Sox2 Promotes Malignancy in Glioblastoma by Regulating
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The T-ALL Related Gene BCL11B Regulates the Initial Stages of Human T-Cell Differentiation
Leukemia (2017) 31, 2503–2514 © 2017 Macmillan Publishers Limited, part of Springer Nature. All rights reserved 0887-6924/17 www.nature.com/leu ORIGINAL ARTICLE The T-ALL related gene BCL11B regulates the initial stages of human T-cell differentiation VL Ha1, A Luong1,FLi2, D Casero3, J Malvar1,YMKim1,4, R Bhatia5, GM Crooks3,6,7,8 and C Parekh1,4 The initial stages of T-cell differentiation are characterized by a progressive commitment to the T-cell lineage, a process that involves the loss of alternative (myelo-erythroid, NK, B) lineage potentials. Aberrant differentiation during these stages can result in T-cell acute lymphoblastic leukemia (T-ALL). However, the mechanisms regulating the initial stages of human T-cell differentiation are obscure. Through loss of function studies, we showed BCL11B, a transcription factor recurrently mutated T-ALL, is essential for T-lineage commitment, particularly the repression of NK and myeloid potentials, and the induction of T-lineage genes, during the initial stages of human T-cell differentiation. In gain of function studies, BCL11B inhibited growth of and induced a T-lineage transcriptional program in T-ALL cells. We found previously unknown differentiation stage-specific DNA binding of BCL11B at multiple T-lineage genes; target genes showed BCL11B-dependent expression, suggesting a transcriptional activator role for BCL11B at these genes. Transcriptional analyses revealed differences in the regulatory actions of BCL11B between human and murine thymopoiesis. Our studies show BCL11B is a key regulator of the initial stages of human T-cell differentiation and delineate the BCL11B transcriptional program, enabling the dissection of the underpinnings of normal T-cell differentiation and providing a resource for understanding dysregulations in T-ALL. -

The Rise and Fall of the Bovine Corpus Luteum
University of Nebraska Medical Center DigitalCommons@UNMC Theses & Dissertations Graduate Studies Spring 5-6-2017 The Rise and Fall of the Bovine Corpus Luteum Heather Talbott University of Nebraska Medical Center Follow this and additional works at: https://digitalcommons.unmc.edu/etd Part of the Biochemistry Commons, Molecular Biology Commons, and the Obstetrics and Gynecology Commons Recommended Citation Talbott, Heather, "The Rise and Fall of the Bovine Corpus Luteum" (2017). Theses & Dissertations. 207. https://digitalcommons.unmc.edu/etd/207 This Dissertation is brought to you for free and open access by the Graduate Studies at DigitalCommons@UNMC. It has been accepted for inclusion in Theses & Dissertations by an authorized administrator of DigitalCommons@UNMC. For more information, please contact [email protected]. THE RISE AND FALL OF THE BOVINE CORPUS LUTEUM by Heather Talbott A DISSERTATION Presented to the Faculty of the University of Nebraska Graduate College in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy Biochemistry and Molecular Biology Graduate Program Under the Supervision of Professor John S. Davis University of Nebraska Medical Center Omaha, Nebraska May, 2017 Supervisory Committee: Carol A. Casey, Ph.D. Andrea S. Cupp, Ph.D. Parmender P. Mehta, Ph.D. Justin L. Mott, Ph.D. i ACKNOWLEDGEMENTS This dissertation was supported by the Agriculture and Food Research Initiative from the USDA National Institute of Food and Agriculture (NIFA) Pre-doctoral award; University of Nebraska Medical Center Graduate Student Assistantship; University of Nebraska Medical Center Exceptional Incoming Graduate Student Award; the VA Nebraska-Western Iowa Health Care System Department of Veterans Affairs; and The Olson Center for Women’s Health, Department of Obstetrics and Gynecology, Nebraska Medical Center. -

