Method Development for Isotope Analysis of Trace and Ultra-Trace Elements in Environmental Matrices
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Analytical Techniques Used for Elemental Analysis in Various Matrices
Helaluddin et al Tropical Journal of Pharmaceutical Research February 2016; 15 (2): 427-434 ISSN: 1596-5996 (print); 1596-9827 (electronic) © Pharmacotherapy Group, Faculty of Pharmacy, University of Benin, Benin City, 300001 Nigeria. All rights reserved. Available online at http://www.tjpr.org http://dx.doi.org/10.4314/tjpr.v15i2.29 Review Article Main Analytical Techniques Used for Elemental Analysis in Various Matrices ABM Helaluddin1, Reem Saadi Khalid1, Mohamed Alaama and Syed Atif Abbas2 1Analytical and Bio-Analytical Research Laboratory, Department of Pharmaceutical Chemistry, Faculty of Pharmacy, International Islamic University Malaysia (IIUM), Jalan Istana, Bandar Indera Mahkota, 25200 Kuantan, Pahang, 2 Malaysia School of Pharmacy, Taylors University, 1 Jalan Taylor’s, 47500 Subang Jaya, Selangor Darul Ehsan, Malaysia *For correspondence: Email: [email protected]; [email protected] Received: 20 August 2015 Revised accepted: 4 January 2016 Abstract Heavy metal pollution is a serious environmental problem. The presence of such metals in different areas of an ecosystem subsequently leads to the contamination of consumable products such as dietary and processed materials. Accurate monitoring of metal concentrations in various samples is of importance in order to minimize health hazards resulting from exposure to such toxic substances. For this purpose, it is essential to have a general understanding of the basic principles for different methods of elemental analysis. This article provides an overview of the most sensitive -

Why the Gas Chromatographic Separation Method Used in the Thermo Scientific Flashsmart Elemental Analyzer Is the Most Reliable for Elemental Analysis?
FlashSmart Elemental Analyzer SmartNotes Why the gas chromatographic separation method used in the Thermo Scientific FlashSmart Elemental Analyzer is the most reliable for elemental analysis? The Thermo Scientific™ FlashSmart™ Elemental Analyzer (Figure 1) operates with the dynamic flash combustion (modified Dumas Method) of the sample for CHNS determination while for oxygen analysis, the system operates in pyrolysis mode. The resulted gases are carried by a helium (or argon) flow till a gas chromatographic column that provides the separation of the gases, and finally, detected by a thermal conductivity detector (TCD). A complete report is automatically generated by the Thermo Scientific™ EagerSmart™ Data Handling Software and displayed at the end of the analysis. Thermo Scientific FlashSmart: The Elemental Analyzer Figure 1. Thermo Scientific FlashSmart Elemental Analyzer. The gas chromatography IC (GC) provides a “real” picture The advantages of the separation method are: of the analytical process during combustion (CHNS) and pyrolysis (O). • “Real” peak of each element. • Easy integration of the peaks by the EagerSmart Data GC technique provides you with complete peak Handling Software. separation and sharp peak shapes, which ensure superior precision and higher sensitivity. • The area of the peak corresponds to the total amount of the element. From the chromatogram you can quantify the amount • Proper quantification of the elements. of elements in your sample and recognize what it is happening inside the analyzer anytime. GC separation • Maintenance free, long lifetime GC column operating features provide you with: for years without the need for replacement: it is not a consumable. • Full insight of the combustion showing complete • GC Column easy to use, directly installed in the analyzer conversion of: • Straightforward continuous flow design from sample – Nitrogen and nitrogen oxide in N 2 processing through gas separation and detection. -

Stark Broadening Measurements in Plasmas Produced by Laser Ablation of Hydrogen Containing Compounds Miloš Burger, Jorg Hermann
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Archive Ouverte en Sciences de l'Information et de la Communication Stark broadening measurements in plasmas produced by laser ablation of hydrogen containing compounds Miloš Burger, Jorg Hermann To cite this version: Miloš Burger, Jorg Hermann. Stark broadening measurements in plasmas produced by laser ablation of hydrogen containing compounds. Spectrochimica Acta Part B: Atomic Spectroscopy, Elsevier, 2016, 122, pp.118-126. 10.1016/j.sab.2016.06.005. hal-02348424 HAL Id: hal-02348424 https://hal.archives-ouvertes.fr/hal-02348424 Submitted on 5 Nov 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Stark broadening measurements in plasmas produced by laser ablation of hydrogen containing compounds Milosˇ Burgera,∗,Jorg¨ Hermannb aUniversity of Belgrade, Faculty of Physics, POB 44, 11000 Belgrade, Serbia bLP3, CNRS - Aix-Marseille University, 13008 Marseille, France Abstract We present a method for the measurement of Stark broadening parameters of atomic and ionic spectral lines based on laser ablation of hydrogen containing compounds. Therefore, plume emission spectra, recorded with an echelle spectrometer coupled to a gated detector, were compared to the spectral radiance of a plasma in local thermal equi- librium. -

