Allergy Testing Instructions
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medication Avoidance List for Skin Testing
D. L. Southern, M.D. A. Pedinoff, M.D. J. Caucino, D.O. H. Skolnick, M.D. K. Sikorski, M.D. S. Shah, M.D. N. Baman, M.D. Princeton 609-921-2202 Plainsboro 609-799-8111 Hamilton 609-888-1555 Flemington 908-782-0093 Fax #: 609-924-1468 Your skin testing or food/medication challenge has been scheduled for ______________________________________. The following is a list of commonly used antihistamines and medications, which can interfere with and reduce the accuracy of allergy testing. Please read carefully the times a medication needs to be discontinued prior to testing: Avoid the following antihistamine medications for 3 full days prior to skin testing: Benadryl=diphenydramine Phenergan cough syrup=promethazine Ru-Tuss, Triaminic=pheniramine Polaramine=tripelenamine Dramamine,Bonine=meclizine Bromfed, Dimetapp=brompheniramine Nolahist, Nolamine=phenindamine Actifed=triprolidine Trinalin=azatadine Avoid the following antihistamine medications for 5 full days prior to skin testing: Atarax=hydroxyzine Allegra=fexofenadine Polaramine=dexachlorpheniramine Zyrtec=cetirizine Avoid the following antihistamine medication for 7 full days prior to skin testing: Claritin=loratidine Clarinex=desloratidine Tavist=clemastine Xyzal=levocetirizine Avoid the following antihistamine for 9 full days prior to skin testing: Periactin=cyproheptadine Avoid the following nasal antihistamine sprays for 5 days prior to skin testing: Dymista, Astepro, Astelin=azelastine Patanase=olapatadine Avoid the following stomach medications for 3 days prior to skin testing: Zantac=ranitidine -

