Apicomplexa 1
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

First Description of Cryptosporidium Parvum in Carrier Pigeons
Veterinary Parasitology 243 (2017) 148–150 Contents lists available at ScienceDirect Veterinary Parasitology journal homepage: www.elsevier.com/locate/vetpar Research paper First description of Cryptosporidium parvum in carrier pigeons (Columba livia) MARK Bruno César Miranda Oliveiraa, Elis Domingos Ferraria, Mariele Fernanda da Cruz Panegossia, Alex Akira Nakamuraa, Flávio Sader Corbuccia, Walter Bertequini Nagataa, Bianca Martins dos Santosb, Jancarlo Ferreira Gomesb, Marcelo Vasconcelos Meirelesa, ⁎ Giovanni Widmerc, Katia Denise Saraiva Brescianid, a Universidade Estadual Paulista (Unesp), Faculdade de Medicina Veterinária, Araçatuba, Brazil b Laboratory of Visual Informatics in Biomedical and Health, Institute of Computing, University of Campinas – UNICAMP, São Paulo, Brazil c Department of Infectious Disease & Global Health, Cummings School of Veterinary Medicine at Tufts University, North Grafton, MA, USA d Universidade Estadual Paulista (Unesp), Faculdade de Medicina Veterinária, Araçatuba. Rua Clóvis Pestana, 793, Jardim Dona Amélia, cep 16050-680, Araçatuba, São Paulo, Brazil ARTICLE INFO ABSTRACT Keywords: The carrier pigeon and the domestic pigeon are different breeds of the species Columba livia. Carrier pigeons are Birds used for recreational activities such as bird contests and exhibitions. Due to the close contact with humans, these Carrier pigeons birds may potentially represent a public health risk, since they can host and disseminate zoonotic parasites, such Columba livia as those belonging to the genus Cryptosporidium (phylum Apicomplexa). The purpose of this work was the de- Cryptosporidiosis tection by microscopic and molecular techniques of Cryptosporidium spp. oocysts in fecal samples of carrier Cryptosporidium parvum pigeons, and subsequently to sequence the 18S ribosomal RNA marker of positive samples to identify the species. Nested PCR A total of 100 fecal samples were collected individually in two pigeon breeding facilities from Formiga and Araçatuba, cities located in Minas Gerais state and São Paulo state, Brazil, respectively. -

An Intestinal Gregarine of Nothria Conchylega (Polychaeta, Onuphidae)
Journal of Invertebrate Pathology 104 (2010) 172–179 Contents lists available at ScienceDirect Journal of Invertebrate Pathology journal homepage: www.elsevier.com/locate/jip Description of Trichotokara nothriae n. gen. et sp. (Apicomplexa, Lecudinidae) – An intestinal gregarine of Nothria conchylega (Polychaeta, Onuphidae) Sonja Rueckert *, Brian S. Leander Canadian Institute for Advanced Research, Program in Integrated Microbial Biodiversity, Departments of Botany and Zoology, University of British Columbia, #3529 – 6270 University Blvd., Vancouver, BC, Canada V6T 1Z4 article info abstract Article history: The trophozoites of a novel gregarine apicomplexan, Trichotokara nothriae n. gen. et sp., were isolated and Received 12 November 2009 characterized from the intestines of the onuphid tubeworm Nothria conchylega (Polychaeta), collected at Accepted 11 March 2010 20 m depth from the North-eastern Pacific Coast. The trophozoites were 50–155 lm long with a mid-cell Available online 23 March 2010 indentation that formed two prominent bulges (anterior bulge, 14–48 lm wide; posterior bulge, 15– 55 lm wide). Scanning electron microscopy (SEM) demonstrated that approximately 400 densely packed, Keywords: longitudinal epicytic folds (5 folds/lm) inscribe the surface of the trophozoites, and a prominently elon- Alveolata gated mucron (14–60 lm long and 6–12 lm wide) was covered with hair-like projections (mean length, Apicomplexa 1.97 m; mean width, 0.2 m at the base). Although a septum occurred at the junction between the cell Lecudinidae l l Lecudina proper and the mucron in most trophozoites, light microscopy (LM) demonstrated that the cell proper Parasite extended into the core of the mucron as a thin prolongation. -

