Toxicological Profile for Cadmium
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

(A) 1) at First a Pair of Each 10G. of Sample Was Extracted with Ether by the Soxhlet's Apparatus for a Week
52 [vol. 6, THE DISTRIBUTION OF PHOSPHORUS IN COW'S MILK AND THE SCHEME FOR THE SEPARATION OF PHOSPHATIDES By Rinjiro SASAKI (From the Institute of Agricultural Chemistry, College of Agriculture, Tokyo Imperial University) (Received 9th September 1930) A. The Distribution of Phosphorus in Cow's milk In order to extract the phosphatides, it is necessary first to determine the distribution of phosphorus in cow's milk. Liquid milk can not be extracted with ether or with other organic solvents owing to its large content of water. The substance applied in this experiment is the milk powder, dried by the Buflovak drum drier below 70•Ž. Its content of moisture is 4.498%, deter mined by the method of drying in the air bath of 105•Ž. The content of total phosphorus was determined(5) in the ash of sample which was burned with a little of fusing mixture. In 100g. of milk powder In 100g. of dry matter Total phosphorus 0.745g. 0.781g. The amounts of phosphorus, which are soluble in the various organic sol vents, were determined by extracting with three solvents in the following orders. (A) Ether-Acetone-Alcohol (B) Acetone-Ether-Alcohol (C) Alcohol-Ether-Acetone Experiment (A) 1) At first a pair of each 10g. of sample was extracted with ether by the Soxhlet's apparatus for a week. After the extraction had been comp leted, the extract was freed from ether and weighed. In 100g.of milkpowder In 100g.of dry matter Total ether matters soluble 3.609g. 3.779g. 2) The above extract was dissolved in a very little of ether. -

Cadmium- Toxfaqs™
Cadmium- ToxFAQs™ CAS # 7440-43-9 This fact sheet answers the most frequently asked health questions (FAQs) about cadmium. For more information, call the CDC Information Center at 1-800-232-4636. This fact sheet is one in a series of summaries about hazardous substances and their health effects. It is important you understand this information because this substance may harm you. The effects of exposure to any hazardous substance depend on the dose, the duration, how you are exposed, personal traits and habits, and whether other chemicals are present. HIGHLIGHTS: Exposure to cadmium happens mostly in the workplace where cadmium products are made. The general population is exposed from breathing cigarette smoke or eating cadmium contaminated foods. Cadmium damages the kidneys, lungs, and bones. Cadmium has been found in at least 1,014 of the 1,669 National Priorities List (NPL) sites identified by the Environmental Protection Agency (EPA). What is cadmium? How might I be exposed to cadmium? Cadmium is a natural element in the earth’s crust. It is • Eating foods containing cadmium; low levels are usually found as a mineral combined with other elements found in all foods (highest levels are found in leafy such as oxygen (cadmium oxide), chlorine (cadmium vegetables, grains, legumes, and kidney meat). chloride), or sulfur (cadmium sulfate, cadmium sulfide). • Smoking cigarettes or breathing cigarette smoke. All soils and rocks, including coal and mineral fertilizers, • Breathing contaminated workplace air. contain some cadmium. Most cadmium used in the United States is extracted during the production of other metals • Drinking contaminated water. like zinc, lead, and copper. -

United States Patent Office Patented Apr
3,030,421 United States Patent Office Patented Apr. 17, 1962 2 3,030,421 perature and under a reduced pressure. In the case of PROCESS FOR PREPARNG TRHYDROXY most of the catalysts the reaction products, which are free METHYL-PHOSPHINE s from solvents, solidify already when being cooled to room Martin Reuter and Ludwig Ortane, Frankfurt an Main, temperature. If, in the case of some catalysts, they Germany, assignors to Farbwerke Hoechst Aktienger 5 solidify only at a lower temperature and still contain to sellschaft vormas Meister Lucius & Briining, Frank a greater extent oily by-products and/or phosphonium furt am Main, Germany, a corporation of Gerinally hydroxide, they can be separated from the latter by filter No Drawing. Fied Jaa. 14, 1958, Ser. No. 708,764 ing or pressing. - - - Claims priority, application Germany Jan. 23, 1957 It is to be assumed that the crystalline main product 6 Canas. (C. 260-606.5) O of the present invention constitutes the hitherto unknown We have found that a new and valuable phosphorus trihydroxymethyl-phosphine. Main and by-products are compound carrying hydroxymethyl groups at the phos easily soluble in water and methanol and sparingly solu phorus atom can be prepared by reacting 1 mol of formal ble in fat dissolvers. dehyde with 4 mol of phosphine, preferably in the pres The reaction products of the invention can be used as ence of water, and in the presence of Small quantities of 5 insecticides, additives for lubricants, flame-proofing agents finely distributed metals that do not belong to the alkali for wood and textiles and as intermediates for these sub metals or alkaline earth metals and/or their compounds Staces. -