MLL1 and DOT1L Cooperate with Meningioma-1 to Induce Acute Myeloid Leukemia
MLL1 and DOT1L cooperate with meningioma-1 to induce acute myeloid leukemia Simone S. Riedel, … , Tobias Neff, Kathrin M. Bernt J Clin Invest. 2016;126(4):1438-1450. https://doi.org/10.1172/JCI80825. Research Article Oncology Meningioma-1 (MN1) overexpression is frequently observed in patients with acute myeloid leukemia (AML) and is predictive of poor prognosis. In murine models, forced expression of MN1 in hematopoietic progenitors induces an aggressive myeloid leukemia that is strictly dependent on a defined gene expression program in the cell of origin, which includes the homeobox genes Hoxa9 and Meis1 as key components. Here, we have shown that this program is controlled by two histone methyltransferases, MLL1 and DOT1L, as deletion of either Mll1 or Dot1l in MN1-expressing cells abrogated the cell of origin–derived gene expression program, including the expression of Hoxa cluster genes. In murine models, genetic inactivation of either Mll1 or Dot1l impaired MN1-mediated leukemogenesis. We determined that HOXA9 and MEIS1 are coexpressed with MN1 in a subset of clinical MN1hi leukemia, and human MN1hi/HOXA9hi leukemias were sensitive to pharmacologic inhibition of DOT1L. Together, these data point to DOT1L as a potential therapeutic target in MN1hi AML. In addition, our findings suggest that epigenetic modulation of the interplay between an oncogenic lesion and its cooperating developmental program has therapeutic potential in AML. Find the latest version: https://jci.me/80825/pdf RESEARCH ARTICLE The Journal of Clinical Investigation MLL1 and DOT1L cooperate with meningioma-1 to induce acute myeloid leukemia Simone S. Riedel,1 Jessica N. Haladyna,1 Matthew Bezzant,1 Brett Stevens,2 Daniel A. -

Universidade Estadual De Campinas Instituto De Biologia
UNIVERSIDADE ESTADUAL DE CAMPINAS INSTITUTO DE BIOLOGIA VERÔNICA APARECIDA MONTEIRO SAIA CEREDA O PROTEOMA DO CORPO CALOSO DA ESQUIZOFRENIA THE PROTEOME OF THE CORPUS CALLOSUM IN SCHIZOPHRENIA CAMPINAS 2016 1 VERÔNICA APARECIDA MONTEIRO SAIA CEREDA O PROTEOMA DO CORPO CALOSO DA ESQUIZOFRENIA THE PROTEOME OF THE CORPUS CALLOSUM IN SCHIZOPHRENIA Dissertação apresentada ao Instituto de Biologia da Universidade Estadual de Campinas como parte dos requisitos exigidos para a obtenção do Título de Mestra em Biologia Funcional e Molecular na área de concentração de Bioquímica. Dissertation presented to the Institute of Biology of the University of Campinas in partial fulfillment of the requirements for the degree of Master in Functional and Molecular Biology, in the area of Biochemistry. ESTE ARQUIVO DIGITAL CORRESPONDE À VERSÃO FINAL DA DISSERTAÇÃO DEFENDIDA PELA ALUNA VERÔNICA APARECIDA MONTEIRO SAIA CEREDA E ORIENTADA PELO DANIEL MARTINS-DE-SOUZA. Orientador: Daniel Martins-de-Souza CAMPINAS 2016 2 Agência(s) de fomento e nº(s) de processo(s): CNPq, 151787/2F2014-0 Ficha catalográfica Universidade Estadual de Campinas Biblioteca do Instituto de Biologia Mara Janaina de Oliveira - CRB 8/6972 Saia-Cereda, Verônica Aparecida Monteiro, 1988- Sa21p O proteoma do corpo caloso da esquizofrenia / Verônica Aparecida Monteiro Saia Cereda. – Campinas, SP : [s.n.], 2016. Orientador: Daniel Martins de Souza. Dissertação (mestrado) – Universidade Estadual de Campinas, Instituto de Biologia. 1. Esquizofrenia. 2. Espectrometria de massas. 3. Corpo caloso. -
