Chemical Safety and Waste Management Manual
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
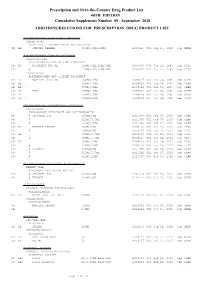
Additions and Deletions to the Drug Product List
Prescription and Over-the-Counter Drug Product List 40TH EDITION Cumulative Supplement Number 09 : September 2020 ADDITIONS/DELETIONS FOR PRESCRIPTION DRUG PRODUCT LIST ACETAMINOPHEN; BUTALBITAL; CAFFEINE TABLET;ORAL BUTALBITAL, ACETAMINOPHEN AND CAFFEINE >A> AA STRIDES PHARMA 325MG;50MG;40MG A 203647 001 Sep 21, 2020 Sep NEWA ACETAMINOPHEN; CODEINE PHOSPHATE SOLUTION;ORAL ACETAMINOPHEN AND CODEINE PHOSPHATE >D> AA WOCKHARDT BIO AG 120MG/5ML;12MG/5ML A 087006 001 Jul 22, 1981 Sep DISC >A> @ 120MG/5ML;12MG/5ML A 087006 001 Jul 22, 1981 Sep DISC TABLET;ORAL ACETAMINOPHEN AND CODEINE PHOSPHATE >A> AA NOSTRUM LABS INC 300MG;15MG A 088627 001 Mar 06, 1985 Sep CAHN >A> AA 300MG;30MG A 088628 001 Mar 06, 1985 Sep CAHN >A> AA ! 300MG;60MG A 088629 001 Mar 06, 1985 Sep CAHN >D> AA TEVA 300MG;15MG A 088627 001 Mar 06, 1985 Sep CAHN >D> AA 300MG;30MG A 088628 001 Mar 06, 1985 Sep CAHN >D> AA ! 300MG;60MG A 088629 001 Mar 06, 1985 Sep CAHN ACETAMINOPHEN; HYDROCODONE BITARTRATE TABLET;ORAL HYDROCODONE BITARTRATE AND ACETAMINOPHEN >A> @ CEROVENE INC 325MG;5MG A 211690 001 Feb 07, 2020 Sep CAHN >A> @ 325MG;7.5MG A 211690 002 Feb 07, 2020 Sep CAHN >A> @ 325MG;10MG A 211690 003 Feb 07, 2020 Sep CAHN >D> AA VINTAGE PHARMS 300MG;5MG A 090415 001 Jan 24, 2011 Sep DISC >A> @ 300MG;5MG A 090415 001 Jan 24, 2011 Sep DISC >D> AA 300MG;7.5MG A 090415 002 Jan 24, 2011 Sep DISC >A> @ 300MG;7.5MG A 090415 002 Jan 24, 2011 Sep DISC >D> AA 300MG;10MG A 090415 003 Jan 24, 2011 Sep DISC >A> @ 300MG;10MG A 090415 003 Jan 24, 2011 Sep DISC >D> @ XIROMED 325MG;5MG A 211690 -