METASTABILITY of CADMIUM SULFATE and ITS EFFECT on ELECTROMOTIVE FORCE of SATURATED STANDARD CELLS by George W
U. S. DEPARTMENT OF C OMMERCE NATIONAL BUREAU OF STANDARDS RESEARCH PAPER RP1389 Part of Journal of Research of the National Bureau of Standards, Volume 26, May 1941 METASTABILITY OF CADMIUM SULFATE AND ITS EFFECT ON ELECTROMOTIVE FORCE OF SATURATED STANDARD CELLS By George W. Vinal and Langhorne H. Brickwedde ABSTRACT Both solubility and electromot ive force measurements are concordant in fixing the transition temperature of CdSOf.8/3HzO to CdSOf.HzO, at 43.4°C. Previously the transition temperature was genera.lly believed to be 74° C. Cells recently were made with each modification. Their respective electromotive forces differ except at the transition temperature, where they, as well as the solubilitiesJ are equal. The temperature coefficient of cells containing CdSO•. HzO was founa to be positive, whereas the temperature coefficient for cells containing the ordi nary salt CdS04.8/3HzO is negative, as is well known. Both hydrates tend to persist in a metastable state, and measurements can be made over a wide range of temperatures. The free energy changes of the cells are discussed, and some practical applications for the cells are described. CONTENTS Page I. Introduction _____ __ ______ __ __________ ________ ______ ___ ___ __ ____ 455 II. Experimental procedure ___ ______________ _____ __ _________________ 457 1. Solubility det erminations __ __ __ ____ _____________ ______ _____ 457 2. Construction of cells __________________ ____ __ ___ __ ____ _____ 457 3. Temperature control of the cells and measurement of electromo- tive fo rce _______________________ _____ ___ _____ __________ 458 III. Experimental results ___________________________________________ _ 458 1. -

Draft Permit
Air Individual Permit Major Amendment 14900013-104 Permittee: DENCO II LLC Facility name: DENCO II LLC 227 County Road 22 Morris, MN 56267-4691 Stevens County Operating permit issuance date: May 1, 2019 Expiration date: May 1, 2024 * All Title I Conditions do not expire Major Amendment: [Action Issue Date] Permit characteristics: State; Limits to avoid Part 70/ Limits to avoid NSR; Limits to avoid NSR The emission units, control equipment and emission stacks at the stationary source authorized in this permit amendment are as described in the submittals listed in the Permit Applications Table. This permit amendment supersedes Air Emission Permit No. 14900013-103 and authorizes the Permittee to operate the stationary source at the address listed above unless otherwise noted in the permit. The Permittee must comply with all the conditions of the permit. Any changes or modifications to the stationary source must be performed in compliance with Minn. R. 7007.1150 to 7007.1500. Terms used in the permit are as defined in the state air pollution control rules unless the term is explicitly defined in the permit. Unless otherwise indicated, all the Minnesota rules cited as the origin of the permit terms are incorporated into the SIP under 40 CFR § 52.1220 and as such are enforceable by U.S. Environmental Protection Agency (EPA) Administrator or citizens under the Clean Air Act. Signature: [ ] This document has been electronically signed. for the Minnesota Pollution Control Agency for Steven S. Pak, P.E., Manager Air Quality Permits Section Industrial Division Table of Contents Page 1. Permit applications table ............................................................................................................................................ -

