Kore” Cittadella Universitaria, 94100 Enna, Italy; E-Mail: [email protected] † These authors contributed equally to this work. * Author to whom correspondence should be addressed; E-Mail: [email protected]; Tel.: +39-075-585-3806; Fax: +39-075-515-3321. Received: 25 June 2014; in revised form: 5 August 2014 / Accepted: 12 August 2014 / Published: 28 August 2014 Abstract: The Biomass Research Centre, section of CIRIAF, has recently developed a biomass boiler (300 kW thermal powered), fed by the poultry manure collected in a nearby livestock. All the thermal requirements of the livestock will be covered by the heat produced by gas combustion in the gasifier boiler. Within the activities carried out by the research project ENERPOLL (Energy Valorization of Poultry Manure in a Thermal Power Plant), funded by the Italian Ministry of Agriculture and Forestry, this paper aims at studying an upgrade version of the existing thermal plant, investigating and analyzing the possible applications for electricity production recovering the exceeding thermal energy. A comparison of Organic Rankine Cycle turbines and Stirling engines, to produce electricity from gasified poultry waste, is proposed, evaluating technical and economic parameters, considering actual incentives on renewable produced electricity. -

Bulletin 173 Plate 1 Smithsonian Institution United States National Museum
U. S. NATIONAL MUSEUM BULLETIN 173 PLATE 1 SMITHSONIAN INSTITUTION UNITED STATES NATIONAL MUSEUM Bulletin 173 CATALOG OF THE MECHANICAL COLLECTIONS OF THE DIVISION OF ENGINEERING UNITED STATES NATIONAL MUSEUM BY FRANK A. TAYLOR UNITED STATES GOVERNMENT PRINTING OFFICE WASHINGTON : 1939 For lale by the Superintendent of Documents, Washington, D. C. Price 50 cents ADVERTISEMENT Tlie scientific publications of the National Museum include two series, known, respectively, as Proceedings and Bulletin. The Proceedings series, begun in 1878, is intended primarily as a medium for the publication of original papers, based on the collec- tions of the National Museum, that set forth newly acquired facts in biology, anthropology, and geology, with descriptions of new forms and revisions of limited groups. Copies of each paper, in pamphlet form, are distributed as published to libraries and scientific organi- zations and to specialists and others interested in the different sub- jects. The dates at which these separate papers are published are recorded in the table of contents of each of the volumes. Tlie series of Bulletins, the first of which was issued in 1875, contains separate publications comprising monographs of large zoological groups and other general systematic treatises (occasionally in several volumes), faunal works, reports of expeditions, catalogs of type specimens and special collections, and other material of simi- lar nature. The majority of the volumes are octavo in size, but a quarto size has been adopted in a few instances in which large plates were regarded as indispensable. In the Bulletin series appear vol- umes under the heading Contrihutions from the United States Na- tional Eerharium, in octavo form, published by the National Museum since 1902, which contain papers relating to the botanical collections of the Museum. -
Stirling Cycle Working Principle
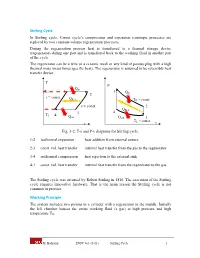
Stirling Cycle In Stirling cycle, Carnot cycle’s compression and expansion isentropic processes are replaced by two constant-volume regeneration processes. During the regeneration process heat is transferred to a thermal storage device (regenerator) during one part and is transferred back to the working fluid in another part of the cycle. The regenerator can be a wire or a ceramic mesh or any kind of porous plug with a high thermal mass (mass times specific heat). The regenerator is assumed to be reversible heat transfer device. T P Qin TH 1 1 Q 2 in v = const. Q Reg. TH = const. v = const. 2 QReg. 3 4 TL 4 Qout Qout 3 T = const. s L v Fig. 3-2: T-s and P-v diagrams for Stirling cycle. 1-2 isothermal expansion heat addition from external source 2-3 const. vol. heat transfer internal heat transfer from the gas to the regenerator 3-4 isothermal compression heat rejection to the external sink 4-1 const. vol. heat transfer internal heat transfer from the regenerator to the gas The Stirling cycle was invented by Robert Stirling in 1816. The execution of the Stirling cycle requires innovative hardware. That is the main reason the Stirling cycle is not common in practice. Working Principle The system includes two pistons in a cylinder with a regenerator in the middle. Initially the left chamber houses the entire working fluid (a gas) at high pressure and high temperature TH. M. Bahrami ENSC 461 (S 11) Stirling Cycle 1 TH TL QH State 1 Regenerator Fig 3-3a: 1-2, isothermal heat transfer to the gas at TH from external source. -

