Phosphorus Compounds
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

5. POTENTIAL for HUMAN EXPOSURE 5.1 OVERVIEW White
WHITE PHOSPHORUS 157 5. POTENTIAL FOR HUMAN EXPOSURE 5.1 OVERVIEW White phosphorus can enter the environment from its production, use, accidental spills during loading and unloading for shipment, and accidental spills during transport. Hazardous wastes sites containing white phosphorus can also be a source of phosphorus in the environment. White phosphorus has been found in at least 77 of the 1,430 current or former EPA National Priorities List (NPL) hazardous waste sites (HazDat 1996). However, the number of sites evaluated for white phosphorus is not known. The frequency of these sites within the United States can be seen in Figure 5-l. The persistence of elemental phosphorus in the air is very short due to oxidation to phosphorus oxides and ultimately to phosphorus acids. However, the particulate phosphorus aerosol may be coated with a protective oxide layer that may prevent further oxidation and extend the lifetime of particulate phosphorus in air. Both wet and dry deposition remove unreacted elemental phosphorus and the degradation products from the air. Similarly, elemental phosphorus oxidizes and hydrolyzes in water and in soil. A small amount of elemental phosphorus is lost from soil and water by volatilization. Phosphorus is used as a fumigant in the storage of grain. Because of ease of application, pellets of aluminum or magnesium phosphide are commonly used (Garry et al. 1993). Phosphine, a highly toxic gas, is generated from phosphide. The rate of formation of phosphine (permissible exposure limit [PEL], 0.4 mg/m3) is dependent on the ambient temperature and humidity. Its release is rapid, and it is extremely fatal to the unprotected person (Garry et al. -

Rational Synthesis of P-Type Zinc Oxide Nanowire Arrays Using Simple Chemical Vapor Deposition
NANO LETTERS 2007 Rational Synthesis of p-Type Zinc Oxide Vol. 7, No. 2 Nanowire Arrays Using Simple Chemical 323-328 Vapor Deposition Bin Xiang,† Pengwei Wang,‡ Xingzheng Zhang,‡ Shadi. A. Dayeh,† David P. R. Aplin,† Cesare Soci,† Dapeng Yu,‡ and Deli Wang*,† Department of Electrical and Computer Engineering, UniVersity of California at San Diego, La Jolla, California 92093-0407, and Electron Microscopy Laboratory, School of Physics, Peking UniVersity, Beijing 100871, China Received October 13, 2006; Revised Manuscript Received December 8, 2006 ABSTRACT We report, for the first time, the synthesis of the high-quality p-type ZnO NWs using a simple chemical vapor deposition method, where phosphorus pentoxide has been used as the dopant source. Single-crystal phosphorus doped ZnO NWs have their growth axis along the 〈001〉 direction and form perfect vertical arrays on a-sapphire. P-type doping was confirmed by photoluminescence measurements at various temperatures and by studying the electrical transport in single NWs field-effect transistors. Comparisons of the low-temperature PL of unintentionally doped ZnO (n-type), as-grown phosphorus-doped ZnO, and annealed phosphorus-doped ZnO NWs show clear differences related to the presence of intragap donor and acceptor states. The electrical transport measurements of phosphorus-doped NW FETs indicate a transition from n-type to p-type conduction upon annealing at high temperature, in good agreement with the PL results. The synthesis of p-type ZnO NWs enables novel complementary ZnO NW devices and opens up enormous opportunities for nanoscale electronics, optoelectronics, and medicines. Zinc oxide (ZnO) is a wide direct band gap semiconductor synthesis of the high-quality p-type ZnO NWs using a simple (Eg ) 3.4 eV) that displays unique features such as large chemical vapor deposition (CVD) method using phosphorus 1 exciton binding energy (Eb ) 60 meV) and large piezo- pentoxide (P2O5) as the dopant source. -

