Assessment of Bycatch in Gill Net Fisheries
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

D13 the Marine Environment Development Consent Order
ENERGY WORKING FOR BRITAIN FOR WORKING ENERGY Wylfa Newydd Project 6.4.13 ES Volume D - WNDA Development D13 - The marine environment PINS Reference Number: EN010007 Application Reference Number: 6.4.13 June 2018 Revision 1.0 Regulation Number: 5(2)(a) Planning Act 2008 Infrastructure Planning (Applications: Prescribed Forms and Procedure) Regulations 2009 Horizon Internal DCRM Number: WN0902-JAC-PAC-CHT-00045 [This page is intentionally blank] Contents 13 The marine environment .................................................................................. 1 13.1 Introduction ...................................................................................................... 1 13.2 Study area ....................................................................................................... 1 13.3 Wylfa Newydd Development Area baseline environment ................................ 2 Conservation designations .............................................................................. 3 Water quality .................................................................................................... 9 Sediment quality ............................................................................................ 13 Phytoplankton and zooplankton ..................................................................... 15 Marine benthic habitats and species .............................................................. 18 Marine fish .................................................................................................... -

Fishes of Terengganu East Coast of Malay Peninsula, Malaysia Ii Iii
i Fishes of Terengganu East coast of Malay Peninsula, Malaysia ii iii Edited by Mizuki Matsunuma, Hiroyuki Motomura, Keiichi Matsuura, Noor Azhar M. Shazili and Mohd Azmi Ambak Photographed by Masatoshi Meguro and Mizuki Matsunuma iv Copy Right © 2011 by the National Museum of Nature and Science, Universiti Malaysia Terengganu and Kagoshima University Museum All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means without prior written permission from the publisher. Copyrights of the specimen photographs are held by the Kagoshima Uni- versity Museum. For bibliographic purposes this book should be cited as follows: Matsunuma, M., H. Motomura, K. Matsuura, N. A. M. Shazili and M. A. Ambak (eds.). 2011 (Nov.). Fishes of Terengganu – east coast of Malay Peninsula, Malaysia. National Museum of Nature and Science, Universiti Malaysia Terengganu and Kagoshima University Museum, ix + 251 pages. ISBN 978-4-87803-036-9 Corresponding editor: Hiroyuki Motomura (e-mail: [email protected]) v Preface Tropical seas in Southeast Asian countries are well known for their rich fish diversity found in various environments such as beautiful coral reefs, mud flats, sandy beaches, mangroves, and estuaries around river mouths. The South China Sea is a major water body containing a large and diverse fish fauna. However, many areas of the South China Sea, particularly in Malaysia and Vietnam, have been poorly studied in terms of fish taxonomy and diversity. Local fish scientists and students have frequently faced difficulty when try- ing to identify fishes in their home countries. During the International Training Program of the Japan Society for Promotion of Science (ITP of JSPS), two graduate students of Kagoshima University, Mr. -

MSC Sustainable Fisheries Certification
FOOD CERTIFICATION INTERNATIONAL LIMITED Findhorn House, Dochfour Business Centre, Dochgarroch, Inverness, IV3 8GY, Scotland, UK Tel: 00 44 (0) 1463 223 039 Fax: 00 44 (0) 1463 246 380 www.foodcertint.com MSC Sustainable Fisheries Certification Public Comment Draft Report for the Irish Pelagic Sustainability Group (IPSG) Western mackerel ( Scomber scombrus ) pelagic trawl fishery JUNE 2009 prepared for: Irish Pelagic Sustainability Group (IPSG) by: Food Certification International Ltd A company incorporated in Scotland No SC313289. Registered address as above IPSG Western mackerel pelagic trawl fishery June 2009 Authors: T. Southall, P. Medley, N. Pfeiffer, M. Gill, M. McFadden Certification Body: Client Contact: Food Certification International Ltd Irish Pelagic Sustainability Group Findhorn House Bruach na Mara Dochfour Business Centre St Catherine’s Road Dochgarroch Killybegs Inverness Co. Donegal Scotland Eire IV3 8GY UK T: +44(0) 1463 223 039 T: +353 74 973 1089 E: [email protected] E: [email protected] W: www.foodcertint.com W: www.kfo.ie Food Certification International Public Comment Draft Report IPSG Western mackerel pelagic trawl fishery Contents 1. Introduction.........................................................................................................................................................1 1.1 Scope. .............................................................................................................................................................1 1.2 Report structure...............................................................................................................................................1 -