Dudley Chemical Corporation
Dudley Chemical 125 Kenyon Drive Lakewood NJ 08701 Corporation (732) 886-3100 • Fax: 732-886-3688 Indicators Code Product CAS Number 02793 Alizarin Complexone 2 Hydrate 3952-78-1 02861 Alizarin Yellow GG 584-42-9 08137 Bromochlorophenol Blue Sodium Salt 102185-52-4 08142 Bromocresol Green Free Acid ACS 76-60-8 08146 Bromocresol Green Sodium Salt ACS 62625-32-5 08152 Bromocresol Purple Free Acid ACS 115-40-2 08156 Bromocresol Purple Sodium Salt ACS 62625-30-3 08162 Bromophenol Blue Free Acid ACS 115-39-9 08166 Bromophenol Blue Sodium Salt ACS 62625-28-9 08168 Bromopyrogallol Red 16574-43-9 08172 Bromothymol Blue Free Acid ACS 76-59-5 08176 Bromothymol Blue Sodium Salt ACS 34722-90-2 08440 Bromophenol Red Free Acid 2800-80-8 08443 Bromophenol Red Sodium Salt 102185-50-2 08490 Bromoxylenol Blue 40070-59-5 09268 Calconcarboxylic Acid 3737-95-9 09330 Calmagite 3147-14-6 09798 Chlorophenol Red 4430-20-0 09801 Chlorophenol Red 4430-20-0 09803 Chlorophenol Red Sodium Salt 123333-64-2 11100 Congo Red 573-58-0 12100 o-Cresolphthalein 596-27-0 12103 o-Cresolphthalein Complexone 2411-89-4 12106 o-Cresolphthalein Compexone Sodium Salt 94442-10-1 12131 Meta-Cresol Purple Free Acid 2303-01-7 12135 meta-Cresol Purple W/S 62625-31-4 12152 Cresol Red Free acid 1733-12-6 12157 Cresol Red Sodium Salt 62625-29-0 20320 Eriochrome Blue Black R 2538-85-4 20507 Ethyl Orange Sodium Salt 62758-12-7 20582 Ethyl Red Indicator 76058-33-8 21140 Fast Sulphon Black F 3682-47-1 Dudley Chemical Corporation • Indicators, Page 1 of 5 23701 Fluorexon / Calcein 1461-15-0 34024 -

Cadmium- Toxfaqs™
Cadmium- ToxFAQs™ CAS # 7440-43-9 This fact sheet answers the most frequently asked health questions (FAQs) about cadmium. For more information, call the CDC Information Center at 1-800-232-4636. This fact sheet is one in a series of summaries about hazardous substances and their health effects. It is important you understand this information because this substance may harm you. The effects of exposure to any hazardous substance depend on the dose, the duration, how you are exposed, personal traits and habits, and whether other chemicals are present. HIGHLIGHTS: Exposure to cadmium happens mostly in the workplace where cadmium products are made. The general population is exposed from breathing cigarette smoke or eating cadmium contaminated foods. Cadmium damages the kidneys, lungs, and bones. Cadmium has been found in at least 1,014 of the 1,669 National Priorities List (NPL) sites identified by the Environmental Protection Agency (EPA). What is cadmium? How might I be exposed to cadmium? Cadmium is a natural element in the earth’s crust. It is • Eating foods containing cadmium; low levels are usually found as a mineral combined with other elements found in all foods (highest levels are found in leafy such as oxygen (cadmium oxide), chlorine (cadmium vegetables, grains, legumes, and kidney meat). chloride), or sulfur (cadmium sulfate, cadmium sulfide). • Smoking cigarettes or breathing cigarette smoke. All soils and rocks, including coal and mineral fertilizers, • Breathing contaminated workplace air. contain some cadmium. Most cadmium used in the United States is extracted during the production of other metals • Drinking contaminated water. like zinc, lead, and copper. -