Ereztech LLC P3553 Safety Data Sheet
EREZTECH LLC 11555 Medlock Bridge Road, Suite 100, Johns Creek, GA 30097, USA T: +1.888.658.1221 F: 1.678.619.2020 E: [email protected] W: https://ereztech.com Section 1. Identification Product Name: Phosphorus trifluoride Product Type: Gas CAS Number: 7783-55-3 Product Number: P3553 Product Manufacturer: Ereztech LLC 11555 Medlock Bridge Road, Suite 100 Johns Creek, GA 30097 Product Information: (888) 658-1221 In Case of an Emergency: CHEMTREC: 1-800-424-9300 (USA); +1 703-527-3887 (International); CCN836180 *** Contact manufacturer for all non-emergency calls. Section 2. Hazards Identification Appearance/Odor: Colorless, odorless gas. Classification: ACUTE TOXICITY, ORAL - Category 4, H302 ACUTE TOXICITY, DERMAL - Category 4, H312 SKIN CORROSION/IRRITATION - Category 1B, H314 SERIOUS EYE DAMAGE/EYE IRRITATION - Category 2, H318 ACUTE TOXICITY, INHALATION - Category 1, H330 SPECIFIC TARGET ORGAN TOXICITY, SINGLE EXPOSURE; RESPIRATORY TRACT IRRITATION – Category 3, H335 GHS Label Elements Hazard Pictograms: Signal Word: DANGER Hazard Statements: H302: Harmful if swallowed. H312: Harmful in contact with skin. H314: Causes severe skin burns and eye damage. H318: Causes serious eye damage. H330: Fatal if inhaled. H335: May cause respiratory irritation. Ereztech P3553 Page 1 of 13 Revision: 1.00 Date of Issue: 11/27/2020 Phosphorus trifluoride Safety Data Sheet Section 2. Hazards Identification Precautionary Statements Prevention: P260: Do not breathe fumes/gases/mists/vapors/sprays. P264: Wash skin thoroughly after handling. P270: Do not eat, drink or smoke when using this product. P271: Use only outdoors or in a well-ventilated area. P280: Wear protective gloves/ protective clothing/ eye protection/ face protection. -

(12) Patent Application Publication (10) Pub. No.: US 2005/0044778A1 Orr (43) Pub
US 20050044778A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2005/0044778A1 Orr (43) Pub. Date: Mar. 3, 2005 (54) FUEL COMPOSITIONS EMPLOYING Publication Classification CATALYST COMBUSTION STRUCTURE (51) Int. CI.' ........ C10L 1/28; C1OL 1/24; C1OL 1/18; (76) Inventor: William C. Orr, Denver, CO (US) C1OL 1/12; C1OL 1/26 Correspondence Address: (52) U.S. Cl. ................. 44/320; 44/435; 44/378; 44/388; HOGAN & HARTSON LLP 44/385; 44/444; 44/443 ONE TABOR CENTER, SUITE 1500 1200 SEVENTEENTH ST DENVER, CO 80202 (US) (57) ABSTRACT (21) Appl. No.: 10/722,127 Metallic vapor phase fuel compositions relating to a broad (22) Filed: Nov. 24, 2003 Spectrum of pollution reducing, improved combustion per Related U.S. Application Data formance, and enhanced Stability fuel compositions for use in jet, aviation, turbine, diesel, gasoline, and other combus (63) Continuation-in-part of application No. 08/986,891, tion applications include co-combustion agents preferably filed on Dec. 8, 1997, now Pat. No. 6,652,608. including trimethoxymethylsilane. Patent Application Publication Mar. 3, 2005 US 2005/0044778A1 FIGURE 1 CALCULATING BUNSEN BURNER LAMINAR FLAME VELOCITY (LFV) OR BURNING VELOCITY (BV) CONVENTIONAL FLAME LUMINOUS FLAME Method For Calculating Bunsen Burner Laminar Flame Velocity (LHV) or Burning Velocity Requires Inside Laminar Cone Angle (0) and The Gas Velocity (Vg). LFV = A, SIN 2 x VG US 2005/0044778A1 Mar. 3, 2005 FUEL COMPOSITIONS EMPLOYING CATALYST Chart of Elements (CAS version), and mixture, wherein said COMBUSTION STRUCTURE element or derivative compound, is combustible, and option 0001) The present invention is a CIP of my U.S. -