Training Manual Series No.15/2018
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by CMFRI Digital Repository DBTR-H D Indian Council of Agricultural Research Ministry of Science and Technology Central Marine Fisheries Research Institute Department of Biotechnology CMFRI Training Manual Series No.15/2018 Training Manual In the frame work of the project: DBT sponsored Three Months National Training in Molecular Biology and Biotechnology for Fisheries Professionals 2015-18 Training Manual In the frame work of the project: DBT sponsored Three Months National Training in Molecular Biology and Biotechnology for Fisheries Professionals 2015-18 Training Manual This is a limited edition of the CMFRI Training Manual provided to participants of the “DBT sponsored Three Months National Training in Molecular Biology and Biotechnology for Fisheries Professionals” organized by the Marine Biotechnology Division of Central Marine Fisheries Research Institute (CMFRI), from 2nd February 2015 - 31st March 2018. Principal Investigator Dr. P. Vijayagopal Compiled & Edited by Dr. P. Vijayagopal Dr. Reynold Peter Assisted by Aditya Prabhakar Swetha Dhamodharan P V ISBN 978-93-82263-24-1 CMFRI Training Manual Series No.15/2018 Published by Dr A Gopalakrishnan Director, Central Marine Fisheries Research Institute (ICAR-CMFRI) Central Marine Fisheries Research Institute PB.No:1603, Ernakulam North P.O, Kochi-682018, India. 2 Foreword Central Marine Fisheries Research Institute (CMFRI), Kochi along with CIFE, Mumbai and CIFA, Bhubaneswar within the Indian Council of Agricultural Research (ICAR) and Department of Biotechnology of Government of India organized a series of training programs entitled “DBT sponsored Three Months National Training in Molecular Biology and Biotechnology for Fisheries Professionals”. -

Record Sized Alepes Vari and Nematalosa Nasus
Mar. Fish. Infor. Serv., T & E Ser., No. 231, 2017 27 Record sized Alepes vari and Nematalosa nasus Subal Kumar Roul, N. Ragesh, T. B. Retheesh, D. Prakasan and A. R. Akhil ICAR-Central Marine Fisheries Research Institute, Kochi e-mail: [email protected] The herring scad, Alepes vari is a common pelagic Nematalosa nasus (Bloch’s gizzard shad), is a fish species in the family carangidae. The species small pelagic mostly captured with ringseines and is the largest fish of the scad genus Alepes and occasionally found in the catches of pelagic trawls maximum length so far recorded all over the world and gill nets. During a regular field visit, on 25th oceans is 560 mm TL (Froese & Pauly, FishBase 2016, October 2016, an unusually large specimen of http://www.fishbase.org.). During a regular field N. nasus (241 mm total length (TL) and 171.5 g total visit, an unusually large specimen of A. vari was weight (TW) was collected from a chinese dip net, recorded from Kalamukku Landing Centre, Kerala operated at Fort Kochi (Fig. 2). The specimen was on 25th May, 2016. This was a male individual deposited in the collections of Marine Biodiversity measuring 600 mm in total length (TL) and weighing Museum of CMFRI, (accession number, GB 1.6 kg (Fig. 1)and was caught by hook and line. Young 13.2.37.6). The largest record of N. nasus (252 mm ones occasionally occur in pelagic trawls and larger TL and 190 g TW) was reported from Chilka Lagoon, ones rarely occur in gill net and hook and line Odisha (Panda et al., 2016 J. -