San Diego ENT
San Diego ENT Allergy Skin Testing Instructions Make sure you review all of your medications with your doctor or the medical assistant when you are scheduled for your allergy test. DON’T’S: • Do note take over-the-counter antihistamines, cold medication, or cough syrup for 10 days prior to the test. This includes Benadryl, Claritin, Zyrtec, Allegra, loratadine, cetirizine, and fexofenadine, Tavist, Dramamine,Atarax, and others. • Do not take prescription antihistamines for 10 days prior to the test including Claritin, Zyrtec, Allegra, loratadine, cetirizine, fexofenadine, and Astelin nasal spray. Also stop antihistamine eye drops 10 days prior to testing. • Do not take beta blockers 5 days prior to testing. These include labetalol, metoprolol, carvedilol, and their brand name equivalents. • Do not take anti-acid medication for 48 hours prior to testing including Zantac, Pepcid, and Tagamet. You may continue to take proton pump inhibitors such as Prilosec, Nexium, Prevacid, Protonix, and Aciphex. • Do not take any sleeping medications for 48 hours prior to testing including Tylenol PM and Excedrin PM. DO’S: • Wear something comfortable that will allow access to your back or both upper arms on the day of testing. • You may continue to use nasal steroid sprays such as Flonase, Nasonex, Nasacort, and Rhinocort. Do not use Astelin. • Review the list of medications that need to be avoided below. Antihistamines to stop 10 days prior to testing: Generic Brand Name Acrivastine Semprex Azatadine Optimine, Trinalin Bropheniramine AccuHist, Bromfed, -
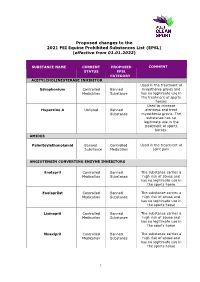
Proposed Changes to the 2021 FEI Equine Prohibited Substances List (EPSL) (Effective from 01.01.2022)
Proposed changes to the 2021 FEI Equine Prohibited Substances List (EPSL) (effective from 01.01.2022) SUBSTANCE NAME CURRENT PROPOSED COMMENT STATUS EPSL CATEGORY ACETYLCHOLINESTERASE INHIBITOR Used in the treatment of Edrophonium Controlled Banned myasthenia gravis and Medication Substance has no legitimate use in the treatment of sports horses Used to increase Huperzine A Unlisted Banned alertness and treat Substance myasthenia gravis. The substance has no legitimate use in the treatment of sports horses. AMIDES Palmitoylethanolamid Banned Controlled Used in the treatment of Substance Medication joint pain ANGIOTENSIN CONVERTING ENZYME INHIBITORS Enalapril Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse Enalaprilat Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse Lisinopril Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse Moexipril Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse 1 Perindoprilat Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse ANTIHISTAMINES Antazoline Controlled Banned The substance has no Medication Substance legitimate use in the sports horse Azatadine Controlled Banned The substance has Medication Substance sedative effects -
![With [3H]Mepyramine (Trieyclic Antidepressants/Antihistamine/Neurotransmitter/Amitriptyline) VINH TAN TRAN, RAYMOND S](https://docslib.b-cdn.net/cover/2862/with-3h-mepyramine-trieyclic-antidepressants-antihistamine-neurotransmitter-amitriptyline-vinh-tan-tran-raymond-s-1512862.webp)
With [3H]Mepyramine (Trieyclic Antidepressants/Antihistamine/Neurotransmitter/Amitriptyline) VINH TAN TRAN, RAYMOND S
Proc. Nati. Acad. Sci. USA Vol. 75, No. 12, pp. 6290-6294,, December 1978 Neurobiology Histamine H1 receptors identified in mammalian brain membranes with [3H]mepyramine (trieyclic antidepressants/antihistamine/neurotransmitter/amitriptyline) VINH TAN TRAN, RAYMOND S. L. CHANG, AND SOLOMON H. SNYDER* Departments of Pharmacology and Experimental Therapeutics, and Psychiatry and Behavioral Sciences, Johns Hopkins University School of Medicine, Baltimore, Maryland 21205 Communicated by Julius Axelrod, August 30,1978 ABSTRACT The antihistamine [3H mepyramine binds to Male Sprague-Dawley rats (150-200 g) were killed by cer- HI histamine receptors in mammalian brain membranes. vical dislocation, their brains were rapidly removed and ho- Potencies of H1 antihistamines at the binding sites correlate mogenized with a Polytron for 30 min (setting 5) in 30 vol of with their pharmacological antihistamine effects in the guinea pig ileum. Specific [3Himepyramine binding is saturable with ice-cold Na/K phosphate buffer (50 mM, pH 7.5), and the a dissociation constant of about 4 nM in both equilibrium and suspension was centrifuged (50,000 X g for 10 min). The pellet kinetic experiments and a density of 10pmolper gram ofwhole was resuspended in the same volume of fresh buffer and cen- brain. Some tricyclic antidepressants are potent inhibitors of trifuged, and the final pellet was resuspended in the original secific [3Hmepamine binding. Regional variations of volume of ice-cold buffer by Polytron homogenization. Calf [3Hjmepyramine ing do not correlate with variations in brains were obtained from a local abattoir within 2 hr after the endogeneous histamine and histidine decarboxylase activity. death of the animals and transferred to the laboratory in ice- Histamine is a neurotransmitter candidate in mammalian brain cold saline. -

Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DIX to the HTSUS—Continued
20558 Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DEPARMENT OF THE TREASURY Services, U.S. Customs Service, 1301 TABLE 1.ÐPHARMACEUTICAL APPEN- Constitution Avenue NW, Washington, DIX TO THE HTSUSÐContinued Customs Service D.C. 20229 at (202) 927±1060. CAS No. Pharmaceutical [T.D. 95±33] Dated: April 14, 1995. 52±78±8 ..................... NORETHANDROLONE. A. W. Tennant, 52±86±8 ..................... HALOPERIDOL. Pharmaceutical Tables 1 and 3 of the Director, Office of Laboratories and Scientific 52±88±0 ..................... ATROPINE METHONITRATE. HTSUS 52±90±4 ..................... CYSTEINE. Services. 53±03±2 ..................... PREDNISONE. 53±06±5 ..................... CORTISONE. AGENCY: Customs Service, Department TABLE 1.ÐPHARMACEUTICAL 53±10±1 ..................... HYDROXYDIONE SODIUM SUCCI- of the Treasury. NATE. APPENDIX TO THE HTSUS 53±16±7 ..................... ESTRONE. ACTION: Listing of the products found in 53±18±9 ..................... BIETASERPINE. Table 1 and Table 3 of the CAS No. Pharmaceutical 53±19±0 ..................... MITOTANE. 53±31±6 ..................... MEDIBAZINE. Pharmaceutical Appendix to the N/A ............................. ACTAGARDIN. 53±33±8 ..................... PARAMETHASONE. Harmonized Tariff Schedule of the N/A ............................. ARDACIN. 53±34±9 ..................... FLUPREDNISOLONE. N/A ............................. BICIROMAB. 53±39±4 ..................... OXANDROLONE. United States of America in Chemical N/A ............................. CELUCLORAL. 53±43±0 -