CHEMICAL STORAGE SEGREGATION GUIDELINES Incompatible Chemicals Should Always Be Handled and Stored So That They Do Not Accidentally Come in Contact with Each Other
Laboratory Safety Reminders January 2007 ♦ Mount Holyoke College – Environmental Health and Safety CHEMICAL STORAGE SEGREGATION GUIDELINES Incompatible chemicals should always be handled and stored so that they do not accidentally come in contact with each other. This list is not complete, nor are all compatibilities shown. These materials can react to produce excessive heat, harmful vapors, and/or other deadly reactions. Always know the hazards and incompatibilities of a chemical before using it. Chemicals Avoid Accidental Contact With Acetic acid Chromic acid, nitric acid, permanganates, peroxides Hydroxyl-containing compounds such as perchloric acid, Acetic anhydride ethylene glycol Concentrated nitric acid and sulfuric acid mixtures, peroxides (i.e. Acetone peracetic acid solution, hydrogen peroxide) Acetylene Chlorine, bromine, copper, silver, fluorine, mercury Alkali, alkaline earth and strongly electropositive metals (powered Carbon dioxide, carbon tetrachloride and other chlorinated aluminum, magnesium, sodium, hydrocarbons potassium) Mercury, chlorine, calcium hypochlorite, iodine, bromine, hydrogen Ammonia (anhydrous) fluoride Acids, metal powders, flammable liquids, chlorates, nitrates, sulfur, Ammonium nitrate finely divided organics, combustibles Aniline Nitric acid, hydrogen peroxide Arsenical compounds Any reducing agent Azides Acids Ammonia, acetylene, butadiene, butane, other petroleum gases, Bromine sodium carbide, turpentine, benzene, finely divided metals Calcium oxide Water Carbon activated Calcium hypochlorite, other -