Visakhapatnam-Chennai Industrial Corridor Development Program-Project 1
Initial Environmental Examination _________________________________________________________________________ Project Number: 48434-003 Grant Number: 0495-IND October 2018 IND: Visakhapatnam-Chennai Industrial Corridor Development Program-Project 1 Mudasarlova Lake Development Project Prepared by Greater Visakhapatnam Municipal Corporation, Government of Andhra Pradesh for the Asian Development Bank. CURRENCY EQUIVALENTS (as of 11 October 2018) Currency Unit = Indian rupees (₹) ₹1.00 = $0.0135 $1.00 = ₹73.785 ABBREVIATIONS ADB – Asian Development Bank ASI – Archeological Survey of India CAPP – community awareness and public participation CFE – consent for establishment CFO – consent for operation GVMC – Greater Visakhapatnam Municipal Corporation DoF – Department of Forest DoL – Department of Labour EAC – Expert Appraisal Committee EARF – environmental assessment and review framework EIA – environmental impact assessment EMP – environmental management plan GRM – grievance redress mechanism IEE – initial environmental examination LARRA – land acquisition, rehabilitation and resettlement authority MFF – multi-tranche financing facility MoEFCC – Ministry of Environment and Forest, Climate Change NGO – non-governmental organization NGT – National Green Tribunal GVMC – Greater Visakhapatnam Municipal Corporation PMC – project management consultancy PMU – program management unit PPTA – project preparatory technical assistance ROW – right of way SPS – Safeguard Policy Statement TOR – terms of reference UGR – underground service reservoir WTP – water treatment plant WEIGHTS AND MEASURES m3 – cubic meter m3/h – cubic meter per hour cm – centimeter dBA – decibel audible °C – degree Celsius ha – hectare km – kilometer m – meter mm – millimeter mm/hr – millimeters per hour km2 – square kilometer m2 – square meter NOTES (i) The fiscal year (FY) of the Government of India and its agencies ends on 31 March. (ii) In this report, "$" refers to United States dollars. This initial environmental examination is a document of the borrower. -

(W. Indian Ocean) Alectis Indicus (Rüppell, 1830) OTHER
click for previous page CARAN Alec 1 1983 FAO SPECIES IDENTIFICATION SHEETS FAMILY: CARANGIDAE FISHING AREA 51 (W. Indian Ocean) Alectis indicus (Rüppell, 1830) OTHER SCIENTIFIC NAMES STILL IN USE: None VERNACULAR NAMES: FAO : En - Indian threadfish Fr - Cordonnier plume Sp - Pámpano Indico NATIONAL: DISTINCTIVE CHARACTERS: Body deep, becoming more elongate with growth, and very compressed. Profile of nape and head somewhat angular; suborbital depth relatively broad, contained 0.8 to 1.0 times in upper jaw length; upper jaw ending slightly before eye (to below anterior margin of eye in young); both jaws with bands of villiform teeth, becoming obsolete with age; gillrakers (excluding rudiments) 8 to 11 upper, 21 to 26 lower and 29 to 37 total on first gill arch. Dorsal fin with 6 short spines (embedded and not apparent at about 17 cm fork length followed by 1 spine and 18 to 20 soft rays; anal fin with 2 spines (embedded and not apparent with growth) followed by 1 spine and 15 to 17 soft rays; anterior soft rays of dorsal and anal fins extremely long and filamentous in young; pectoral fins falcate, longer than, head; pelvic fins elongate in young. Lateral line anteriorly with a strong and moderately long arch, its posterior (straight) part with 6 plus 11 scutes; body superficially naked, scales minute and embedded where present. Vertebrae 10 + 14; supraoccipital and distal ends of predorsal bones hyperossified and enlarged distally in specimens larger than 25 cm fork length. (young) Colour: mostly silvery with a dusky green tinge dorsally; juveniles with dark bars on body; second dorsal fin pale with leading edges and tips of rays dusky; anal fin pale yellow and caudal fin dusky green. -

National Report on the Fish Stocks and Habitats of Regional, Global
United Nations UNEP/GEF South China Sea Global Environment Environment Programme Project Facility NATIONAL REPORT on The Fish Stocks and Habitats of Regional, Global, and Transboundary Significance in the South China Sea THAILAND Mr. Pirochana Saikliang Focal Point for Fisheries Chumphon Marine Fisheries Research and Development Center 408 Moo 8, Paknum Sub-District, Muang District, Chumphon 86120, Thailand NATIONAL REPORT ON FISHERIES – THAILAND Table of Contents 1. MARINE FISHERIES DEVELOPMENT........................................................................................2 / 1.1 OVERVIEW OF THE FISHERIES SECTOR ...................................................................................2 1.1.1 Total catch by fishing area, port of landing or province (by species/species group).7 1.1.2 Fishing effort by gear (no. of fishing days, or no. of boats) .......................................7 1.1.2.1 Trawl ...........................................................................................................10 1.1.2.2 Purse seine/ring net....................................................................................10 1.1.2.3 Gill net.........................................................................................................12 1.1.2.4 Other gears.................................................................................................12 1.1.3 Economic value of catch..........................................................................................14 1.1.4 Importance of the fisheries sector -

