Food and Drug Administration, HHS § 352.20
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Thesis Entitled Evaluating UVB and UVA Boosting Technologies For
A Thesis entitled Evaluating UVB and UVA Boosting Technologies for Chemical and Physical Sunscreens by An Ngoc Hiep Huynh Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Master of Science Degree in Pharmaceutical Sciences Industrial Pharmacy ___________________________________________ Gabriella Baki, Ph.D., Committee Chair ___________________________________________ Jerry Nesamony, Ph.D., Committee Member ___________________________________________ Matthew W. Liberatore, Ph.D., Committee Member ___________________________________________ Dr. Amanda C. Bryant-Friedrich, Dean College of Graduate Studies The University of Toledo May 2020 Copyright 2020 An Ngoc Hiep Huynh This document is copyrighted material. Under copyright law, no parts of this document may be reproduced without the expressed permission of the author. An Abstract of Evaluating UVB and UVA Boosting Technologies for Chemical and Physical Sunscreens by An Ngoc Hiep Huynh Submitted to the Graduate Faculty as partial fulfillment of the requirements for the Master of Science Degree in Pharmaceutical Sciences Industrial Pharmacy The University of Toledo May 2020 There are currently 14 organic and 2 inorganic UV filters approved in the United States. Due to coral reef safety concerns, octinoxate and oxybenzone have been banned in Hawaii, Key West, FL and the US Virgin Islands; and octocrylene is also being studied for its potential impact on coral reef safety, leaving 11 organic UV filters as viable options for sunscreen manufacturers – with limitations on their combination. Since consumers are always looking for sunscreens with high SPF and broad-spectrum protection, the need for UVB and UVA protection boosting technologies is greater than ever. In a preliminary study, about two dozen emollients were scanned for their SPF boosting capability with selected organic UV filters. -

Octinoxate, Octisalate, Avobenzone, Ensulizole, Homosalate
TONYMOLY MAGIC FOOD MANGO MILD SUN BLOCK- octinoxate, octisalate, avobenzone, ensulizole, homosalate cream TONYMOLY CO.,LTD Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- ACTIVE INGREDIENT Active ingredients: Ethylhexyl Methoxycinnamate 6.9%, Ethylhexyl Salicylate 4.5%, Butyl Methoxydibenzoylmethane 3.5%, Phenylbenzimidazole Sulfonic Acid 3.5%, Homosalate 3.0% INACTIVE INGREDIENT Inactive ingredients: Water, Butylene Glycol, Alcohol Denat., Octocrylene, Phenethyl Benzoate, Aminomethyl Propanol, Bis-Ethylhexyloxyphenol Methoxyphenyl Triazine, Triceteareth-4 Phosphate, Glycol Stearate, Ammonium Acryloyldimethyltaurate/VP Copolymer, Carbomer, PEG-2 Stearate, Fragrance(Parfum), Phenoxyethanol, Glyceryl Caprylate, Caprylyl Glycol, Mangifera Indica (Mango) Seed Butter, Disodium EDTA, Citrus Limon (Lemon) Fruit Extract, Musa Sapientum (Banana) Fruit Extract, Propylene Glycol, 1,2-Hexanediol, Citrus Aurantium Dulcis (Orange) Fruit Extract, Mangifera Indica (Mango) Fruit Extract, Psidium Guajava Fruit Extract, Citrus Paradisi (Grapefruit) Fruit Extract, Cocos Nucifera (Coconut) Fruit Extract, Actinidia Chinensis (Kiwi) Fruit Extract, Carica Papaya (Papaya) Fruit Extract, Ethylhexylglycerin PURPOSE Purpose: Sunscreen WARNINGS Warnings: For external use only Do not use on damaged or broken skin Stop use and ask a doctor if irritation occurs Keep out of reach of children DESCRIPTION Uses: - helps prevent sunburn - If used as directed with other sun protection measures Directions: decreases the risk of skin cancer and early skin aging caused by the sun Directions: For sunscreen use: - apply liberally 15 minutes before sun exposure - use a water resistant sunscreen if swimming or sweating - reapply at least every 2 hours - Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. -
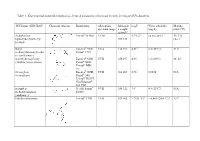
Table 1 - Experimental and Predicted Physical-Chemical Parameters of the Most Recently Investigated UV-Absorbers
Table 1 - Experimental and predicted physical-chemical parameters of the most recently investigated UV-absorbers. INCI name (INN/XAN) Chemical structure Brand name Absorption Molecula LogP Water solubility Melting spectrum range r weight (mg/L) point (°C) (g/mol)4 diethylamino Uvinul® A Plus UVA1 5.7-6.21 <0.01 (20°C) 1 54; 314 hydroxybenzoyl hexyl 397.515 (dec.) 1 benzoate Butyl Eusolex® 9020, UVA 310.393 4.514 2.2 (25°C)4 83.54 methoxydibenzoylmetha Parsol® 1789 ne (avobenzone) 4-methylbenzylidene Eusolex® 6300 UVB 258.397 4.95 1.3 (20°C) 66–68 camphor (enzacamene) Parsol® 5000 Uvinul® MBC 95 Octocrylene Eusolex® OCR, UVB 361.485 6.783 0.00383 N/A (octocrilene) Parsol® 340, Uvinul® N539T, NeoHeliopan® 303 USP isoamyl p- Neo Heliopan® UVB 248.322 3.61 4.9 (25°C)1 N/A methoxycinnamate E1000 (amiloxate) Ethylhexyl triazone Uvinul® T150 UVB 823.092 > 7(20 °C) 6 < 0.001 (20.0 °C) 6 1296 Ethylhexyl Parsol® MCX, UVB 290.403 6.14 0.041 (24 °C and N/A methoxycinnamate Heliopan® New pH 7.1) 4 (octinoxate) Ethylhexyl dimethyl Escalol™ 507 UVB 277.4084 5.774 0.54 (25 °C) 4 N/A PABA (padimate-O) Arlatone 507 Eusolex 6007 benzophenone-3 Eusolex® 4360 UVA2+ UVB 228.247 3.72 3.7 (20°C) 2 62-652 (oxybenzone) bis-ethylhexyloxyphenol Tinosorb® S UVA1+UVB 627.826 12.61 <10-4 80.401 methoxyphenol triazine (bemotrizinol) Phenylbenzimidazole Eusolex® 232 UVA2+ UVB 274.2945 -1.1 (pH 5) > 30% (As sodium N/A sulfonic acid Parsol® HS -2.1 (pH 8)5 or (ensulizole) Neo Heliopan® triethanolammoniu Hydro m salt at 20°C) 5 1 (3) 2 (34) 3 (44) 4 Pubchem 5 SCCP/1056/06 Opinion on phenylbenzimidazole sulfonic acid and its salts 6 BASF safety data sheet Table 2 – In vitro studies for the assessment of skin permeation/penetration of sunscreens. -