Good Practice Guide for Isotope Ratio Mass Spectrometry, FIRMS (2011)
Good Practice Guide for Isotope Ratio Mass Spectrometry Good Practice Guide for Isotope Ratio Mass Spectrometry First Edition 2011 Editors Dr Jim Carter, UK Vicki Barwick, UK Contributors Dr Jim Carter, UK Dr Claire Lock, UK Acknowledgements Prof Wolfram Meier-Augenstein, UK This Guide has been produced by Dr Helen Kemp, UK members of the Steering Group of the Forensic Isotope Ratio Mass Dr Sabine Schneiders, Germany Spectrometry (FIRMS) Network. Dr Libby Stern, USA Acknowledgement of an individual does not indicate their agreement with Dr Gerard van der Peijl, Netherlands this Guide in its entirety. Production of this Guide was funded in part by the UK National Measurement System. This publication should be cited as: First edition 2011 J. F. Carter and V. J. Barwick (Eds), Good practice guide for isotope ratio mass spectrometry, FIRMS (2011). ISBN 978-0-948926-31-0 ISBN 978-0-948926-31-0 Copyright © 2011 Copyright of this document is vested in the members of the FIRMS Network. IRMS Guide 1st Ed. 2011 Preface A few decades ago, mass spectrometry (by which I mean organic MS) was considered a “black art”. Its complex and highly expensive instruments were maintained and operated by a few dedicated technicians and its output understood by only a few academics. Despite, or because, of this the data produced were amongst the “gold standard” of analytical science. In recent years a revolution occurred and MS became an affordable, easy to use and routine technique in many laboratories. Although many (rightly) applaud this popularisation, as a consequence the “black art” has been replaced by a “black box”: SAMPLES GO IN → → RESULTS COME OUT The user often has little comprehension of what goes on “under the hood” and, when “things go wrong”, the inexperienced operator can be unaware of why (or even that) the results that come out do not reflect the sample that goes in. -

Elemental Analysis: CHNS/O Determination in Carbon
APPLICATION NOTE 42182 Elemental Analysis: CHNS/O determination in carbon Authors Introduction Dr. Liliana Krotz and Carbon occurs as a variety of allotropes. There are two crystalline forms, Dr. Guido Giazzi diamond and graphite, and a number of amorphous (non-crystalline) forms, Thermo Fisher Scientific, such as charcoal, coke, and carbon black. The most common use of carbon Milan, Italy black is as a pigment and reinforcing phase in automobile tires. Coke is the solid carbonaceous material derived from destructive distillation of low-ash, Keywords low-sulfur bituminous coal. Coke is also used as a fuel and as a reducing Coal, Coke, Carbon Black, agent in smelting iron ore in a blast furnace. Graphite, CHNS/O, Heat Value For quality control purposes, the organic elements in carbon need to be Goal determined. For the determination of carbon, hydrogen, nitrogen, sulfur and This application note reports data oxygen, the combustion method is used. on CHNS/O determination on carbon samples needed for quality The Thermo Scientific™ FlashSmart™ Elemental Analyzer (Figure 1) allows control purposes, performed with the quantitative determination of carbon, hydrogen, nitrogen and oxygen in the FlashSmart EA. carbon. The FlashSmart EA based on the dynamic flash combustion of the sample, provides automated and simultaneous CHNS determination in a single analysis run and oxygen determination by pyrolysis in a second run. To perform total sulfur determination at trace levels, the analyzer has been coupled with the Flame Photometric Detector (FPD). Methods For CHNS determination, the FlashSmart EA operates according to the dynamic flash combustion of the sample. Liquid samples are weighed in tin containers and introduced into the combustion reactor via the Thermo Scientific™ MAS Plus Autosampler. -