2 12/ 35 74Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 22 March 2012 (22.03.2012) 2 12/ 35 74 Al (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every A61K 9/16 (2006.01) A61K 9/51 (2006.01) kind of national protection available): AE, AG, AL, AM, A61K 9/14 (2006.01) AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, (21) International Application Number: DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, PCT/EP201 1/065959 HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, (22) International Filing Date: KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, 14 September 201 1 (14.09.201 1) ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PE, PG, PH, PL, PT, QA, RO, RS, RU, (25) Filing Language: English RW, SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, (26) Publication Language: English TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: 61/382,653 14 September 2010 (14.09.2010) US (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (71) Applicant (for all designated States except US): GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, NANOLOGICA AB [SE/SE]; P.O Box 8182, S-104 20 ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, Stockholm (SE). -

Bulk Drug Substances Nominated for Use in Compounding Under Section 503B of the Federal Food, Drug, and Cosmetic Act
Updated June 07, 2021 Bulk Drug Substances Nominated for Use in Compounding Under Section 503B of the Federal Food, Drug, and Cosmetic Act Three categories of bulk drug substances: • Category 1: Bulk Drug Substances Under Evaluation • Category 2: Bulk Drug Substances that Raise Significant Safety Risks • Category 3: Bulk Drug Substances Nominated Without Adequate Support Updates to Categories of Substances Nominated for the 503B Bulk Drug Substances List1 • Add the following entry to category 2 due to serious safety concerns of mutagenicity, cytotoxicity, and possible carcinogenicity when quinacrine hydrochloride is used for intrauterine administration for non- surgical female sterilization: 2,3 o Quinacrine Hydrochloride for intrauterine administration • Revision to category 1 for clarity: o Modify the entry for “Quinacrine Hydrochloride” to “Quinacrine Hydrochloride (except for intrauterine administration).” • Revision to category 1 to correct a substance name error: o Correct the error in the substance name “DHEA (dehydroepiandosterone)” to “DHEA (dehydroepiandrosterone).” 1 For the purposes of the substance names in the categories, hydrated forms of the substance are included in the scope of the substance name. 2 Quinacrine HCl was previously reviewed in 2016 as part of FDA’s consideration of this bulk drug substance for inclusion on the 503A Bulks List. As part of this review, the Division of Bone, Reproductive and Urologic Products (DBRUP), now the Division of Urology, Obstetrics and Gynecology (DUOG), evaluated the nomination of quinacrine for intrauterine administration for non-surgical female sterilization and recommended that quinacrine should not be included on the 503A Bulks List for this use. This recommendation was based on the lack of information on efficacy comparable to other available methods of female sterilization and serious safety concerns of mutagenicity, cytotoxicity and possible carcinogenicity in use of quinacrine for this indication and route of administration. -