Neonatal Intensive Care Drug Therapy Update: a Bibliography
LWW/JPNN AS310-13 July 28, 2004 23:11 Char Count= 0 J Perinat Neonat Nurs Vol. 18, No. 3, pp. 292–306 c 2004 Lippincott Williams & Wilkins, Inc. Neonatal Intensive Care Drug Therapy Update: A Bibliography Jason Sauberan, PharmD BIBLIOGRAPHY I. Overview A. Clark RH, Bloom BT, Gerstmann DR Medications Used in Neonatal Intensive Care Units—A Descriptive Study [abstract 3047]. In: Program and abstracts of the 2004 Pediatric Academic Societies’ Annual Meeting, San Francisco, CA. B. Barr J, Brenner-Zada G, Heiman E, Pareth G, Bulkowstein M, Greenberg R, Berkovitch M. Unlicensed and off-label medication use in a neonatal intensive care unit: a prospective study. Am J Perinatol. 2002 Feb;19(2):67–72. C. O’Donnell CP, Stone RJ, Morley CJ. Unlicensed and off-label drug use in an Australian neonatal intensive care unit. Pediatrics. 2002 Nov;110(5):e52. D. Committee on Drugs. American Academy of Pediatrics. Uses of drugs not described in the package insert (off-label uses). Pediatrics. 2002 Jul;110(1 Pt 1):181–3. II. Anti-infectives A. Linezolid 1. Deville JG, Adler S, Azimi PH, Jantausch BA, Morfin MR, Beltran S, Edge-Padbury B, Naberhuis-Stehouwer S, Bruss JB. Linezolid versus vancomycin in the treatment of known or suspected resistant gram-positive infections in neonates. Pediatr Infect Dis J. 2003 Sep;22(9 Suppl):S158–63. 2. Vo M, Cirincione BB, Rubino CM, Jungbluth GL. Pharmacokinetics of Linezolid in Neonates and Young Infants [abstract A-1409]. In: Program and abstracts of the 42nd Interscience Conference on Antimicrobial Agents and Chemotherapy, San Diego, CA. -

The Preparation and Reactions of Certain Quarternary Ammonium And
THE PREPARATION AND REACTIONS OF CERTAIN QUATERNARY AMMONIUM AND RELATED COMPOUNDS By CARL L MICHAELIS A DISSERTATION PRESENTED TO THE GRADUATE COUNCIL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY UNIVERSITY OF FLORIDA August, 1953 ACKNOWLEDGMENTS The author wishes to express his sincere appreci- ation for the valuable assistance and inspiring direction of Dr. George B. Butler. Special thanks are due Dr. and Mrs. Armin Gropp for the help and encouragement in carrying out this investi- gation. The author also wishes to express his appreciation for the assistance and advice so readily given him by his fellow students, graduate and undergraduate alike. \ ii TABLE OF CONTENTS Page ACKNOWLEDGMENTS ii TABLE OF CONTENTS iii INTRODUCTION 1 A. Review of Literature and Statement of Problem 1 EXPERIMENTAL 14 A. General Discussion 14 B. Procedure 18 DISCUSSION . 51 SUMMARY 62 BIBLIOGRAPHY 64 BIOGRAPHICAL ITEMS 66 COMMITTEE REPORT 67 iii INTRODUCTION The need for compounds of good adrenergic blooking activity has been the object of extensive investigation in recent years. In order to get a better idea of the nature of adrenergic blocking activity, it might be best to under- stand first how an adrenergic compound functions. Certain compounds like epinephrine or adrenaline, are powerful agents in stimulating the sympathetic nervous system, that part of the nervous system springing from the thoracic lumbar cord. The substance epinephrine is much like, if not identical to, the substance called sympathin, which is produced at the ending of a nerve in the smooth muscle or gland when a nerve is stimulated. -

Chromatographic Separation of Alkaline Earth Metals Using Alpha-Hydroxyisobutyric Acid
AN ABSTRACT OF THE THESIS OF JOHN ARTHUR HAUSCHILD for the MASTER OF SCIENCE (Name) (Degree) in CHEMISTRY (ANALYTICAL) presented on (Major) Title: CHROMATOGRAPHIC SEPARATION OF ALKALINE EARTH METALS USING ALPHA-HYDROXYISOBUYRIC ACID Abstract approved: Redacted for Privacy Max B. Williams A systematic study of the elution of magnesium and calcium from Dowex 50 X 8 resin using a-hydroxyisobutyric acid (a-HIBA) at various pH values and concentrations, indicated that the difference in the equilibrium distribution coefficients of these two elements was large enough for a good separation.This fact was applied to develop a chromatographic procedure for the separationof milligram quantities of magnesium, calcium, strontium, and barium.After magnesium was eluted with 0. 22M a-HIBA at pH 4. 5, thethree remaining elements were eluted by varying the concentration and pH of a-HIBAduring the course of the elution (exponential gradient elution).After its respec- tive elution, each alkaline earth metal was directly determined by atomic absorption spectroscopy.Using this method, several success- ful analyses of synthetic samples (similar to the composition of sea water) were performed.Yield determinations of the alkaline earth metals from these analyses were consistently greater than 93%, with the overall average yield being 98%. Chromatographic Separation of Alkaline Earth Metals Using Alpha-Hydroxyisobutyric Acid by John Arthur Haus child A THESIS submitted to Oregon State University in partial fulfillment of the requirements for the degree of Master -