The Role of Nanoanalytics in the Development of Organic-Inorganic Nanohybrids—Seeing Nanomaterials As They Are
nanomaterials Review The Role of Nanoanalytics in the Development of Organic-Inorganic Nanohybrids—Seeing Nanomaterials as They Are Daria Semenova 1 and Yuliya E. Silina 2,* 1 Process and Systems Engineering Center (PROSYS), Department of Chemical and Biochemical Engineering, Technical University of Denmark, 2800 Kgs. Lyngby, Denmark; [email protected] 2 Institute of Biochemistry, Saarland University, 66123 Saarbrücken, Germany * Correspondence: [email protected] or [email protected]; Tel.: +49-681-302-64717 Received: 23 October 2019; Accepted: 19 November 2019; Published: 23 November 2019 Abstract: The functional properties of organic-inorganic (O-I) hybrids can be easily tuned by combining system components and parameters, making this class of novel nanomaterials a crucial element in various application fields. Unfortunately, the manufacturing of organic-inorganic nanohybrids still suffers from mechanical instability and insufficient synthesis reproducibility. The control of the composition and structure of nanosurfaces themselves is a specific analytical challenge and plays an important role in the future reproducibility of hybrid nanomaterials surface properties and response. Therefore, appropriate and sufficient analytical methodologies and technical guidance for control of their synthesis, characterization and standardization of the final product quality at the nanoscale level should be established. In this review, we summarize and compare the analytical merit of the modern analytical methods, viz. Fourier transform infrared spectroscopy (FTIR), RAMAN spectroscopy, surface plasmon resonance (SPR) and several mass spectrometry (MS)-based techniques, that is, inductively coupled plasma mass spectrometry (ICP-MS), single particle ICP-MS (sp-ICP-MS), laser ablation coupled ICP-MS (LA-ICP-MS), time-of-flight secondary ion mass spectrometry (TOF-SIMS), liquid chromatography mass spectrometry (LC-MS) utilized for characterization of O-I nanohybrids. -
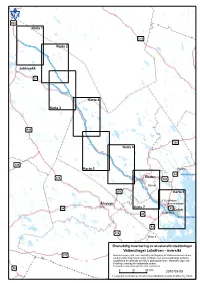
Lulealven.Pdf
± E45 Karta 1 E10 Karta 2 Jokkmokk 97 Karta 4 Karta 3 E45 356 Karta 6 E45 Karta 5 Råneå E4 Rånefjärden Boden 374 383 Sävast 356 Karta 8 S Sunderbyn Älvsbyn Gammelstaden Karta 7 94 97 Luleå Bergnäset 94 Brändöfjärden E4 374 Rosvik Kallfjärden Översiktlig inventering avStorfjärden erosionsförutsättningar Vattendraget Luleälven - översikt 373 Redovisningen ingår i en översiktligPiteå kartläggning av stranderosionen utmed landets vattendrag.Bergsviken Kartan visar områden med erosionskänsliga jordarter. Uppgifterna är baserade på SGU:s geologiska kartor. Materialet utgör inte tillräckligt underlag för detaljerade studier. Eventuella erosionsskydd har inte inventerats. 95 0 10 20 Km Bondöfjärden2010-09-09 © Copyright Lantmäteriet. Ur GSD-Översiktskartan ärende nr MS2010_10049 Översiktlig inventering av erosionsförutsättningar ± Vattendraget Luleälven - karta 1 Redovisningen ingår i en översiktlig kart- Teckenförklaring läggning av stranderosionen utmed landets vattendrag. Kartan visar områden med Grovsand-finsand erosionskänsliga jordarter. Uppgifterna är Silt baserade på SGU:s geologiska kartor. Materialet utgör inte tillräckligt underlag Svämsediment för detaljerade studier. Fyllning Eventuella erosionsskydd har inte inventerats. Tätort >200 invånare 0 1 2 3 4 Km 2010-09-09 © Copyright Lantmäteriet. Ur GSD-Översiktskartan ärende nr MS2010_10049 E45 Lu leälven Översiktlig inventering av erosionsförutsättningar ± Vattendraget Luleälven - karta 2 Redovisningen ingår i en översiktlig kart- Teckenförklaring läggning av stranderosionen utmed landets -