209089Orig1s000 209090Orig1s000
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 209089Orig1s000 209090Orig1s000 CLINICAL REVIEW(S) CLINICAL REVIEW Application Type NDA (Rx to OTC switch) Application Number(s) 209-089 Xyzal Allergy 24 HR tablets 209-090 Xyzal Allergy 24 HR solution Priority or Standard Standard Submit Date(s) March 18, 2016 Received Date(s) March 31, 2016 PDUFA Goal Date January 31, 2017 Division / Office DPARP/ODE 2/OND Reviewer Name(s) Xu Wang, M.D., Ph.D. Review Completion Date December 9, 2016 Established Name Levocetirizine dihydrochloride Rx Trade Name Xyzal tablets/solution OTC Trade Name Xyzal Allergy 24HR tablets/solution Therapeutic Class Antihistamine Applicant UCB Inc. Formulation(s) tablets/solution Dosing Regimen ≥12 years: One tablet (5 mg) daily/10 mL solution (5 mg) daily; 1 6 – 11 years: /2 tablet (2.5 mg) daily/5 mL solution (2.5 mg) daily; 2 – 5 years: 2.5 mL solution (1.25 mg) daily Indication(s) Temporarily relieve these symptoms due to hay fever or other upper respiratory allergies: • running nose, • sneezing, • itchy, watering eyes, • itching of nose or throat Intended Population(s) Tablets: ≥6 years of age Solution: ≥2 years of age Template Version: March 6, 2009 Reference ID: 4025819 Clinical Review Xu Wang, M.D., Ph.D. NDA 209-089/NDA 209-090 Rx to OTC switch Xyzal Allergy 24HR (levocetirizine dihydrochloride) tablets/solution Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT .........................................4 1.1 Recommendation on Regulatory Action ............................................................. 4 1.2 Risk Benefit Assessment .................................................................................... 4 2 INTRODUCTION AND REGULATORY BACKGROUND ........................................ 4 2.1 Product Information ............................................................................................ 4 2.2 Currently Available Treatments for Proposed Indications.................................. -

Medications Which Must Be Withheld Prior to Skin Testing
Medications Which Must Be Withheld Prior to Skin Testing All the medications listed below must be held at least 48 hours prior to skin testing, and the medications denoted by a * or + must be held longer than 48 hours. The majority of these medications are antihistamines used to treat allergy symptoms or hives. However, the sleep aids and medications used to prevent dizziness or nausea also have antihistaminic effects. If you are taking a tricyclic antidepressant, consult with your doctor before stopping it. These medications are listed by brand name, with the (generic name) in parentheses. Actifed (triprolidine HCL) Disophrol Polaramine, Polargen Alka-Seltzer Plus Cold (chlorpheniramine) Dorcol (dexchlorpheniramine) *Allegra (fexofenadine) Dristan Pyribenzamine Allerest (chlorpheniramine) Drixoral Pyrroxate (chlorpheniramine) *Alavert (loratadine) Extendryl Rhinex *Atarax,Vistaril (hydroxyzine) Fedahist (chlorpheniramine) Rondec (carbinoxamine) ARM Hispril (diphenylpyramine) Rynatan (chlorpheniramine/ Atrohist Histabid pyrliamine) BENADRYL (diphenhydramine) Histadyl Rynatuss Benylin (diphenhydramine) Histaspan Sine-Aid Bromfed (bromphenaramine) Isoclor Sinutab Chlor-Trimeton (chlorpheniramine) Kronafed-A Sinutin (chlorpheniramine) *Claritin (loratadine) Naldecon (phenytolozamine) Sudafed-Plus (chlorpheniramine) *Clarinex (desloratadine) Napril Tacaryl (methdilazine) Clistin (carbinoxamine) Nolahist (phenindamine) Tamine Comhist Nolamine (chlorpheniramine) Tavist (clemastine) Congesprin Novafed A Teldrin Contac Novahistine Temaril (trimeprazine) -