Sun Protection, Sunscreens and Vitamin D
SunSun protection,protection, sunscreenssunscreens andand VitaminVitamin DD GPGP NationalNational ConferenceConference RotoruaRotorua EnergyEnergy EventsEvents CentreCentre JuneJune 20092009 Dr. Louise Reiche Dermatologist New Zealand Dermatological Society Incorporated MelanomaMelanoma SkinSkin cancercancer andand sunlightsunlight Exposure to UVR causes > 90% of skin cancers Skin cancer is commonest cancer in NZ >50,000 new cases per year ~300 deaths per year ~$33.4 NZ million per year International Agency for Research on Cancer. IARC Monographs on the evaluation of carcinogenic risks to humans. Solar ultraviolet radiation. Lyon: International Agency for Research on Cancer, 1992. Armstrong BK. How sun exposure causes skin cancer. In: Hill D, Elwood JM, English DR, Eds. Prevention of Skin Cancer. Dordrecht: Kluwer Academic Publishers, 2004. O’Dea D. The Costs of Skin Cancer to New Zealand. Wellington: Cancer Society of New Zealand, 2000. New Zealand Health Information Service. Cancer, New Registrations and Deaths. Wellington: New Zealand Health Information Service, 2004. MelanomaMelanoma 1842 new cases in 2002 328 directly attributable to severe sunburn (Sneyd and Cox 2006) Authors recommended, “to reduce burden of melanoma in NZ, need to prevent excessive sun exposure and (facilitate) early diagnosis” Whilst cancer overall is rare in adolescence, melanoma was commonest cancer MelanomaMelanoma NZ incidence and death rate among world highest 56.2/100,000 in European population of Auckland highest reported worldwide men -