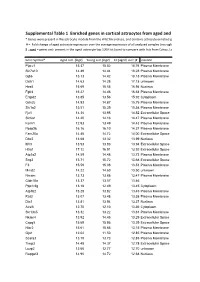
Supplemental Table 1 Enriched Genes in Cortical Astrocytes from Aged
Supplemental Table 1 Enriched genes in cortical astrocytes from aged and young-adult mice * Genes were present in the astrocyte module from the WGCNA analysis, and contains astrocyte enriched genes compared to microglia and oligodendrocytes # = Fold change of aged astrocyte expression over the average expression of all analyzed samples (microglia, astrocytes: young, old, with and without myelin contamination) $ ; aged = genes only present in the aged astrocyte top 1000 list (used to compare with lists from Cahoy, Lovatt, Doyle; see Fig. 4B), all = genes present in all astrocyte top 1000 lists Gene Symbol* Aged astr. (log2) Young astr.(log2) FC (aged/ aver.)# Location Ptprz1 15.37 15.02 18.76 Plasma Membrane Slc7a10 14.49 14.44 18.28 Plasma Membrane Gjb6 15.13 14.42 18.18 Plasma Membrane Dclk1 14.63 14.28 17.18 unknown Hes5 15.69 15.55 16.94 Nucleus Fgfr3 15.27 14.46 16.54 Plasma Membrane Entpd2 13.85 13.56 15.92 Cytoplasm Grin2c 14.93 14.87 15.75 Plasma Membrane Slc1a2 15.51 15.39 15.58 Plasma Membrane Fjx1 14.36 13.98 14.52 Extracellular Space Slc6a1 14.20 14.16 14.47 Plasma Membrane Kcnk1 12.93 13.49 14.43 Plasma Membrane Ppap2b 16.16 16.10 14.37 Plasma Membrane Fam20a 14.48 14.72 14.00 Extracellular Space Dbx2 13.68 13.32 13.99 Nucleus Itih3 13.93 13.93 13.94 Extracellular Space Htra1 17.12 16.91 13.92 Extracellular Space Atp1a2 14.59 14.48 13.73 Plasma Membrane Scg3 15.71 15.72 13.68 Extracellular Space F3 15.59 15.08 13.51 Plasma Membrane Mmd2 14.22 14.60 13.50 unknown Nrcam 13.73 13.88 13.47 Plasma Membrane Cldn10a 13.37 13.57 13.46 -

Propranolol-Mediated Attenuation of MMP-9 Excretion in Infants with Hemangiomas
Supplementary Online Content Thaivalappil S, Bauman N, Saieg A, Movius E, Brown KJ, Preciado D. Propranolol-mediated attenuation of MMP-9 excretion in infants with hemangiomas. JAMA Otolaryngol Head Neck Surg. doi:10.1001/jamaoto.2013.4773 eTable. List of All of the Proteins Identified by Proteomics This supplementary material has been provided by the authors to give readers additional information about their work. © 2013 American Medical Association. All rights reserved. Downloaded From: https://jamanetwork.com/ on 10/01/2021 eTable. List of All of the Proteins Identified by Proteomics Protein Name Prop 12 mo/4 Pred 12 mo/4 Δ Prop to Pred mo mo Myeloperoxidase OS=Homo sapiens GN=MPO 26.00 143.00 ‐117.00 Lactotransferrin OS=Homo sapiens GN=LTF 114.00 205.50 ‐91.50 Matrix metalloproteinase‐9 OS=Homo sapiens GN=MMP9 5.00 36.00 ‐31.00 Neutrophil elastase OS=Homo sapiens GN=ELANE 24.00 48.00 ‐24.00 Bleomycin hydrolase OS=Homo sapiens GN=BLMH 3.00 25.00 ‐22.00 CAP7_HUMAN Azurocidin OS=Homo sapiens GN=AZU1 PE=1 SV=3 4.00 26.00 ‐22.00 S10A8_HUMAN Protein S100‐A8 OS=Homo sapiens GN=S100A8 PE=1 14.67 30.50 ‐15.83 SV=1 IL1F9_HUMAN Interleukin‐1 family member 9 OS=Homo sapiens 1.00 15.00 ‐14.00 GN=IL1F9 PE=1 SV=1 MUC5B_HUMAN Mucin‐5B OS=Homo sapiens GN=MUC5B PE=1 SV=3 2.00 14.00 ‐12.00 MUC4_HUMAN Mucin‐4 OS=Homo sapiens GN=MUC4 PE=1 SV=3 1.00 12.00 ‐11.00 HRG_HUMAN Histidine‐rich glycoprotein OS=Homo sapiens GN=HRG 1.00 12.00 ‐11.00 PE=1 SV=1 TKT_HUMAN Transketolase OS=Homo sapiens GN=TKT PE=1 SV=3 17.00 28.00 ‐11.00 CATG_HUMAN Cathepsin G OS=Homo -

SUPPLEMENTAL DATA Supplemental Materials And