Medicines That Adversely Affect Allergy Skin Testing
1 OSU Department of Otolaryngology–Head and Neck Surgery Division of Sinus and Allergy at the OSU Eye and Ear Institute 915 Olentangy River Rd. 4th Floor Columbus, Oh 43212 614-366-ENTS (3687) Medicines that affect allergy skin testing: Do not take these medications 5 days or longer (as listed below), prior to your appointment: *This is not an all-inclusive list, review all the ingredients on the package or ask your doctor or pharmacist.* Antihistamines - First Generation Generic Name Brand Names Azatadine Optimine Bromphenarimine BroveX, Dimetane, Lodrane Carbinoxamine Maleate Histex Pd, Palgic and Pediatex AHIST, Aller-Chlor, C.P.M, Chlo-Amine, Chlor-Allergy, Chlor-Mal, ChlorTrimeton, Chlorphen, Chlorpheniramine (6 days) Effidac-24, Histex, Ridraman Clemastine (10 days) Allerhist-1, Contact 12hr Allergy, Tavist-1 Cyproheptadine (11 days) Periactin Dexchlorpheniramine Polaramine Actifed Sinus Day, Aler-Tab, Allergy, AllergySinus, Allermax, Aler-Dryl, Altaryl, Banophren, Benadryl, Calm-Aid, Children's Allergy, Compoz Nighttime, Diphedryl, Diphen-Allergy, Diphenhist, Dormin Sleep Aide, Dytan, Dytuss, Genahist, Hydramine, Ibuprofen PM, Nu-Med, Nytol, PediaCare Diphenhydramine Children's Allergy, Q-Dryl, Quenalin, Scot-Tussin Allergy, Siladryl, Silphen, Simply Allergy, Simply Sleep, Sleep-ettes, Sleep Formula, Sleepinal, Sominex, Tavist, Theraflu, Triaminic, Twilite, Tylenol PM, Unisom Sleep Gels, Valu-Dryl Dimenhydrinate Dramamine Hydroxyzine (8 days) Atarax, Rezine, Vistaril Ketotifen Zatiden Meclizine HCl Antivert, Bonine Methdilazine -

Decongestants, Antihistamines and Nasal Irrigation for Acute Sinusitis in Children (Review)
Decongestants, antihistamines and nasal irrigation for acute sinusitis in children (Review) Shaikh N, Wald ER This is a reprint of a Cochrane review, prepared and maintained by The Cochrane Collaboration and published in The Cochrane Library 2014, Issue 10 http://www.thecochranelibrary.com Decongestants, antihistamines and nasal irrigation for acute sinusitis in children (Review) Copyright © 2014 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd. TABLE OF CONTENTS HEADER....................................... 1 ABSTRACT ...................................... 1 PLAINLANGUAGESUMMARY . 2 BACKGROUND .................................... 2 OBJECTIVES ..................................... 3 METHODS ...................................... 3 RESULTS....................................... 5 Figure1. ..................................... 6 DISCUSSION ..................................... 8 AUTHORS’CONCLUSIONS . 8 ACKNOWLEDGEMENTS . 8 REFERENCES ..................................... 8 CHARACTERISTICSOFSTUDIES . 12 DATAANDANALYSES. 14 APPENDICES ..................................... 14 WHAT’SNEW..................................... 17 HISTORY....................................... 17 CONTRIBUTIONSOFAUTHORS . 17 DECLARATIONSOFINTEREST . 17 SOURCESOFSUPPORT . 17 DIFFERENCES BETWEEN PROTOCOL AND REVIEW . .... 18 INDEXTERMS .................................... 18 Decongestants, antihistamines and nasal irrigation for acute sinusitis in children (Review) i Copyright © 2014 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd. [Intervention -

Final1-Aritmo-Final-Report-V2-0Final.Pdf
ARITMO Final Report PROJECT FINAL REPORT Grant Agreement number: 241679 Project acronym: ARITMO Project title: Arrhythmogenic potential of drugs Funding Scheme: Small or Medium-Scale Focused Research Project Period covered: from 1st January 2010 to 30th June 2013 Name of the scientific representative of the project's co-ordinator, Title and Organisation: Prof. Miriam CJM Sturkenboom, Erasmus Universitair Medisch Centrum Rotterdam Tel: +31 10 704 4126 Fax: +31 10 704 4722 E-mail: [email protected] Project website1 address: www.aritmo-project.org 1 The home page of the website should contain the generic European flag and the FP7 logo which are available in electronic format at the Europa website (logo of the European flag: http://europa.eu/abc/symbols/emblem/index_en.htm ; logo of the 7th FP: http://ec.europa.eu/research/fp7/index_en.cfm?pg=logos). The area of activity of the project should also be mentioned. © Copyright 2013 ARITMO Consortium 1 ARITMO Final Report Table of contents Table of contents ................................................................................................................................................................. 2 1. Final publishable summary report ................................................................................................................................ 3 1.1 Executive summary ................................................................................................................................................. 3 1.2 Description of project context and