A Bottom-Up Approach to Photoprotection
Journal of Photochemistry and Photobiology A: Chemistry 353 (2018) 376–384 Contents lists available at ScienceDirect Journal of Photochemistry and Photobiology A: Chemistry journal homepage: www.elsevier.com/locate/jphotochem Photophysics of the sunscreen ingredient menthyl anthranilate and its precursor methyl anthranilate: A bottom-up approach to photoprotection a a a a,b c N.D.N. Rodrigues , N.C. Cole-Filipiak , M.D. Horbury , M. Staniforth , T.N.V. Karsili , d a, Y. Peperstraete , V.G. Stavros * a University of Warwick, Department of Chemistry, Coventry, CV4 7AL, UK b University of Warwick, Department of Physics, Coventry, CV4 7AL, UK c Temple University, Department of Chemistry, Philadelphia, PA 19122, USA d Synchrotron SOLEIL, AILES Beamline, L’Orme des Merisiers, Saint Aubin, BP48, 91192 Gif Sur Yvette Cedex, France A R T I C L E I N F O A B S T R A C T Article history: Received 3 October 2017 The ultrafast excited state dynamics of the sunscreen ingredient menthyl anthranilate (MenA) and its Received in revised form 22 November 2017 precursor methyl anthranilate (MA) were studied in vacuum (using time-resolved ion yield spectroscopy) Accepted 23 November 2017 and in solution (using transient electronic absorption spectroscopy). MenA and MA both show long-lived Available online 1 December 2017 dynamics, with the observation of a kinetic isotope effect suggesting that hydrogen motion acts as the rate determining process in the overall decay. Complementary computational studies exploring the Keywords: intuitive decay pathways of MA revealed a bound S1 state with a shallow ‘up-hill’ gradient with respect to Photochemistry proton transfer. -

Sunscreen Faqs
Sunscreen FAQs What type of sunscreen should I use? There are two main types of sunscreen ingredients: physical sun blockers and chemical sunscreens. Physical sunscreens prevent ultraviolet light from reaching your skin, and contain either zinc oxide or titanium dioxide. Physical sunscreens protect against UVA and UVB damage. Physical sunscreens are preferred for patients with sensitive skin or children. Chemical sunscreens rely on an interaction between the sun and the chemical to protect your skin. Examples of such chemicals are avobenzone, octinoxate, homosalate, padimate O, and many others. Chemical sunscreens can protect against UVA, UVB, or both types of damage, depending on which ingredient is used. Read the label to ensure coverage for both UVA and UVB exposure. There are new ingredients being developed that take chemical sunscreens and stabilize them to prolong their activity (i.e., Helioplex, Mexoryl). Does the SPF really matter? In laboratory testing, there are minor differences between SPF 15 and SPF 30 or greater products. However, that data is based on using 1 oz. (30 gm.) of sunscreen for each full body application (the size of a shot glass). Most people use far less in real-life settings. For that reason, we recommend an SPF of at least 30. It is common to find good sunscreens with SPF ranging from 45-70. To maintain protection, reapply sunscreen every 1-3 hours, depending on sweating, water exposure, and sun intensity. Is sunscreen alone enough to protect my skin in the sun? In some cases, yes. However, if you are in the sun for longer periods on a regular basis (such as gardening, golfing, boating, sports), it may be better to add sun protective clothing or habits. -

FDA Proposes Sunscreen Regulation Changes February 2019
FDA Proposes Sunscreen Regulation Changes February 2019 The U.S. Food and Drug Administration (FDA) regulates sunscreens to ensure they meet safety and eectiveness standards. To improve the quality, safety, and eectiveness of sunscreens, FDA issued a proposed rule that describes updated proposed requirements for sunscreens. Given the recognized public health benets of sunscreen use, Americans should continue to use broad spectrum sunscreen with SPF 15 or higher with other sun protective measures as this important rulemaking eort moves forward. Highlights of FDA’s Proposals Sunscreen active ingredient safety and eectiveness Two ingredients (zinc oxide and titanium dioxide) are proposed to be safe and eective for sunscreen use and two (aminobenzoic acid (PABA) and trolamine salicylate) are 1 proposed as not safe and eective for sunscreen use. FDA proposes that it needs more safety information for the remaining 12 sunscreen ingredients (cinoxate, dioxybenzone, ensulizole, homosalate, meradimate, octinoxate, octisalate, octocrylene, padimate O, sulisobenzone, oxybenzone, avobenzone). New proposed sun protection factor Sunscreen dosage forms (SPF) and broad spectrum Sunscreen sprays, oils, lotions, creams, gels, butters, pastes, ointments, and sticks are requirements 2 proposed as safe and eective. FDA 3 • Raise the maximum proposed labeled SPF proposes that it needs more data for from SPF 50+ to SPF 60+ sunscreen powders. • Require any sunscreen SPF 15 or higher to be broad spectrum • Require for all broad spectrum products SPF 15 and above, as SPF increases, broad spectrum protection increases New proposed label requirements • Include alphabetical listing of active ingredients on the front panel • Require sunscreens with SPF below 15 to include “See Skin Cancer/Skin Aging alert” on the front panel 4 • Require font and placement changes to ensure SPF, broad spectrum, and water resistance statements stand out Sunscreen-insect repellent combination 5 products proposed not safe and eective www.fda.gov. -