SUPPLEMENTAL DATA Supplemental Materials and Methods Cells and Cell Culture Human breast carcinoma cell lines, MDA-MB-231 and MCF7, were purchased from American Type Tissue Culture Collection (ATCC). 231BoM-1833, 231BrM-2a, CN34, CN34-BoM2d, CN34-BrM2c and MCF7- BoM2d cell lines were kindly provided by Dr. Joan Massagué (Memorial Sloan-Kettering Cancer Center) (1-3). Luciferase-labeled cells were generated by infecting the lentivirus carrying the firefly luciferase gene. The immortalized mouse bone microvascular endothelial cell (mBMEC) was a generous gift from Dr. Isaiah J. Fidler (M.D. Anderson Cancer Center) (4). MCF10A and MCF10DCIS.com cells were purchased from ATCC and Asterand, respectively. MDA-MB-231, its variant cells, MCF7 and MCF-BoM2d cells were cultured in DMEM medium supplemented with 10% FBS and antibiotics. CN34 and its variant cells were cultured in Medium199 supplemented with 2.5% FBS, 10 µg/ml insulin, 0.5 µg/ml hydrocortisone, 20 ng/ml EGF, 100 ng/ml cholera toxin and antibiotics. MCF10DCIS.com cells were cultured in RPMI-1640 medium supplemented with 10% FBS and antibiotics. MCF10A cells were cultured in MEGM mammary epithelial cell growth medium (Lonza). mBMEC was maintained at 8% CO2 at 33 °C in DMEM with 10% FBS, 2 mM L-glutamine, 1 mM sodium pyruvate, 1% non-essential amino acids and 1% vitamin mixture. Bone marrow stromal fibroblast cell lines HS5 and HS27A, and osteoblast cell line, hFOB1.19, were purchased from ATCC. Bone marrow derived human mesenchymal stem cells, BM-hMSC, were isolated for enrichment of plastic adherent cells from unprocessed bone marrow (Lonza) which was depleted of red blood cells. -

New Approaches to Functional Process Discovery in HPV 16-Associated Cervical Cancer Cells by Gene Ontology
Cancer Research and Treatment 2003;35(4):304-313 New Approaches to Functional Process Discovery in HPV 16-Associated Cervical Cancer Cells by Gene Ontology Yong-Wan Kim, Ph.D.1, Min-Je Suh, M.S.1, Jin-Sik Bae, M.S.1, Su Mi Bae, M.S.1, Joo Hee Yoon, M.D.2, Soo Young Hur, M.D.2, Jae Hoon Kim, M.D.2, Duck Young Ro, M.D.2, Joon Mo Lee, M.D.2, Sung Eun Namkoong, M.D.2, Chong Kook Kim, Ph.D.3 and Woong Shick Ahn, M.D.2 1Catholic Research Institutes of Medical Science, 2Department of Obstetrics and Gynecology, College of Medicine, The Catholic University of Korea, Seoul; 3College of Pharmacy, Seoul National University, Seoul, Korea Purpose: This study utilized both mRNA differential significant genes of unknown function affected by the display and the Gene Ontology (GO) analysis to char- HPV-16-derived pathway. The GO analysis suggested that acterize the multiple interactions of a number of genes the cervical cancer cells underwent repression of the with gene expression profiles involved in the HPV-16- cancer-specific cell adhesive properties. Also, genes induced cervical carcinogenesis. belonging to DNA metabolism, such as DNA repair and Materials and Methods: mRNA differential displays, replication, were strongly down-regulated, whereas sig- with HPV-16 positive cervical cancer cell line (SiHa), and nificant increases were shown in the protein degradation normal human keratinocyte cell line (HaCaT) as a con- and synthesis. trol, were used. Each human gene has several biological Conclusion: The GO analysis can overcome the com- functions in the Gene Ontology; therefore, several func- plexity of the gene expression profile of the HPV-16- tions of each gene were chosen to establish a powerful associated pathway, identify several cancer-specific cel- cervical carcinogenesis pathway. -

UNIVERSITY of CALIFORNIA Los Angeles