Stirling Engine Configuration Selection
energies Article Stirling Engine Configuration Selection Jose Egas 1 ID and Don M. Clucas 2,∗ ID 1 Department of Mechanical Engineering, University of Canterbury, Christchurch 8041, New Zealand; [email protected] 2 Department of Mechanical Engineering, University of Canterbury, Civil Mechanical E521, 20 Kirkwood Ave, Christchurch 8041, New Zealand * Correspondence: [email protected]; Tel: +64-3-364-2987 (ext. 92212) Received: 3 December 2017; Accepted: 6 February 2018; Published: 7 March 2018 Abstract: Unlike internal combustion engines, Stirling engines can be designed to work with many drive mechanisms based on the three primary configurations, alpha, beta and gamma. Hundreds of different combinations of configuration and mechanical drives have been proposed. Few succeed beyond prototypes. A reason for poor success is the use of inappropriate configuration and drive mechanisms, which leads to low power to weight ratio and reduced economic viability. The large number of options, the lack of an objective comparison method, and the absence of a selection criteria force designers to make random choices. In this article, the pressure—volume diagrams and compression ratios of machines of equal dimensions, using the main (alpha, beta and gamma) crank based configurations as well as rhombic drive and Ross yoke mechanisms, are obtained. The existence of a direct relation between the optimum compression ratio and the temperature ratio is derived from the ideal Stirling cycle, and the usability of an empirical low temperature difference compression ratio equation for high temperature difference applications is tested using experimental data. It is shown that each machine has a different compression ratio, making it more or less suitable for a specific application, depending on the temperature difference reachable. -

Stirling Engine for Solar Thermal Electric Generation
Stirling Engine for Solar Thermal Electric Generation Mike Miao He Electrical Engineering and Computer Sciences University of California at Berkeley Technical Report No. UCB/EECS-2018-15 http://www2.eecs.berkeley.edu/Pubs/TechRpts/2018/EECS-2018-15.html May 1, 2018 Copyright © 2018, by the author(s). All rights reserved. Permission to make digital or hard copies of all or part of this work for personal or classroom use is granted without fee provided that copies are not made or distributed for profit or commercial advantage and that copies bear this notice and the full citation on the first page. To copy otherwise, to republish, to post on servers or to redistribute to lists, requires prior specific permission. Acknowledgement I would like to thank Professor Seth Sanders for his mentorship and helping me to be a better engineer, for his perpetual good humor and kindness, and for always being available for his students. I would also like to thank Artin Der Minassians for his guidance and help. Thanks to Aron, Matt, Roger, Sarah, and Shai on the Cleantech-to-Market team for their research on the market viability of the technology. Thanks to Alan, Shong, Kelvin, Alex, and Samir and others for their help. Stirling Engine for Solar Thermal Electric Generation by Mike Miao He A dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Engineering { Electrical Engineering and Computer Sciences and the Designated Emphasis in Energy Science and Technology in the Graduate Division of the University -

A Handbook on the Steam Engine, with Especial Reference to Small And
ADVERTlSEMElfTS. i+t\\i JD, 3.E. YS. (YS, Tl sPOUS U ^rcacuteb to PI nd), of I tt of Toronto id). Professor E.A.Allcut AL. Is. Castings in Bronze, Brass and Gun and White Metals, Machined if required. ROLLED PHOSPHOR BRONZE, Strip and Sheet, for Air Pump Valves, Eccentric Strap Liners, etc. HIKE find TELEPHONE WIRE. AD VER TISEMENTS. THE PHOSPHOR BRONZE CO, LIMITED, 87, Sumner Street, Southwark, London, S.E. And at BIRMINGHAM. r "COG WHEEL" and Sole Makers of the j3S %jg "VULCAN" iSPI^flfiC Brands of The best and most durable Alloys for Slide Valves, Bearings, Bushes, Eccentric Straps, and other parts of Machinery exposed to friction and Piston Motor wear ; Pump Rods, Pumps, Rings, Pinions, Worm Wheels, Gearing, etc. "DURO METAL" (REGISTERED TRADE MARK). Alloy B, specially adapted for BEARINGS for HOT-NECK ROLLS. CASTINGS In BRONZE, BRASS, GUN and WHITE METALS, in the Rough, or Machined, if required. ROLLED AND DRAWN BRONZE, GERMAN SILVER, GUN METAL, TIN, WHITE METALS AND ALUMINIUM BRONZE ALLOYS PHOSPHOR TIN & PHOSPHOR COPPER, "Cog-Wheel" Brand. Please specify the Manufacture of THE PHOSPHOR BRONZE CO., Ltd., of Southwark, London. lii ADVERTISEMENTS, ROBEY & Co GLOBE WORKS, LINCOLN. Mod Compound Horizontal Fixed Engine, it.- 1 iiii I'.ii.-nt Ti-ip Kxitiimioii (ic:ir. tolnijthe limpl >nUit ml miTrt economical at uuv in tin- market, and working ;.|iei to the Newcastle-nil -Tvm .tit Station (MX large engines), also St. Helens mint lam. BrUbane Electric Tram- Open-front High-speed Vertical e, for electric lighting. All thete Engine* are specially Dengned and Adapted for Electric Lighting. -