Thermodynamic Properties of Mixtures of Aqueous Solutions of Hydrochloric Acid and Cadmium Chloride, Copper Chloride, Manganese Chloride, and Zinc Chloride
Western Michigan University ScholarWorks at WMU Master's Theses Graduate College 12-1993 Thermodynamic Properties of Mixtures of Aqueous Solutions of Hydrochloric Acid and Cadmium Chloride, Copper Chloride, Manganese Chloride, and Zinc Chloride Samia A. Kosa Follow this and additional works at: https://scholarworks.wmich.edu/masters_theses Part of the Chemistry Commons Recommended Citation Kosa, Samia A., "Thermodynamic Properties of Mixtures of Aqueous Solutions of Hydrochloric Acid and Cadmium Chloride, Copper Chloride, Manganese Chloride, and Zinc Chloride" (1993). Master's Theses. 4363. https://scholarworks.wmich.edu/masters_theses/4363 This Masters Thesis-Open Access is brought to you for free and open access by the Graduate College at ScholarWorks at WMU. It has been accepted for inclusion in Master's Theses by an authorized administrator of ScholarWorks at WMU. For more information, please contact [email protected]. THERMODYNAMIC PROPERTIES OF MIXTURES OF AQUEOUS SOLUTIONS OF HYDROCHLORIC ACID AND CADMIUM CHLORIDE, COPPER CHLORIDE, MANGANESE CHLORIDE, AND ZINC CHLORIDE by Samia A. Kosa A Thesis Submitted to the Faculty of The Graduate College in partial fulfillment of the requirements for the Degree of Master of Arts Department of Chemistry Western Michigan University Kalamazoo, Michigan December 1993 ACKNOWLEDGEMENTS I wish to express my deep gratitude and sincere appreciation to my committee advisor, Dr. Donald Schriber, for his continual guidance, direction, and assistance in this research. I would also like to acknowledge the help of the other committee members, Dr. Thomass Houser and Dr. Ralph Steinhaus, towards this work. My family continuously provided me support and encouragement and to them I owe more than I can say. -

Download Author Version (PDF)
Journal of Materials Chemistry A Accepted Manuscript This is an Accepted Manuscript, which has been through the Royal Society of Chemistry peer review process and has been accepted for publication. Accepted Manuscripts are published online shortly after acceptance, before technical editing, formatting and proof reading. Using this free service, authors can make their results available to the community, in citable form, before we publish the edited article. We will replace this Accepted Manuscript with the edited and formatted Advance Article as soon as it is available. You can find more information about Accepted Manuscripts in the Information for Authors. Please note that technical editing may introduce minor changes to the text and/or graphics, which may alter content. The journal’s standard Terms & Conditions and the Ethical guidelines still apply. In no event shall the Royal Society of Chemistry be held responsible for any errors or omissions in this Accepted Manuscript or any consequences arising from the use of any information it contains. www.rsc.org/materialsA Page 1 of 9 Journal of Materials Chemistry A ARTICLE JMCA Safer Salts for CdTe Nanocrystal Solution Processed Solar Cells: The Dual Roles of Ligand Exchange and Grain Growth Received 00th January 20xx, a b c d e Accepted 00th January 20xx Troy K. Townsend, † William B. Heuer, Edward E. Foos, Eric Kowalski, Woojun Yoon and Joseph G. Tischler e DOI: 10.1039/x0xx00000x Inorganic CdSe/CdTe nanocrystals for solid-state photovoltaic devices are typically sintered into a bulk-like material after www.rsc.org/ annealing in the presence of solid cadmium chloride. -
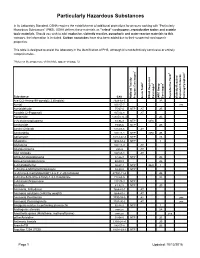
Particularly Hazardous Substances
Particularly Hazardous Substances In its Laboratory Standard, OSHA requires the establishment of additional protections for persons working with "Particularly Hazardous Substances" (PHS). OSHA defines these materials as "select" carcinogens, reproductive toxins and acutely toxic materials. Should you wish to add: explosive, violently reactive, pyrophoric and water-reactve materials to this category, the information is included. Carbon nanotubes have also been added due to their suspected carcinogenic properties. This table is designed to assist the laboratory in the identification of PHS, although it is not definitively conclusive or entirely comprehensive. *Notes on the proper use of this table appear on page 12. 1 6 5 2 3 4 Substance CAS National Toxicity National Program Carcinogen Toxin Acute Regulated OSHA Carcinogen Group IARC Carcinogen Toxin Reproductive Violently Reactive/ Explosive/Peroxide Forming/Pyrophoric A-a-C(2-Amino-9H-pyrido[2,3,b]indole) 2648-68-5 2B Acetal 105-57-7 yes Acetaldehyde 75-07-0 NTP AT 2B Acrolein (2-Propenal) 107-02-8 AT Acetamide 126850-14-4 2B 2-Acetylaminofluorene 53-96-3 NTP ORC Acrylamide 79-06-6 NTP 2B Acrylyl Chloride 814-68-6 AT Acrylonitrile 107-13-1 NTP ORC 2B Adriamycin 23214-92-8 NTP 2A Aflatoxins 1402-68-2 NTP 1 Allylamine 107-11-9 AT Alkylaluminums varies AT Allyl Chloride 107-05-1 AT ortho-Aminoazotoluene 97-56-3 NTP 2B para-aminoazobenzene 60-09-3 2B 4-Aminobiphenyl 92-67-1 NTP ORC 1 1-Amino-2-Methylanthraquinone 82-28-0 NTP (2-Amino-6-methyldipyrido[1,2-a:3’,2’-d]imidazole) 67730-11-4 2B -