UNIVERSITY OF CALIFORNIA Los Angeles Circadian Gene Networks In Bone Regeneration A thesis submitted in partial satisfaction of the requirements for the degree Master of Science in Oral Biology by Nathaniel Raphael Hassan 2012 ABSTRACT OF THESIS Circadian Gene Networks In Bone Regeneration By Nathaniel Raphael Hassan Master of Science in Oral Biology University of California, Los Angeles, 2012 Professor Ichiro Nishimura, Chair BACKGROUND: Previous studies suggested that vitamin D played a significant role in bone regeneration, facilitating the establishment of implant osseointegration. A whole genome microarray study further suggested that the vitamin D axis might involve circadian rhythm gene expression in the bone peripheral tissue. OBJECTIVES: To identify key gene networks involved with vitamin D receptor in the bone regeneration process and to explore any correlation with circadian rhythm gene expression in bone marrow mesenchymal stromal/stem cells (BMSC). METHODS: The available whole gene microarray data was analyzed using the weighted gene correlation network analysis (WGCNA) R package and Cytoscape software. Any gene expression correlation was examined for vitamin D receptor (VDR) as well as circadian rhythm ii genes. Separately, Per 1 luciferase transgenic Wistar rats were then applied for in vitro evaluation on BMSC circadian rhythm. Per1::luc BMSCs were seeded on 35mm dishes and forskolin-synchronized luminescence was recorded across different media conditions. The recording media included growth medium containing F12 (10% Fetal Bovine Serum, 1% Pen- Strep and antibiotic) supplemented with or without 1 nM 1,25D. Luminescence was also recorded in F12 growth medium containing bone differentiation factors (beta glycerophosphate, Ascorbic acid and Dexamethasone) supplemented with 0, 1 or 10 nM 1,25D. -

NPAS2 As a Transcriptional Regulator of Non-Rapid Eye Movement Sleep: Genotype and Sex Interactions
NPAS2 as a transcriptional regulator of non-rapid eye movement sleep: Genotype and sex interactions Paul Franken*†‡, Carol A. Dudley§, Sandi Jo Estill§, Monique Barakat*, Ryan Thomason¶, Bruce F. O’Hara¶, and Steven L. McKnight‡§ §Department of Biochemistry, University of Texas Southwestern Medical Center, Dallas, TX 75390; *Department of Biological Sciences, Stanford University, Stanford, CA 94305; ¶Department of Biology, University of Kentucky, Lexington, KY 40506; and †Center for Integrative Genomics, University of Lausanne, CH-1015 Lausanne-Dorigny, Switzerland Contributed by Steven L. McKnight, March 13, 2006 Because the transcription factor neuronal Per-Arnt-Sim-type sig- delta frequency range is a sensitive marker of time spent awake (4, nal-sensor protein-domain protein 2 (NPAS2) acts both as a sensor 7) and local cortical activation (8) and is therefore widely used as and an effector of intracellular energy balance, and because sleep an index of NREMS need and intensity. is thought to correct an energy imbalance incurred during waking, The PAS-domain proteins, CLOCK, BMAL1, PERIOD-1 we examined NPAS2’s role in sleep homeostasis using npas2 (PER1), and PER2, play crucial roles in circadian rhythm gener- knockout (npas2؊/؊) mice. We found that, under conditions of ation (9). The NPAS2 paralog CLOCK, like NPAS2, can induce the increased sleep need, i.e., at the end of the active period or after transcription of per1, per2, cryptochrome-1 (cry1), and cry2. PER and sleep deprivation (SD), NPAS2 allows for sleep to occur at times CRY proteins, in turn, inhibit CLOCK- and NPAS2-induced when mice are normally awake. Lack of npas2 affected electroen- transcription, thereby closing a negative-feedback loop that is cephalogram activity of thalamocortical origin; during non-rapid thought to underlie circadian rhythm generation. -