S42 AW 05 OSMOSE-WFS.Pdf
Estimating age- and size-specific natural mortality rates for Gulf of Mexico red grouper (Epinephelus morio) using the ecosystem model OSMOSE-WFS A. Grüss, M. J. Schirripa, D. Chagaris, P. Verley, Y.-J. Shin, L. Velez, C. H. Ainsworth, S. R. Sagarese, and L. Lombardi-Carlson SEDAR42-AW-05 11 March 2015 This information is distributed solely for the purpose of pre-dissemination peer review. It does not represent and should not be construed to represent any agency determination or policy. Please cite this document as: Grüss, A., M. J. Schirripa, D. Chagaris, P. Verley, Y.-J. Shin, L. Velez, C. H. Ainsworth, S. R. Sagarese, and L. Lombardi-Carlson. 2015. Estimating age- and size-specific natural mortality rates for Gulf of Mexico red grouper (Epinephelus morio) using the ecosystem model OSMOSE- WFS. SEDAR42-AW-05. SEDAR, North Charleston, SC. 33 pp. SEDAR42-AW-05 Estimating age- and size-specific natural mortality rates for Gulf of Mexico red grouper (Epinephelus morio) using the ecosystem model OSMOSE-WFS A. Grüss1,2, M. J. Schirripa2, D. Chagaris3, P. Verley4, Y.-J. Shin4,5, L. Velez4, C. H. Ainsworth6, S. R. Sagarese1,2, and L. Lombardi-Carlson7 1University of Miami, Rosenstiel School of Marine and Atmospheric Science, Cooperative Institute for Marine and Atmospheric Studies, 4600 Rickenbacker Causeway, Miami, FL, 33149, USA 2Southeast Fisheries Science Center, Sustainable Fisheries Division, 75 Virginia Beach Drive, Miami, FL, 33149-1099, USA 3Florida Fish and Wildlife Conservation Commission, Fish and Wildlife Research Institute, 100 8th Ave SE, St. Petersburg, FL 33701, USA 4Institut de Recherche pour le Développement, UMR EME 212, Centre de Recherche Halieutique Méditerranéenne et Tropicale, Avenue Jean Monnet, CS 30171, 34203 Sète cedex, France 5University of Cape Town, Marine Research Institute Ma-Re, Department of Biological Sciences, Private Bag X3, Rondebosch, Cape Town 7701, South Africa 6University of South Florida, College of Marine Science, 140 7th Avenue South, St. -
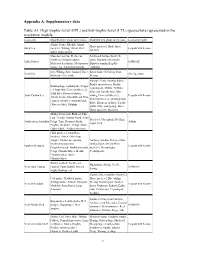
And Low-Trophic-Level (LTL) Species/Taxa Represented in the Ecosystem Models
Appendix A. Supplementary data Table A1. High-trophic-level (HTL) and low-trophic-level (LTL) species/taxa represented in the ecosystem models. Ecosystem Modelled HLT group species/taxa Modelled LTL group species/taxa Ecosystem model Atlantic bonito, Bluefish, Atlantic Horse mackerel, Shad, Sprat, Black Sea mackerel, Whiting, Turbot, Red Ecopath with Ecosim Anchovy mullet, Spiny dogfish Mustelus mustelus, Merluccius Sardina pilchardus, Sardinella merlucciu, Octopus vulgaris, aurita, Engraulis encrasicolus, Gulf of Gabes OSMOSE Melicertus kerathurus, Metapenaeus Diplodus annularisPagellus monoceros, Trachurus trachurus, erythrinus Dab, Whiting, Sole, Gurnard, Plaice, Sprat, Sandeel, Norway Pout, North Sea Size Spectrum Haddock, Cod, Saithe Herring Shrimps, Crabs, Norway lobster, Benthic invertebrates, Benthic Benthopelagic cephalopods, Conger cephalopods, Mullets, Flatfishes, eel, Anglerfish, Demersal fishes (3), Poor cod, Juvenile hake, Blue Adult hake, Demersal sharks, South Catalan Sea whiting, Demersal fishes (1), Ecopath with Ecosim Atlantic bonito, Swordfish and Tuna, Demersal fishes (2), Benthopelagic Loggerhead turtles, Audouin's gull, fishes, European anchovy, Sardine Other sea birds, Dolphins adults, Other small pelagic fishes, Horse mackerel, Mackerel Shallow Demersals, Flathead, Pink Ling, Trevalla, Gummy Shark, Small Mackerel, Myctophids, Red Bait, Southeastern Australian Pelagic Tuna, Demersal Shark, Atlantis Squid, Krill Dogfish, Grenadier, Pelagic Shark, Gulper Shark, Shallow Piscivores Chub mackerel, Adult Horse mackerel, -