Inorganic Chemistry Test for Cadmium Radical
Chemistry Inorganic Chemistry Test for Cadmium Radical General Aim Method Detection of the presence of cadmium ion as a base Detection of the presence of cadmium as a base radical radical in inorganic salts such as cadmium sulfate. using specic chemical reagents. Learning Objectives (ILOs) Dene and dierentiate between members of the second group cations and those of other cation groups. Classify inorganic salts according to their base radicals. Compare between cadmium containing salts and other members of the same group in terms of chemical structures, properties and reactions. Identify cadmium radicals containing salts experimentally. Select the appropriate reagents to detect the presence of cadmium radical. Balance the chemical equations of chemical reactions. Theoretical Background/Context Cadmium (Cd) is one of the transition metals that are located in the d-block of the periodic table. Cadmium is located in the fth period and twelfth group of the periodic table. Cd possesses an atomic number of 48 and an atomic mass of 112.411g. It was rst discovered by the German scientist, Friedrich Strohmeyer in 1817 in Germany. At that time, cadmium was commonly used to protect iron and steel from corrosion as it was inserted as a sacricial anode. Additionally, it was used in the manufacture of nickel-cadmium batteries. Cadmium is a highly toxic element so it has to be handled with great caution. Abundance of Cadmium in Nature Cadmium cannot be easily found in its elemental form naturally. It has been detected in the Earth's Crust in very minute amounts that do not exceed 0.1 to 0.2 ppm. -

The Effect of Sargassum Angustifolium Ethanol Extract on Cadmium Chloride-Induced Hypertension in Rat
Research Journal of Pharmacognosy (RJP) 8(1), 2021: 81-89 Received: 31 Oct 2020 Accepted: 17 Dec 2020 Published online: 19 Dec 2020 DOI: 10.22127/RJP.2020.255203.1637 Original article The Effect of Sargassum angustifolium Ethanol Extract on Cadmium Chloride-Induced Hypertension in Rat Leila Safaeian1* , Afsaneh Yegdaneh2, Masoud Mobasherian1 1Department of Pharmacology and Toxicology, Isfahan Pharmaceutical Sciences Research Center, School of Pharmacy and Pharmaceutical Sciences, Isfahan University of Medical Sciences, Isfahan, Iran. 2Department of Pharmacognosy, School of Pharmacy and Pharmaceutical Sciences, Isfahan University of Medical Sciences, Isfahan, Iran. Abstract Background and objectives: Sargassum angustifolium is a brown alga in southwestern coastline of Persian Gulf. Regarding the presence of various bioactive compounds and evidence of antihypertensive effects in other species of Sargassum, we evaluated the effect of S. angustifolium ethanol extract in CdCl2-induced hypertension in Wistar rats. Methods: Alga extract was prepared by maceration method using 70% ethanol and assessed for total phenolics and salt content. CdCl2 (1.5 mg/kg/day) was administered intraperitoneally to the rats for two weeks. Treatment groups received S. angustifolium extract (20, 40 and 80 mg/kg) or nifedipine (10 mg/kg) orally and simultaneously were given CdCl2 for two weeks. Systolic blood pressure (SBP) and heart rate were measured using tail-cuff method. Total antioxidant capacity, urea, creatinine, electrolytes including sodium, potassium, calcium and chloride were estimated in blood samples. The weight and histopathology of kidney tissues were also evaluated. Results: The content of total phenolic as gallic acid equivalent and the salt as NaCl was 67.42 ± 9.5 mg/g and 6.9 g/100 g in dried ethanol extract, respectively. -