Operation of Thermoacoustic Stirling Heat Engine Driven Large Multiple Pulse Tube Refrigerators
LA-UR-04-2820, submitted to the proceedings of the 13th Int'l Cryocooler Conf. 1 Operation of Thermoacoustic Stirling Heat Engine Driven Large Multiple Pulse Tube Refrigerators Bayram Arman, John Wollan, and Vince Kotsubo Praxair, Inc. Tonawanda, NY 14150 Scott Backhaus and Greg Swift Los Alamos National Laboratory Los Alamos, NM 87545 ABSTRACT With support from Los Alamos National Laboratory, Praxair has been developing ther- moacoustic Stirling heat engines and refrigerators for liquefaction of natural gas. The combina- tion of thermoacoustic engines with pulse tube refrigerators is the only technology capable of producing significant cryogenic refrigeration with no moving parts. A prototype, powered by a natural-gas burner and with a projected natural-gas-liquefaction capacity of 500 gal/day, has been built and tested. The unit has liquefied 350 gal/day, with a projected production efficiency of 70% liquefaction and 30% combustion of an incoming gas stream. A larger system, intended to have a liquefaction capacity of 20,000 gal/day and an efficiency of 80 to 85% liquefaction, has undergone preliminary design. In the 500 gal/day system, the combustion-powered thermoacoustic Stirling heat engine drives three pulse tube refrigerators to generate refrigeration at methane liquefaction tempera- tures. Each refrigerator was designed to produce over 2 kW of refrigeration. The orifice valves of the three refrigerators were adjusted to eliminate Rayleigh streaming in the pulse tubes. This pa- per describes the hardware, operating experience, and some recent test results. INTRODUCTION Praxair has been developing thermoacoustic liquefiers and refrigerators for liquefaction of natural gas and for other cryogenic applications. -

Lecture 3 Thermodynamic Principles of Energy Conversion
Thermodynamic principles of energy conversion Md. Mizanur Rahman School of Mechanical Engineering Universiti Teknologi Malaysia 1 Introduction • Mechanical and electrical power developed from the combustion of fossil fuels or the fission of nuclear fuel or renewable sources • This released energy is never lost but is transformed into other forms • This conservation of energy is explicitly expressed by the first law of thermodynamics • The work of an engine cannot be the same as the energy available in the fuel sources • Second law of thermodynamics is to meet to happen the process 2 Forms of energy • Energy can exist in numerous forms such as thermal, mechanical, kinetic, potential, electric, magnetic, chemical, and nuclear, and their sum constitutes the total energy, E of a system. • Mechanical Energy • Kinetic energy, KE: The energy that a system possesses as a result of its motion relative to some reference frame, KE=1/2mV2. • Potential energy, PE: The energy that a system possesses as a result of its elevation in a gravitational field, PE=mgh • For a moving body, E=PE+KE remains constant 3 Internal energy, U: The sum of all the microscopic forms of energy. • For gases, molecules are so widely separated in space that they may be considered to be moving independently of each other, each possessing a distinct total energy. • In the case of liquids or solids, each molecule is under the influence of forces exerted by nearby molecules • Changes in internal energy are measurable by changes in temperature, pressure, and density. 4 • Chemical energy: The internal energy associated with the atomic bonds in a molecule. -

The Stirling Cycle the Ultimate Way to Generate Cooling Power
The Stirling Cycle The ultimate way to generate cooling power History The Stirling cycle is a thermodynamic closed cycle invented in 1816 by the Scottish minister Robert Stirling. It was used as an engine and was considered at the time to be capable of replacing the steam engine since their steam boilers were prone to life- threatening explosions. The counterpart of the Stirling engine, the refrigerator, was first recognized in 1832. Both machines experienced high and low points during the nineteenth century. The principle behind the machines was almost condemned to obscurity after the invention of the internal combustion engine (gas-, petrol-, and diesel motors) and compressor refrigerators with external evaporation. In 1938 the famous Dutch Philips Research Laboratory was looking for a means to power electricity generators for short wave communication systems in remote areas without electricity supply. The practically-forgotten Stirling engine attracted their attention. In 1946 Philips started optimizing the Stirling cycle to be used for cryogenic cooling. The result was the development of the world conquering Stirling Cryogenerator, marking the start of significant cryogenic activities at Philips. Though the Stirling engine itself never became a commercial success, the Stirling Cryogenerator has been selling by thousands worldwide and has been incorporated in equipment and projects used from Antarctica to the North Pole. Today In the beginning of the 90’s the Philips Cryogenic Division became independent and continued its worldwide activities under the name of Stirling Cryogenics BV. Since 2014, DH Industries BV incorporated the Stirling Cryogenics products as a brand alongside its CryoZone brand. DH Industries is the only company in the world that successfully produces Stirling cycle- based cryogenerators with cooling powers of 1,000-4,000 watt (at 77K) per unit.