) (51) International Patent Classification: Columbia V5G 1G3
) ( (51) International Patent Classification: Columbia V5G 1G3 (CA). PANDEY, Nihar R.; 10209 A 61K 31/4525 (2006.01) C07C 39/23 (2006.01) 128A St, Surrey, British Columbia V3T 3E7 (CA). A61K 31/05 (2006.01) C07D 405/06 (2006.01) (74) Agent: ZIESCHE, Sonia et al.; Gowling WLG (Canada) A61P25/22 (2006.01) LLP, 2300 - 550 Burrard Street, Vancouver, British Colum¬ (21) International Application Number: bia V6C 2B5 (CA). PCT/CA2020/050165 (81) Designated States (unless otherwise indicated, for every (22) International Filing Date: kind of national protection av ailable) . AE, AG, AL, AM, 07 February 2020 (07.02.2020) AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DJ, DK, DM, DO, (25) Filing Language: English DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, HN, (26) Publication Language: English HR, HU, ID, IL, IN, IR, IS, JO, JP, KE, KG, KH, KN, KP, KR, KW, KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, (30) Priority Data: MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, 16/270,389 07 February 2019 (07.02.2019) US OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, (63) Related by continuation (CON) or continuation-in-part SC, SD, SE, SG, SK, SL, ST, SV, SY, TH, TJ, TM, TN, TR, (CIP) to earlier application: TT, TZ, UA, UG, US, UZ, VC, VN, WS, ZA, ZM, ZW. US 16/270,389 (CON) (84) Designated States (unless otherwise indicated, for every Filed on 07 Februaiy 2019 (07.02.2019) kind of regional protection available) . -

Figure S1. Representative Report Generated by the Ion Torrent System Server for Each of the KCC71 Panel Analysis and Pcafusion Analysis
Figure S1. Representative report generated by the Ion Torrent system server for each of the KCC71 panel analysis and PCaFusion analysis. (A) Details of the run summary report followed by the alignment summary report for the KCC71 panel analysis sequencing. (B) Details of the run summary report for the PCaFusion panel analysis. A Figure S1. Continued. Representative report generated by the Ion Torrent system server for each of the KCC71 panel analysis and PCaFusion analysis. (A) Details of the run summary report followed by the alignment summary report for the KCC71 panel analysis sequencing. (B) Details of the run summary report for the PCaFusion panel analysis. B Figure S2. Comparative analysis of the variant frequency found by the KCC71 panel and calculated from publicly available cBioPortal datasets. For each of the 71 genes in the KCC71 panel, the frequency of variants was calculated as the variant number found in the examined cases. Datasets marked with different colors and sample numbers of prostate cancer are presented in the upper right. *Significantly high in the present study. Figure S3. Seven subnetworks extracted from each of seven public prostate cancer gene networks in TCNG (Table SVI). Blue dots represent genes that include initial seed genes (parent nodes), and parent‑child and child‑grandchild genes in the network. Graphical representation of node‑to‑node associations and subnetwork structures that differed among and were unique to each of the seven subnetworks. TCNG, The Cancer Network Galaxy. Figure S4. REVIGO tree map showing the predicted biological processes of prostate cancer in the Japanese. Each rectangle represents a biological function in terms of a Gene Ontology (GO) term, with the size adjusted to represent the P‑value of the GO term in the underlying GO term database.