Chemical Chemical Hazard and Compatibility Information
Chemical Chemical Hazard and Compatibility Information Acetic Acid HAZARDS & STORAGE: Corrosive and combustible liquid. Serious health hazard. Reacts with oxidizing and alkali materials. Keep above freezing point (62 degrees F) to avoid rupture of carboys and glass containers.. INCOMPATIBILITIES: 2-amino-ethanol, Acetaldehyde, Acetic anhydride, Acids, Alcohol, Amines, 2-Amino-ethanol, Ammonia, Ammonium nitrate, 5-Azidotetrazole, Bases, Bromine pentafluoride, Caustics (strong), Chlorosulfonic acid, Chromic Acid, Chromium trioxide, Chlorine trifluoride, Ethylene imine, Ethylene glycol, Ethylene diamine, Hydrogen cyanide, Hydrogen peroxide, Hydrogen sulfide, Hydroxyl compounds, Ketones, Nitric Acid, Oleum, Oxidizers (strong), P(OCN)3, Perchloric acid, Permanganates, Peroxides, Phenols, Phosphorus isocyanate, Phosphorus trichloride, Potassium hydroxide, Potassium permanganate, Potassium-tert-butoxide, Sodium hydroxide, Sodium peroxide, Sulfuric acid, n-Xylene. Acetone HAZARDS & STORAGE: Store in a cool, dry, well ventilated place. INCOMPATIBILITIES: Acids, Bromine trifluoride, Bromine, Bromoform, Carbon, Chloroform, Chromium oxide, Chromium trioxide, Chromyl chloride, Dioxygen difluoride, Fluorine oxide, Hydrogen peroxide, 2-Methyl-1,2-butadiene, NaOBr, Nitric acid, Nitrosyl chloride, Nitrosyl perchlorate, Nitryl perchlorate, NOCl, Oxidizing materials, Permonosulfuric acid, Peroxomonosulfuric acid, Potassium-tert-butoxide, Sulfur dichloride, Sulfuric acid, thio-Diglycol, Thiotrithiazyl perchlorate, Trichloromelamine, 2,4,6-Trichloro-1,3,5-triazine -
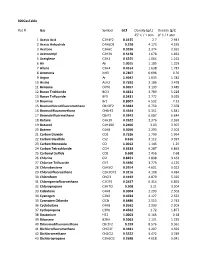
Gas Conversion Factor for 300 Series
300GasTable Rec # Gas Symbol GCF Density (g/L) Density (g/L) 25° C / 1 atm 0° C / 1 atm 1 Acetic Acid C2H4F2 0.4155 2.7 2.947 2 Acetic Anhydride C4H6O3 0.258 4.173 4.555 3 Acetone C3H6O 0.3556 2.374 2.591 4 Acetonitryl C2H3N 0.5178 1.678 1.832 5 Acetylene C2H2 0.6255 1.064 1.162 6 Air Air 1.0015 1.185 1.293 7 Allene C3H4 0.4514 1.638 1.787 8 Ammonia NH3 0.7807 0.696 0.76 9 Argon Ar 1.4047 1.633 1.782 10 Arsine AsH3 0.7592 3.186 3.478 11 Benzene C6H6 0.3057 3.193 3.485 12 Boron Trichloride BCl3 0.4421 4.789 5.228 13 Boron Triflouride BF3 0.5431 2.772 3.025 14 Bromine Br2 0.8007 6.532 7.13 15 Bromochlorodifluoromethane CBrClF2 0.3684 6.759 7.378 16 Bromodifluoromethane CHBrF2 0.4644 5.351 5.841 17 Bromotrifluormethane CBrF3 0.3943 6.087 6.644 18 Butane C4H10 0.2622 2.376 2.593 19 Butanol C4H10O 0.2406 3.03 3.307 20 Butene C4H8 0.3056 2.293 2.503 21 Carbon Dioxide CO2 0.7526 1.799 1.964 22 Carbon Disulfide CS2 0.616 3.112 3.397 23 Carbon Monoxide CO 1.0012 1.145 1.25 24 Carbon Tetrachloride CCl4 0.3333 6.287 6.863 25 Carbonyl Sulfide COS 0.668 2.456 2.68 26 Chlorine Cl2 0.8451 2.898 3.163 27 Chlorine Trifluoride ClF3 0.4496 3.779 4.125 28 Chlorobenzene C6H5Cl 0.2614 4.601 5.022 29 Chlorodifluoroethane C2H3ClF2 0.3216 4.108 4.484 30 Chloroform CHCl3 0.4192 4.879 5.326 31 Chloropentafluoroethane C2ClF5 0.2437 6.314 6.892 32 Chloropropane C3H7Cl 0.308 3.21 3.504 33 Cisbutene C4H8 0.3004 2.293 2.503 34 Cyanogen C2N2 0.4924 2.127 2.322 35 Cyanogen Chloride ClCN 0.6486 2.513 2.743 36 Cyclobutane C4H8 0.3562 2.293 2.503 37 Cyclopropane C3H6 0.4562 -

Stabilisation of Clayey Soil with Lime, Sodium Hydroxide, Aluminium
ISSN 2321 3361 © 2020 IJESC Research Article Volume 10 Issue No.5 Stabilisation of Clayey Soil with Lime, Sodium Hydroxide, Aluminium Oxide and Phosphorus Pentoxide Bhawna Sahay Assistant Professor Noida International University, India Abstract: Soil Stabilisation is a method to improve the geotechnical properties of soil such as bearing capacity, shear strength, plastic limit, liquid limit, etc. it is important before construction of any project whether it is buildings and pavement construction. There are numerous method available for stabilisation of soil but with chemical stabilisation is a quick method. Keywords: Stabilisation, bearing capacity, plastic limit, liquid limit. I. INTRODUCTION with water which results into a powerful compaction and giving higher density for same compaction effort. Clayey soil is originated from weathering and disintegration of rocks. It is generally classified as low compressible soil, medium compressible soil and highly compressible soil. The volume changes and shrinkage is a major problem occur in it. It is generally classified as highly plastic soil which depends on the mineral content of the soil. Stabilisation of soil with chemicals increase the strength of soil and improve the general properties of soil. Lime, sodium hydroxide, aluminium oxide and phosphorus pentoxide reacts vigorously with soil which interacts with the clay particles of soil and increases the resisting capacity of soil. II. CHEMICALS Lime: Calcium oxide or lime is available in white colour powder form, highly reactive in nature, modify all fine grained soil and improves the plasticity, moisture holding capacity, swell and shrinkage behaviour of soil and stability of soil. Aluminium Oxide Aluminium oxide is white coloured shining powder form which reacts with soil to form a sample little bit cool due to its affinity with water. -