PRICELIST-1920-FINAL.Pdf
INDEX Page No. MD Speech 01 Our Vision / Our Mission 02 Product Classification and Grade Information 03 Label Information 04 GHS Compliance 05 Technical Data Sheet and COA 06 Qualikems Product Range 07 ISO Certificate 08 - 09 Company Details 10 Ordering Information 11 Terms & Conditions 12 Rate List 13 - 52 Images of Lab / Plant / R & D 53 - 58 Rate List 59 -116 BELIEVING yourselfIN IS THE FIRST SECRET TO Success Dear Reader, The document you are holding is the result of work performed by the team of professionals of QUALIKEMS. It is the fruit of our teams extensive technical experience combine with the collaboration of our customers, who have offered us their valuable comments and proposals for improvement. At Qualikems, we have been working and investing for many years with our thoughts focused on the long term. Only thus can this comprehensive catalogue be kept up to date with the products you need. Our highly trained workforce, using state of the art technology, is the driving force behind the management of our modern factory, and our principal aim is to guarantee that the QUALIKEMS product range meets the conditions you require. QUALIKEMS reinforces industrial character and the path to progress we have continuously forged over the years. This path requires the responsible use of resources and the sustainability of our business activity. It is likewise requires and ability to keep on growing as the way to earn and to preserve our status as the leading supplier of laboratory reagents to our Clients Ashok Sahni Managing Director QUALIKEMS FINE CHEM PVT. -

162 Part 175—Indirect Food Addi
§ 174.6 21 CFR Ch. I (4–1–19 Edition) (c) The existence in this subchapter B Subpart B—Substances for Use Only as of a regulation prescribing safe condi- Components of Adhesives tions for the use of a substance as an Sec. article or component of articles that 175.105 Adhesives. contact food shall not be construed as 175.125 Pressure-sensitive adhesives. implying that such substance may be safely used as a direct additive in food. Subpart C—Substances for Use as (d) Substances that under conditions Components of Coatings of good manufacturing practice may be 175.210 Acrylate ester copolymer coating. safely used as components of articles 175.230 Hot-melt strippable food coatings. that contact food include the fol- 175.250 Paraffin (synthetic). lowing, subject to any prescribed limi- 175.260 Partial phosphoric acid esters of pol- yester resins. tations: 175.270 Poly(vinyl fluoride) resins. (1) Substances generally recognized 175.300 Resinous and polymeric coatings. as safe in or on food. 175.320 Resinous and polymeric coatings for (2) Substances generally recognized polyolefin films. as safe for their intended use in food 175.350 Vinyl acetate/crotonic acid copoly- mer. packaging. 175.360 Vinylidene chloride copolymer coat- (3) Substances used in accordance ings for nylon film. with a prior sanction or approval. 175.365 Vinylidene chloride copolymer coat- (4) Substances permitted for use by ings for polycarbonate film. 175.380 Xylene-formaldehyde resins con- regulations in this part and parts 175, densed with 4,4′-isopropylidenediphenol- 176, 177, 178 and § 179.45 of this chapter. -