North Western Waters Fisheries Ecosystem Plan
Making the European Fisheries Ecosystem Plan Operational NORTH WESTERN WATERS FISHERIES ECOSYSTEM PLAN: WORK PACKAGE 7 REPORT EC FP7 PROJECT # 212881 “....we need to make sustainability our primary goal; we need to base our management decisions strictly on science; we need to adopt an ecosystem approach that is geographically specified, adaptive and capable of balancing diverse social objectives....forgive me for borrowing a somewhat trite old saying: when the wind of change is blowing, some build walls, others build windmills.” Maria Damanaki European Commissioner for Maritime Affairs and Fisheries Brussels, 16th November 2010 Please cite as: Bloomfield, H., Nolan, C., Le Quesne, W.J.F., Raakjær, J., Christensen, A.S., Aanesen, M., Armstrong, C., Piet, G.J. & Frid, C. (2011) Fisheries Ecosystem Plan: North Western Waters. Making the European Fisheries Ecosystem Plan Operation (MEFEPO): Work Package 7 Report. http://www.liv.ac.uk/mefepo/reports-and-outputs/wp7/ EXECUTIVE SUMMARY Science based fisheries management developed late in the 19th century with a narrow focus on the dynamics of fish stocks. Now, early in the 21st century fisheries management is becoming integrated into wider environmental management. The July 2011 Green Paper on the Reform of the Common Fisheries Policy (CFP) identified the need reform European fisheries management and advocated the application of an Ecosystem Based Fisheries Management (EBFM) approach to deliver ecological, social and economic sustainability, stated an intention to move towards a longer term approach to fisheries management, and made commitments to greater stakeholder involvement in management. The Marine Strategy Framework Directive (MSFD) defines environmental objectives for European seas, based on sustainable utilisation of healthy marine ecosystems in support of sustainable development, and the Integrated Maritime Policy requires that individual sectors (e.g. -

Technology of Mackerel Fishery
DFO - Library MPO Bibliothèque III 11 1 1111 1 11 11 11 1 11 12038818 FD3('_IDD-!_cgraphy End -,ZWey oq Li%elati-Te TECHNOLOGY OF MACKEREL FISHERY Bibliography and Survey of Literature In addition to the Miscellaneous Special Publication series, the Fisheries and Marine Service, Department of the Environment publishes the Journal of the Fisheries Research Board of Canada in annual volumes of monthly issues and a Bulletin series. These publications are for sale by Information Canada, Ottawa KlA 0S9. Remittances must be in advance, payable in Canadian funds to the order of the Receiver General for Canada. Editor and Director of Scientific J. C. STEVENSON, PH.D. Information Deputy Editor J. WATSON, PH.D. Assistant Editors JOHANNA M. REINHART, M.SC. D. U. LOOK, PH.D. J. CAMP Production-Docutnentation G. J. NEVILLE MONA SMITH, B.H.SC. Department of the Environment Fisheries and Marine Service Office of the Editor, 116 Lisgar Street Ottawa, Canada KlA OH3 "1r.. '‘1%.■71WAillaiillidftele71114‘b‘ .:7114A11"et'llti4ZW1 Handling perishable goods. Landing mackerel from the inshore trap fishery. 3LMISCELLANEOUS SPECIAL PUBLICATION 30 (La version française est en préparation) Technology of Mackerel Fishery Bibliography and Survey of Literature J. R. DINGLE Department of the Environment Fisheries and Marine Service Halifax Laboratory Halifax, N.S. B3J 2R3 DEPARTMENT OF THE ENVIRONMENT Le_P■e) DAD-MISHERIES AND MARINE SERVICE. a Ottawa 1976 ui No. 30 C)Crown Copyrights reserved Available by mail from Information Canada, Ottawa KlA 0S9 and at the following Information Canada bookshops: HALIFAX c'2 1683 Barrington Street MONTREAL 640 St. Catherine Street West OTTAWA 171 Slater Street TORONTO 221 Yonge Street WINNIPEG 393 Portage Avenue VANCOUVER 800 Granville Street or through your bookseller A deposit copy of thi's publication is also available for reference in public libraries across Canada Canada: $3.00 Catalog No.