Reconstruction of the Evolutionary History of Haemosporida
Parasitology International 65 (2016) 5–11 Contents lists available at ScienceDirect Parasitology International journal homepage: www.elsevier.com/locate/parint Reconstruction of the evolutionary history of Haemosporida (Apicomplexa) based on the cyt b gene with characterization of Haemocystidium in geckos (Squamata: Gekkota) from Oman João P. Maia a,b,c,⁎, D. James Harris a,b, Salvador Carranza c a CIBIO Research Centre in Biodiversity and Genetic Resources, InBIO, Universidade do Porto, Campus Agrário de Vairão, Rua Padre Armando Quintas, N° 7, 4485-661 Vairão, Vila do Conde, Portugal b Departamento de Biologia, Faculdade de Ciências, Universidade do Porto, Rua do Campo Alegre FC4 4169-007 Porto, Portugal c Institut de Biologia Evolutiva (CSIC-Universitat Pompeu Fabra), Passeig Maritím de la Barceloneta, 37-49, 08003 Barcelona, Spain article info abstract Article history: The order Haemosporida (Apicomplexa) includes many medically important parasites. Knowledge on the diver- Received 4 April 2015 sity and distribution of Haemosporida has increased in recent years, but remains less known in reptiles and their Received in revised form 7 September 2015 taxonomy is still uncertain. Further, estimates of evolutionary relationships of this order tend to change when Accepted 10 September 2015 new genes, taxa, outgroups or alternative methodologies are used. We inferred an updated phylogeny for the Available online 12 September 2015 Cytochrome b gene (cyt b) of Haemosporida and screened a total of 80 blood smears from 17 lizard species from Oman belonging to 11 genera. The inclusion of previously underrepresented genera resulted in an alterna- Keywords: Haemoproteus tive estimate of phylogeny for Haemosporida based on the cyt b gene. -

Control of Intestinal Protozoa in Dogs and Cats
Control of Intestinal Protozoa 6 in Dogs and Cats ESCCAP Guideline 06 Second Edition – February 2018 1 ESCCAP Malvern Hills Science Park, Geraldine Road, Malvern, Worcestershire, WR14 3SZ, United Kingdom First Edition Published by ESCCAP in August 2011 Second Edition Published in February 2018 © ESCCAP 2018 All rights reserved This publication is made available subject to the condition that any redistribution or reproduction of part or all of the contents in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise is with the prior written permission of ESCCAP. This publication may only be distributed in the covers in which it is first published unless with the prior written permission of ESCCAP. A catalogue record for this publication is available from the British Library. ISBN: 978-1-907259-53-1 2 TABLE OF CONTENTS INTRODUCTION 4 1: CONSIDERATION OF PET HEALTH AND LIFESTYLE FACTORS 5 2: LIFELONG CONTROL OF MAJOR INTESTINAL PROTOZOA 6 2.1 Giardia duodenalis 6 2.2 Feline Tritrichomonas foetus (syn. T. blagburni) 8 2.3 Cystoisospora (syn. Isospora) spp. 9 2.4 Cryptosporidium spp. 11 2.5 Toxoplasma gondii 12 2.6 Neospora caninum 14 2.7 Hammondia spp. 16 2.8 Sarcocystis spp. 17 3: ENVIRONMENTAL CONTROL OF PARASITE TRANSMISSION 18 4: OWNER CONSIDERATIONS IN PREVENTING ZOONOTIC DISEASES 19 5: STAFF, PET OWNER AND COMMUNITY EDUCATION 19 APPENDIX 1 – BACKGROUND 20 APPENDIX 2 – GLOSSARY 21 FIGURES Figure 1: Toxoplasma gondii life cycle 12 Figure 2: Neospora caninum life cycle 14 TABLES Table 1: Characteristics of apicomplexan oocysts found in the faeces of dogs and cats 10 Control of Intestinal Protozoa 6 in Dogs and Cats ESCCAP Guideline 06 Second Edition – February 2018 3 INTRODUCTION A wide range of intestinal protozoa commonly infect dogs and cats throughout Europe; with a few exceptions there seem to be no limitations in geographical distribution. -