268 Part 522—Implantation Or Injectable Dosage Form
§ 520.2645 21 CFR Ch. I (4–1–18 Edition) (ii) Indications for use. For the control 522.82 Aminopropazine. of American foulbrood (Paenibacillus 522.84 Beta-aminopropionitrile. larvae). 522.88 Amoxicillin. 522.90 Ampicillin injectable dosage forms. (iii) Limitations. The drug should be 522.90a Ampicillin trihydrate suspension. fed early in the spring or fall and con- 522.90b Ampicillin trihydrate powder for in- sumed by the bees before the main jection. honey flow begins, to avoid contamina- 522.90c Ampicillin sodium. tion of production honey. Complete 522.144 Arsenamide. treatments at least 4 weeks before 522.147 Atipamezole. main honey flow. 522.150 Azaperone. 522.161 Betamethasone. [40 FR 13838, Mar. 27, 1975, as amended at 50 522.163 Betamethasone dipropionate and FR 49841, Dec. 5, 1985; 59 FR 14365, Mar. 28, betamethasone sodium phosphate aque- 1994; 62 FR 39443, July 23, 1997; 68 FR 24879, ous suspension. May 9, 2003; 70 FR 69439, Nov. 16, 2005; 73 FR 522.167 Betamethasone sodium phosphate 76946, Dec. 18, 2008; 75 FR 76259, Dec. 8, 2010; and betamethasone acetate. 76 FR 59024, Sept. 23, 2011; 77 FR 29217, May 522.204 Boldenone. 17, 2012; 79 FR 37620, July 2, 2014; 79 FR 53136, 522.224 Bupivacaine. Sept. 8, 2014; 79 FR 64116, Oct. 28, 2014; 80 FR 522.230 Buprenorphine. 34278, June 16, 2015; 81 FR 48702, July 26, 2016] 522.234 Butamisole. 522.246 Butorphanol. § 520.2645 Tylvalosin. 522.275 N-Butylscopolammonium. 522.300 Carfentanil. (a) Specifications. Granules containing 522.304 Carprofen. 62.5 percent tylvalosin (w/w) as 522.311 Cefovecin. -

Review Article
REVIEW ARTICLE COLLAGEN METABOLISM COLLAGEN METABOLISM Types of Collagen 228 Structure of Collagen Molecules 230 Synthesis and Processing of Procollagen Polypeptides 232 Transcription and Translation 233 Posttranslational Modifications 233 Extracellular Processing of Procollagen and Collagen Fibrillogenesis 240 Functions of Collagen in Connective rissue 243 Collagen Degradation 245 Regulation of the Metabolism of Collagen 246 Heritable Diseases of Collagen 247 Recessive Dermatosparaxis 248 Recessive Forms of EDS 251 EDS VI 251 EDS VII 252 EDS V 252 Lysyl Oxidase Deficiency in the Mouse 253 X-Linked Cutis Laxa 253 Menke's Kinky Hair Syndrome 253 Homocystinuria 254 EDS IV 254 Dominant Forms of EDS 254 Dominant Collagen Packing Defect I 255 Dominant and Recessive Forms of Osteogenesis Imperfecta 258 Dominant and Recessive Forms of Cutis Laxa 258 The Marfan Syndrome 259 Acquired Diseases and Repair Processes Affecting Collagen 259 Acquired Changes in the Types of Collagen Synthesis 260 Acquired Changes in Amounts of Collagen Synthesized 263 Acquired Changes in Hydroxylation of Proline and Lysine 264 Acquired Changes in Collagen Cross-Links 265 Acquired Defects in Collagen Degradation 267 Conclusion 267 Bibliography 267 Collagen Metabolism A Comparison of Diseases of Collagen and Diseases Affecting Collagen Ronald R. Minor, VMD, PhD COLLAGEN CONSTITUTES approximately one third of the body's total protein, and changes in synthesis and/or degradation of colla- gen occur in nearly every disease process. There are also a number of newly described specific diseases of collagen in both man and domestic animals. Thus, an understanding of the synthesis, deposition, and turnover of collagen is important for the pathologist, the clinician, and the basic scientist alike. -