Examples of Hazardous Chemicals
Examples of Hazardous Chemicals A-Acids Liquids 1 Acetic acid 64-19-7 2 Acetic anhydride 108-24-7 3 Butyric acid 107-92-6 4 Chromosulfuric acid 14489-25-9 5 Formic acid 64-18-6 6 Hydrobromic acid 10035-10-6 7 Hydrochloric acid 7647-01-0 8 Hydrofluoric acid 7664-39-3 9 Lactic acid 50-21-5 10 Nitric acid 7697-37-2 11 Perchloric acid 7601-90-3 12 Phenol solution (carbolic acid) 108-95-2 13 Phosphoric acid 7664-38-2 14 Propionic acid 79-09-4 15 Sulfuric acid 7664-93-9 16 Trifluoroacetic acid 76-05-1 Solids 17 Benzoic acid 65-85-0 18 Cacodylic acid 75-60-5 19 Maleic acid 110-16-7 20 Oxalic acid 144-62-7 21 Periodic acid 10450-60-9 22 Salicylic acid 69-72-7 23 Tannic acid 1401-55-4 24 Trichloroacetic acid 76-03-9 B-Corrosive Materials Liquids 25 Ammonium hydroxide 1336-21-6 26 Cresol 1319-77-3 27 Hydrazine 302-01-2 28 Morpholine 110-91-8 29 Potassium hydroxide solution 1310-58-3 30 Sodium hydroxide solution 1310-73-2 Solids 31 Aluminum sulfate 10043-01-3 32 Calcium hydroxide 1305-62-0 33 Chromic trioxide 1333-82-0 34 Ferric chloride 7705-08-0 35 Ferrous sulfate 7720-78-7 36 Iodine 7553-56-2 37 Phosphorus pentoxide 1314-56-3 38 Potassium dichromate 7778-50-9 39 Potassium hydroxide 1310-58-3 40 Sodium hydroxide 1310-73-2 C-Cyrogenic Materials 41 Nitrogen, refrigerated liquid 7727-37-9 42 Carbon dioxide, solid 124-38-9 D-Gases 43 Compressed air 44 Germane 7782-65-2 45 Helium 7440-59-7 46 Hydrogen 1333-74-0 47 Mixtures of N2, O2, CO2 48 Oxygen, 100% 7782-44-7 49 Propane 74-98-6 50 Silane 7803-62-5 51 Sulfur dioxide 7446-09-5 E-Flammable Liquids -

Chromic Acid, Nitric Acid, Hydroxyl Compounds, Ethylene Glycol
Acetic acid - Chromic acid, nitric acid, hydroxyl compounds, ethylene glycol, perchloric acid, peroxides, permanganates, ammonium nitrate Acetic anhydride - Hydroxyl-containing compounds such as ethylene glycol, perchloric acid Acetone - Concentrated nitric and sulfuric acid mixtures Acetaldehyde - Acetic acid, acetic anhydride Acetylene - Chlorine, bromine, copper, fluorine, silver, mercury Acrolein - Ammonia(aqueous), any alkali or amine, strong oxidizing agents Alkali and alkaline earth metals (such as powdered aluminum or magnesium, calcium, lithium, sodium, potassium) - Water, carbon tetrachloride or other chlorinated hydrocarbons, carbon dioxide, halogens. Aluminum metal - Ammonium nitrate, antimony trichloride, bromate Ammonia (anhydrous) - Mercury (in manometers, for example), chlorine, calcium hypochlorite, iodine, bromine, hydrofluoric acid (anhydrous) Ammonium nitrate - Acids, powdered metals, flammable liquids, chlorates, nitrites, sulfur, finely divided organic combustible materials Aniline - Nitric acid, hydrogen peroxide, strong acids, oxidizers Arsenic materials - Any reducing agent Azides – Acids Bromine - Ammonia, acetylene, butadiene, butane, methane, propane (or other petroleum gases), hydrogen, sodium carbide, benzene, finely divided metals, turpentine Calcium oxide – Water Carbon (activated) - Calcium hypochlorite, all oxidizing agents Carbon tetrachloride – Sodium Chlorates - Ammonium salts, acids, powdered metals, sulfur, finely divided organic or combustible materials Chromic acid and chromium trioxide - Acetic -