Kroger Co Disclaimer: Most OTC Drugs Are Not Reviewed and Approved by FDA, However They May Be Marketed If They Comply with Applicable Regulations and Policies
SUNSCREEN- avobenzone, homosalate, octisalate, octocrylene, oxybenzone lotion The Kroger Co Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies. ---------- Kroger 711 Active Ingredients Avobenzone 3% Homosalate 13% Octisalate 5% Octocrylene 7% Oxybenzone 4% Purpose Sunscreen Use helps prevent sunburn if used as directed with other sun protections measures (see Directions), decreases the rist of skin cancer and early skin aging caused by the sun Warnings For external use only Do not use on damaged or broken skin When using this product keep out of eyes. Rinse with water to remove Stop use and ask a doctor if rash occurs Keep out of reach of children if swallowed, get medical help or contact a Poison Control Center right away Directions apply liberally 15 minutes before sun exposure reapply: after 80 minutes of swimming or sweating immediately after towel drying at least every 2 hours Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk use a reqularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including: limit time in the sun, especially from 10 a.m. - 2 p.m. wear long-sleeved shirts, pants, hats and sunglasses children under 6 months of age: Ask a doctor Other Information Protect the product from excessive heat and direct sun Inactive ingredients water, sorbitol, triethanolamine, VP/eicosene copolymer, stearic acid, sorbitan isostearate, aluminum starch octenylsuccinate, benzyl alcohol, dimethicone, tocopheryl, chlorphenesin, polyglyceryl-3 distearate, fragrance, carbomer, disodium EDTA Questions or comments? 1-800-632-6900 Adverse Reaction DISTRIBUTED BY THE KROGER CO. -

Chemical UVR Absorbers
Chemical UVR Absorbers The names given in bold and used Diisopropyl methyl cinnamate Glyceryl ethyihexanoate dimethoxy- throughout this handbook are those of Empirical formula: cinnamate the International Nomenclature of C 6H22O2 Chemical names. Cosmetic Ingredients. Glyceryl octanoate dimethoxycinnamate; Chemical names: 2-propenoic acid, 3-(4-methoxyphenyl)-, 2-Propenoic acid, 3-12,4bis(1 diester with 1 ,3-dihydroxy-2-(2-ethyl-1 - methylethyphenyl-methyl ester; 2,5- oxohexyl)oxypropane diisopropyl methyl cinnamate _ lsoamyl-para-methoxycinnamate Ethyihexyl methoxycinnamate Empirical formula: Empirical formula: C151-12003 C 8H26O3 Chemical names: Cinnamates Chemical names: Amyl4-methoxycinnamate; isopentyl-4- 2-Ethylhexyl-4-methoxycin nam ate; methoxycinnamate; isopenlyl-para- Cinoxate 2-ethyl-hexyl-para-methoxycinnamate; methoxy-cinnamate; 3-(4-methoxyphenyl)- Empirical formula: para-methoxycinnamic acid, 2-ethylhexyl 2-propenoic acid, isopentyl ester Ci4HieO4 ester; 3-(4-methoxyphenyl)-2-propenoic acid, 2-ethylhexyl ester; octinoxate; octyl Trade names: Chemical names: methoxycinnamate; 2-propenoic acid, 3- Neo Heliopan type E 1000; Solarum AMC 2- Ethoxyothyl-para-methoxyci n nam ate; (4-methoxyphenyl)-2-ethylhexyl ester 2-propenoic acid, 3-(4-methoxyphery- para-A minobenzoic acids (PA BAs) 2-ethoxyethyl ester; 2-ethoxyethyl-4- Trade names: methoxycinnamate AEC Octyl Methoxycinnamate; Escalol Amyl dimethyl FABA 557; Eusolex 2292; Heliosol 3; Empirical formula: Trade names: Jeescreen OMC; Katoscreen OMC; Nec C14H21 NO2 Giv Tan F; Phiasol -