Industrial Applications of Nano Cu-Chromite Catalyzer
International Journal of Chemical Studies 2018; 6(4): 86-90 P-ISSN: 2349–8528 E-ISSN: 2321–4902 IJCS 2018; 6(4): 86-90 Industrial applications of nano Cu-Chromite © 2018 IJCS Received: 26-05-2018 catalyzer Accepted: 29-06-2018 Pouresmaeily Seyed Mohammad Pouresmaeily Seyed Mohammad, H Shirkavand Behzad, Shahidzadeh Department of Chemistry & Mansour and Ebrahimi Sobhan Chemical Engineering Malek-e- Ashtar University of Technology, Shabanlou, Iran Abstract Nano Cu-Chromite catalyzer is a high usage catalyzer in chemical reactions. This catalyzer has many H Shirkavand Behzad applications in oxidation, hydrogenation, dehydrogenation, alkylation, alcohol decomposition, useful Department of Chemistry & reactions in petroleum include: reforming. Recyclable is one of advantages of this catalyzer. This Chemical Engineering Malek-e- catalyzer produces by sol-gel, co-precipitating, deposition in vacuum and ceramic methods. This paper Ashtar University of explores applications of this catalyzer in various industries. Technology, Shabanlou, Iran Shahidzadeh Mansour Keywords: Nano Cu-Chromite, hydrogen producing, nano catalyzer Institute for Color Science & Technology, Shams, Iran Introduction Cu-Chromite is an important catalyzer in chemical reactions that has wide application in Ebrahimi Sobhan synthesis of organic chemistry processes. Uses in many reactions such as: hydrogenation, Department of Material Science and Engineering, Sharif dehydrogenation, hydrogenolysis, oxidation, alkylation, cycloaddition, as catalyzer for University of Technology, -

Simultaneous Removal of Calconcarboxylic Acid, + 3− NH4 and PO4 from Pharmaceutical Effluent Using Iron Oxide-Biochar Nanocomposite Loaded with Pseudomonas Putida
processes Article Simultaneous Removal of Calconcarboxylic Acid, + 3− NH4 and PO4 from Pharmaceutical Effluent Using Iron Oxide-Biochar Nanocomposite Loaded with Pseudomonas putida Saifeldin M. Siddeeg 1,2 , Mohamed A. Tahoon 1 and Faouzi Ben Rebah 1,3,* 1 Department of Chemistry, College of Science, King Khalid University, P.O. Box 9004, Abha 61413, Kingdom of Saudi Arabia; [email protected] (S.M.S.); [email protected] (M.A.T.) 2 Chemistry and Nuclear Physics Institute, Atomic Energy Commission, P.O. Box 3001, Khartoum 11111, Sudan 3 Higher Institute of Biotechnology of Sfax (ISBS), Sfax University, P.O. Box 263, Sfax 3000, Tunisia * Correspondence: [email protected] Received: 28 September 2019; Accepted: 23 October 2019; Published: 3 November 2019 Abstract: In the current study, the Fe2O3/biochar nanocomposite was synthesized through a self-assembly method, followed by the immobilization of Pseudomonas putida (P. putida) on its surface to produce the P. putida/Fe2O3/biochar magnetic innovative nanocomposite. The synthesized nanocomposite was characterized using different techniques including X-ray diffraction, transmission electron microscopy (TEM), scanning electron microscopy (SEM), and Fourier-transform infrared spectroscopy (FT-IR). Then, the efficiencies of this material to + remove calconcarboxylic acid (CCA) organic dye, ammonium ions (NH4 ), and phosphate ions 3 (PO4 −) from industrial wastewater were analyzed. The removal rates of up to 82%, 95%, and 85% 3 + were achieved for CCA dye, PO4 −, NH4 , respectively, by the synthesized composite. Interestingly, even after 5 cycles of reuse, the prepared nanocomposite remains efficient in the removal of pollutants. Therefore, the P. putida/Fe3O4/biochar composite was found to be an actual talented nanocomposite for industrial wastewater bioremediation.