Distribution of Sulfur Dioxide and Phosphorus Pentoxide in an Air Pool Obtained by Correlation and Regression Analysis
JournalJournal of Chemical of Chemical Technology Technology and Metallurgy,and Metallurgy, 54, 4, 54, 2019, 4, 2019 826-830 DISTRIBUTION OF SULFUR DIOXIDE AND PHOSPHORUS PENTOXIDE IN AN AIR POOL OBTAINED BY CORRELATION AND REGRESSION ANALYSIS Mаlik N. Abdikarimov1, Raushan Kh. Turgumbayeva2, Saule S. Sagintayeva1, Rasylbek Mussabekov1, Gulzhan Sh. Ospanova1 1 Almaty University of Power Engineering&Telecommunications, Received 17 May 2017 126, Baytursunov str., Almaty, 050013, Republic of Kazakhstan Accepted 30 May 2018 2 Kazakh National Pedagogical University named after Abai (KazNPU) 050010, Almaty, Dostyk str. 13, Almaty, Republik Kazakhstan E-mail: [email protected] ABSTRACT Results of studying an aerosol of sulfur dioxide аnd phosphorus pentoxide released to the atmosphere by a chemical company processing phosphorites are presented. The effect of wind direction and speed on sulfur dioxide аnd phosphorous pentoxide distribution in a ground layer of the atmosphere is studied. The points of wind direction leading to pollution of the nearby city atmosphere are allocated. The statistical analysis of the environmental pollu- tion is carried out by method of correlation and regression analysis. The dependence of the amount of sulfur dioxide аnd phosphorous pentoxide reaching the atmosphere on the volume released by the enterprise producing them is defined. The results obtained are recommended for environmental control, regulation and management. Keywords: phosphorus oxide, atmosphere, correlation regression analysis, industrial enterprise, sulfur dioxide, phosphorous pentoxide, statistical correlation regression analysis, equation. INTRODUCTION character and intensity of their investigation – on the other. The Republic of Kazakhstan has no powerful A modern chemical industry produces several tens natural factors to counteract the pollution forces. -

Determination of Phosphoric Anhydride in Phosphate Rock, Superphospha Te, and "Meta
U. S. DEPARTMENT OF COMMERCE NATIONAL BUREAU OF STANDARDS RESEARCH PAPER RP1010 Part of Journal of Research of the :National Bureau of Standards, Volume 19, July 1937 DETERMINATION OF PHOSPHORIC ANHYDRIDE IN PHOSPHATE ROCK, SUPERPHOSPHA TE, AND "META. PHOSPHATE" By James I. Hoffman and G. E. F. Lundell ABSTRACT A gravimetric method requiring no molybdate reagent is described for the determination of phosphoric anhydride in phosphate rock and similar materials. In this method a precipitation with a large excess of magnesia mixture in the presence of ammonium citrate serves t o separate the phosphate ion from the other constituents. A second precipitation yields a precipitate of defi nite composition which is converted to magnesium pyrophosphate, Mg2P 20 7, on ignition. The method has been su ccessfully applied to phosphate rock, superphosphate, "meta phosphate", and liquids used with silicate cements in dentistry. It is recom mended for use in standardization and umpire analyses of phosphate rocks and similar materials, but it is not sufficiently rapid to replace the usual volumetric method now employed in routine analyses. CONTENTS Page I. Introduction___ __ _ _ ___ _ _ __ _ _ _ __ _ _ _ _ _ _ _ __ _ __ __ __ __ __ _ _ _ _ _ __ _ __ _ 59 II. Procedure_ ____ ________________ __ ___ ______ _____ __ _________ ____ 60 1. Preparation of the solution ______________________________ _ 60 2. Determination of phosphoric anhydride _ _ _ _ _ _ _ _ _ _ _ __ _ _ _ __ _ 61 III. Results obtained in the use of the method__________________ ____ __ 62 IV.