Elemental and Isotopic Characterization of Tobacco from Umbria
H OH metabolites OH Article Elemental and Isotopic Characterization of Tobacco from Umbria Luana Bontempo 1,*, Daniela Bertoldi 2, Pietro Franceschi 1, Fabio Rossi 3 and Roberto Larcher 2 1 Research and Innovation Centre, Fondazione Edmund Mach (FEM), Via E. Mach 1, 38098 San Michele all’Adige, Italy; [email protected] 2 Technology Transfer Centre, Experiment and Technological Services Department, Fondazione Edmund Mach (FEM), Via E. Mach 1, 38098 San Michele all’Adige, Italy; [email protected] (D.B.); [email protected] (R.L.) 3 Fattoria Autonoma Tabacchi Soc. Coop Agricola, Via G. Oberdan, 06012 Città di Castello, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-461-615-138 Abstract: Umbrian tobacco of the Virginia Bright variety is one of the most appreciated tobaccos in Europe, and one characterized by an excellent yield. In recent years, the Umbria region and local producers have invested in introducing novel practices (for production and processing) focused on environmental, social, and economic sustainability. Due to this, tobacco from Umbria is a leading commodity in the global tobacco industry, and it claims a high economic value. The aim of this study is then to assess if elemental and isotopic compositions can be used to protect the quality and geographical traceability of this particular tobacco. For the first time the characteristic value ranges of the stable isotope ratios of the bio-elements as a whole (δ2H, δ13C, δ15N, δ18O, and δ34S) and of the concentration of 56 macro- and micro-elements are now available, determined in Virginia Bright tobacco produced in two different areas of Italy (Umbria and Veneto), and from other worldwide Citation: Bontempo, L.; Bertoldi, D.; geographical regions. -

Spectrometric Techniques for Elemental Profile Analysis Associated with Bitter Pit in Apples
Postharvest Biology and Technology 128 (2017) 121–129 Contents lists available at ScienceDirect Postharvest Biology and Technology journal homepage: www.elsevier.com/locate/postharvbio Spectrometric techniques for elemental profile analysis associated with bitter pit in apples a a a,b a Carlos Espinoza Zúñiga , Sanaz Jarolmasjed , Rajeev Sinha , Chongyuan Zhang , c,d c,d a,b, Lee Kalcsits , Amit Dhingra , Sindhuja Sankaran * a Department of Biological Systems Engineering, Washington State University, Pullman, WA, USA b Center for Precision and Automated Agricultural Systems, Department of Biological Systems Engineering, IAREC, Washington State University, Prosser, WA, USA c Department of Horticulture, Washington State University, Pullman, WA, USA d Tree Fruit Research and Extension Center, Washington State University, Wenatchee, WA, USA A R T I C L E I N F O A B S T R A C T Article history: ‘ ’ ‘ ’ ‘ ’ Received 6 December 2016 Bitter pit and healthy Honeycrisp , Golden Delicious , and Granny Smith apples were collected from Received in revised form 20 February 2017 three commercial orchards. Apples were scanned using Fourier transform infrared (FTIR) and X-ray Accepted 21 February 2017 fluorescence (XRF) spectrometers to associate the elemental profile with bitter pit occurrence in apples. Available online xxx The FTIR spectra were acquired from apple peel and flesh; while XRF spectra were acquired from the apple surface (peel). Destructive elemental analysis was also performed to estimate calcium, magnesium, Keywords: and potassium concentrations in the apples. There were significant differences between healthy and Apple disorder bitter pit affected apples in calcium, magnesium, and potassium concentrations, in addition to Support vector machine magnesium/calcium and potassium/calcium ratios (5% level of significance). -

Läs Hela Analysen
Bostadsförsörjningsanalys Luleå December 2020 Linda Lövgren, Maria Pleiborn, Ebba Älvgren Förord Syftet med analysen att analysera och bedöma det kommande behovet av bostäder för Luleå kommun och vilken typ av bostäder som kommer att efterfrågas med avseende på de målgrupper som är aktuella. Analysen är en grund till riktlinjerna för bostadsförsörjning och innehåller en analys av kommunens demografiska utveckling, efterfrågan på bostäder, bostadsbehovet för särskilda grupper och marknadsförutsättningarna för bostäder totalt sett samt för kommunens delområden. Analysen har även kopplats till bostadsmarknadsläget i grannkommunerna och den regionala bostads- och arbetsmarknaden i stort. 2 Författare: Linda Lövgren, Maria Pleiborn och Ebba Älvgren. Stockholm, december 2020 WSP Sverige AB SE-121 88 Stockholm-Globen Arenavägen 7 Org. nr: 556057-4880 Innehåll — Slutsatser och åtgärdsförslag för bostadsmarknaden i Luleå kommun — Allmänt om bostadsmarknaden i Sverige — Omvärldsanalys — Demografisk analys — Bostadsbestånd och nyproduktion 3 — Bostadssituationen för särskilda grupper — Efterfrågan på bostäder — Geografisk analys Slutsatser Luleå är en kommun med en stabil Däremot är efterfrågan oftast på bostäder befolkningsökning och en förhållandevis som är billigare än vad marknaden förmår låg arbetslöshet. Luleå kommun har i stort att producera. Detta leder till att en hög sett en god bostadsförsörjning i nuläget grad av exploatering inte leder till en bättre och det finns ingen underliggande bostadsförsörjning i kommunen. strukturell bostadsbrist. Däremot är en god planeringsberedskap Däremot finns ett behov av bostäder med alltid positiv, eftersom det ger flexibilitet i 4 en lägre prisnivå än vad marknaden förmår planeringen. att producera redan hos den existerande befolkningen. En övervägande andel av de nya invånarna är invandrare, som oftast har en något lägre betalningsförmåga. -