Table of Contents
UNIVERSITY OF MISSOURI-KANSAS CITY CHEMICAL MANAGEMENT PLAN Revised May 2016 UMKC CHEMICAL MANAGEMENT PLAN This document constitutes the Chemical Management Plan (CMP) for the University of Missouri-Kansas City (UMKC). It was developed by the Environmental Health and Safety Department (EHS), to ensure the safe and proper use of hazardous and non- hazardous chemicals and to comply with applicable governmental regulations addressing the disposal of these chemicals. In addition, it was developed to foster waste minimization, and to provide the faculty and the staff with a management program to reduce the potential for accidents involving hazardous chemicals and/or wastes. Elements of the CMP include: a. a procedure for identifying potential or actual hazardous chemicals or wastes b. a procedure for periodic reexamination of those hazardous chemicals or wastes identified by the procedure in (a.) above as well as a systematic method for identification and evaluation of any new potential or actual hazardous chemicals or wastes c. procedures for labeling, and inventorying hazardous chemicals or wastes d. a procedure for identification and training of personnel directly responsible for ensuring that (a.), (b.), and (c.) are implemented e. a procedure for monitoring, recording, and reporting compliance with the CMP f. a procedure by which information generated by the CMP is provided to the persons performing waste analyses Each element is addressed as part of the complete CMP in the following paragraphs. 4 Table of Contents 1 Definitions 7 2 Identification -

Cadmium Sulfide
SIGMA-ALDRICH sigma-aldrich.com SAFETY DATA SHEET Version 6.1 Revision Date 05/28/2017 Print Date 08/08/2019 1. PRODUCT AND COMPANY IDENTIFICATION 1.1 Product identifiers Product name : Cadmium sulfide Product Number : 208183 Brand : Aldrich Index-No. : 048-010-00-4 CAS-No. : 1306-23-6 1.2 Relevant identified uses of the substance or mixture and uses advised against Identified uses : Laboratory chemicals, Synthesis of substances 1.3 Details of the supplier of the safety data sheet Company : Sigma-Aldrich Inc. 3050 Spruce Street ST. LOUIS MO 63103 UNITED STATES Telephone : +1 314 771-5765 Fax : +1 800 325-5052 1.4 Emergency telephone number Emergency Phone # : +1-703-527-3887 2. HAZARDS IDENTIFICATION 2.1 Classification of the substance or mixture GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) Acute toxicity, Oral (Category 4), H302 Germ cell mutagenicity (Category 2), H341 Carcinogenicity (Category 1B), H350 Reproductive toxicity (Category 2), H361 Specific target organ toxicity - repeated exposure (Category 1), H372 Acute aquatic toxicity (Category 1), H400 Chronic aquatic toxicity (Category 1), H410 For the full text of the H-Statements mentioned in this Section, see Section 16. 2.2 GHS Label elements, including precautionary statements Pictogram Signal word Danger Hazard statement(s) H302 Harmful if swallowed. H341 Suspected of causing genetic defects. H350 May cause cancer. Aldrich- 208183 Page 1 of 9 H361 Suspected of damaging fertility or the unborn child. H372 Causes damage to organs through prolonged or repeated exposure. H410 Very toxic to aquatic life with long lasting effects. Precautionary statement(s) P201 Obtain special instructions before use.