WO 2013/036901 A2 14 March 2013 (14.03.2013) P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2013/036901 A2 14 March 2013 (14.03.2013) P O P C T (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every A61K 8/30 (2006.01) kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, (21) International Application Number: BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, PCT/US2012/054376 DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (22) International Filing Date: HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, 10 September 2012 (10.09.2012) KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, (25) Filing Language: English NO, NZ, OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, (26) Publication Language: English RW, SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, (30) Priority Data: ZM, ZW. 61/532,701 9 September 201 1 (09.09.201 1) US (84) Designated States (unless otherwise indicated, for every (71) Applicant (for all designated States except US): UNIVER¬ kind of regional protection available): ARIPO (BW, GH, SITY OF FLORIDA RESEARCH FOUNDATION, GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, SZ, TZ, INC. -

Sunscreen Alternatives Sunscreen Alternatives
Sunscreen Alternatives We all know that the sun can be damaging in many ways, but most sunscreens on the market have some questionable, if not dangerous, ingredients. Here are a few ways to play it safe in the sun without putting your health at risk. 1. Common Sense We all know it but often forget: Avoid midday (10 am-2 pm) sun as much as possible, stay in the shade when possible, wear sunglasses and sun hats, don’t stay out exposed for too long. 2. Cover Up! For short periods, especially early or late in the day, sunscreen may not be necessary. In fact, a little exposure every day is vital to keep our Vitamin D levels up! Instead cover the skin with lightweight, tightly woven clothing, and protect your face with a hat. Babies under 6 months especially need to be covered up, since they don’t yet have the protection of melanin. 3. Be a Label Reader Not all sunscreens are created equal. The “worst” chemicals to avoid include Oxybenzone and PABA/Padimate O. Some active ingredients that have been found less toxic include Titanium Dioxide and Zinc Oxide. 4. CaliCaliforniafornia Baby This very eco-friendly company makes three of the top ten sunscreens on Environmental Working Group’s “Low Health Hazard/Low Sun Hazard/Most Effective” list: the SPF 30+ No Fragrance Sunblock Stick, the SPF 30+ Everyday Sunblock Stick, and the SPF 30+ Everyday Lotion. They are widely available and reasonably priced. 5. Homemade There are many recipes out there for DIY sunscreen. Here’s one to try: In a double boiler over medium heat, mix 2 ½ oz sesame oil, 1 ½ oz coconut oil, and 1 ½ oz beeswax. -

Annex VI, Last Update: 02/08/2021
File creation date: 03/10/2021 Annex VI, Last update: 22/09/2021 LIST OF UV FILTERS ALLOWED IN COSMETIC PRODUCTS Substance identification Conditions Wording of Reference Maximum conditions of Product Type, concentration Update date number Chemical name / INN / XAN Name of Common Ingredients Glossary CAS Number EC Number Other use and body parts in ready for use warnings preparation 2 N,N,N-Trimethyl-4-(2-oxoborn-3-ylidenemethyl CAMPHOR BENZALKONIUM 52793-97-2 258-190-8 6% 15/10/2010 ) anilinium methyl sulphate METHOSULFATE 3 Benzoic acid, 2-hydroxy-, HOMOSALATE 118-56-9 204-260-8 10% 02/08/2021 3,3,5-trimethylcyclohexyl ester / Homosalate 4 2-Hydroxy-4-methoxybenzophenone / BENZOPHENONE-3 131-57-7 205-031-5 6% Reg (EU) Not more than Contains 02/08/2021 Oxybenzone 2017/238 of 10 0,5 % to protect Benzophenone-3 February 2017- product (1) date of formulation application from September 2017 6 2-Phenylbenzimidazole-5-sulphonic acid and its PHENYLBENZIMIDAZOLE SULFONIC 27503-81-7 248-502-0 8%(as acid) 08/03/2011 potassium, sodium and triethanolamine salts / ACID Ensulizole 7 3,3'-(1,4-Phenylenedimethylene) bis TEREPHTHALYLIDENE DICAMPHOR 92761-26-7 / 410-960-6 / - 10%(as acid) 26/10/2010 (7,7-dimethyl-2-oxobicyclo-[2.2.1] SULFONIC ACID 90457-82-2 hept-1-ylmethanesulfonic acid) and its salts / Ecamsule 8 1-(4-tert-Butylphenyl)-3-(4-methoxyphenyl) BUTYL 70356-09-1 274-581-6 5% 15/10/2010 propane-1,3-dione / Avobenzone METHOXYDIBENZOYLMETHANE 9 alpha-(2-Oxoborn-3-ylidene)toluene-4-sulphoni BENZYLIDENE CAMPHOR SULFONIC 56039-58-8 - 6%(as acid)