(Elemental) Analysis
Modern Methods in Heterogeneous Catalysis Research 29.10.2004 Chemical (Elemental) Analysis Raimund Horn Dep. of Inorganic Chemistry, Group Functional Characterization, Fritz-Haber-Institute of the Max-Planck-Society Outline 1. Introduction 1.1 Concentration Ranges 1.2 Accuracy and Precision 1.3 Decision Limit, Detection Limit, Determination Limit 2. Methods for Quantitative Elemental Analysis 2.1 Chemical Methods 2.1.1 Volumetric Methods 2.1.2 Gravimetric Methods 2.2 Electroanalytical Methods 2.2.1 Potentiometry 2.2.2 Polarography Outline 2.6 Spectroscopic Methods 2.6.1 Atomic Emission Spectroscopy 2.6.2 Atomic Absorption Spectroscopy 2.6.3 Inductively Coupled Plasma Mass Spectrometry 2.6.4 X-Ray Fluorescence Spectroscopy 2.6.5 Electron Probe X-Ray Microanalysis 3 Specification of an Analytical Result 4 Summary 5 Literature Backup Slide The Analytical Process strategy problem solution characterization representative sampling chemical sample object of investigation information preparation measurement evaluation data diminution analysis smaller sample for measurement analytical sample measurement data result Backup Slide The Analytical Process strategy problem interpretation characterization representative sampling chemical sample object of investigation information preparation measurement evaluation data diminution analysis smaller sample for measurement analytical sample measurement data result Backup Slide Analytical Problems in Science, Industry, Law… Science Industry Law example: Determination by X- example: manufacturer of iron - example: Is the accused guilty ray Absorption Spectroscopy of Shall I by this iron ore or not ? or not guilty ? the Fe-Fe Separation in the Oxidized Form of the Hydroxylase of Methane Monooxygenase Alone and in the Presence of MMOD Rudd D. J., Sazinsky M. -

The Application of Elemental Analysis for the Determination of the Elemental 2013: Analysis of the Influence of Cutting Parameters on Surface Roughness
6. VARASQUIM F., ALVES M., GONCALVES M., SANTIAGO L., SOUZA A., Annals of Warsaw University of Life Sciences - SGGW 2011: Influence of belt speed, grit size and pressure on the sanding of Eucalyptus Forestry and Wood Technology № 92, 2015: 477-482 (Ann. WULS - SGGW, For. and Wood Technol. 92, 2015) grandis wood. Cerne, Lavras, v.18, n 2:231-237 7. WILKOWSKI J., ROUSEK M., SVOBODA E., KOPECKÝ Z., CZARNIAK P., The application of elemental analysis for the determination of the elemental 2013: Analysis of the influence of cutting parameters on surface roughness. Annals of Warsaw University of Life Sciences – SGGW 84: 321-325. composition of lignocellulosic materials Streszczenie: Wpływ parametrów skrawania na chropowatość powierzchni płyty MDF po MAGDALENA WITCZAK, MAŁGORZATA WALKOWIAK, WOJCIECH CICHY, frezowaniu i szlifowaniu. W pracy zbadano wpływ podstawowych parametrów skrawania MAGDALENA KOMOROWICZ takich jak posuw na ząb lub obrót oraz prędkość obrotowa wrzeciona na chropowatość Environmental Protection and Wood Chemistry Department, Wood Technology Institute, Poznan powierzchni płyt MDF. Próbki poddano dwóm rodzajom operacji tj. frezowaniu i szlifowaniu. Przy pomocy profilometru stykowego uzyskano 2 podstawowe parametry Abstract: The application of elemental analysis for the determination of the elemental composition of opisujące strukturę geometryczną powierzchni (Ra, Rz,). Wyniki pokazują, że istotny lignocellulosic materials. The presented work includes research on different types of lignocellulosic materials: woody materials, agricultural materials and fruity materials. The aim of the study was the determination of the statystycznie wpływ na chropowatość powierzchni płyty MDF ma posuw na ząb (przy elemental composition of lignocellulosic materials using elemental analysis by high-temperature combustion, frezowaniu) lub posuw na obrót (przy szlifowaniu).