Feline Immune Response to Infection with Cytauxzoon Felis and The
FELINE IMMUNE RESPONSE TO INFECTION WITH CYTAUXZOON FELIS AND THE ROLE OF CD18 IN THE PATHOGENESIS OF CYTAUXZOONOSIS by KARELMA FRONTERA-ACEVEDO (Under the Direction of Kaori Sakamoto) ABSTRACT Cytauxzoonosis is a highly fatal, hemoprotozoal disease of cats in the Mid-Western, Mid- Atlantic, and Southeastern United States, caused by Cytauxzoon felis. Although the causative agent has been recognized since 1976, no study has profiled the immune response of infected cats, there is no definitive cure, and C. felis has not been successfully maintained in cell cultures in vitro, thwarting research efforts. One of the main histopathologic characteristics of this disease is the presence of giant, infected, intravascular macrophages, many of which are adhered to the vascular endothelium. The main goals of this project are: 1) to characterize the feline immune response to C. felis; 2) to develop a cell culture system in order to study C. felis in vitro; and 3) to determine whether CD18 plays a role in the pathogenesis of cytauxzoonosis. INDEX WORDS: Cat, Cytauxzoon felis, pathogenesis, protozoal disease, veterinary pathology FELINE IMMUNE RESPONSE TO INFECTION WITH CYTAUXZOON FELIS AND THE ROLE OF CD18 IN THE PATHOGENESIS OF CYTAUXZOONOSIS by KARELMA FRONTERA-ACEVEDO BS, University of Florida, 2004 DVM, Louisiana State University, 2008 A Dissertation Submitted to the Graduate Faculty of The University of Georgia in Partial Fulfillment of the Requirements for the Degree DOCTOR OF PHILOSOPHY ATHENS, GEORGIA 2013 © 2013 Karelma Frontera-Acevedo All -
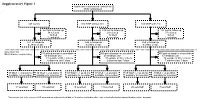
Supplementary Figure 1 Multicenter Randomised Control Trial 2746 Randomised
Supplementary Figure 1 Multicenter randomised control trial 2746 randomised 947 control 910 MNP without zinc 889 MNP with zinc 223 lost to follow up 219 lost to follow up 183 lost to follow up 34 refused 29 refused 37 refused 16 died 12 died 9 died 3 excluded 4 excluded 1 excluded 671 in follow-up 646 in follow-up 659 in follow-up at 24mo of age at 24mo of age at 24mo of age Selection for Microbiome sequencing 516 paired samples unavailable 469 paired samples unavailable 497 paired samples unavailable 69 antibiotic use 63 antibiotic use 67 antibiotic use 31 outside of WLZ criteria 37 outside of WLZ criteria 34 outside of WLZ criteria 6 diarrhea last 7 days 2 diarrhea last 7 days 7 diarrhea last 7 days 39 WLZ > -1 at 24 mo 10 WLZ < -2 at 24mo 58 WLZ > -1 at 24 mo 17 WLZ < -2 at 24mo 48 WLZ > -1 at 24 mo 8 WLZ < -2 at 24mo available for selection available for selection available for selection available for selection available for selection1 available for selection1 14 selected 10 selected 15 selected 14 selected 20 selected1 7 selected1 1 Two subjects (one in the reference WLZ group and one undernourished) had, at 12 months, no diarrhea within 1 day of stool collection but reported diarrhea within 7 days prior. Length, cm kg Weight, Supplementary Figure 2. Length (left) and weight (right) z-scores of children recruited into clinical trial NCT00705445 during the first 24 months of life. Median and quantile values are shown, with medians for participants profiled in current study indicated by red (undernourished) and black (reference WLZ) lines. -

Why the –Omic Future of Apicomplexa Should Include Gregarines Julie Boisard, Isabelle Florent
Why the –omic future of Apicomplexa should include Gregarines Julie Boisard, Isabelle Florent To cite this version: Julie Boisard, Isabelle Florent. Why the –omic future of Apicomplexa should include Gregarines. Biology of the Cell, Wiley, 2020, 10.1111/boc.202000006. hal-02553206 HAL Id: hal-02553206 https://hal.archives-ouvertes.fr/hal-02553206 Submitted on 24 Apr 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Article title: Why the –omic future of Apicomplexa should include Gregarines. Names of authors: Julie BOISARD1,2 and Isabelle FLORENT1 Authors affiliations: 1. Molécules de Communication et Adaptation des Microorganismes (MCAM, UMR 7245), Département Adaptations du Vivant (AVIV), Muséum National d’Histoire Naturelle, CNRS, CP52, 57 rue Cuvier 75231 Paris Cedex 05, France. 2. Structure et instabilité des génomes (STRING UMR 7196 CNRS / INSERM U1154), Département Adaptations du vivant (AVIV), Muséum National d'Histoire Naturelle, CP 26, 57 rue Cuvier 75231 Paris Cedex 05, France. Short Title: Gregarines –omics for Apicomplexa studies -

Tracing the Origin of Planktonic Protists in an Ancient Lake
microorganisms Article Tracing the Origin of Planktonic Protists in an Ancient Lake Nataliia V. Annenkova 1,* , Caterina R. Giner 2,3 and Ramiro Logares 2,* 1 Limnological Institute Siberian Branch of the Russian Academy of Sciences 3, Ulan-Batorskaya St., 664033 Irkutsk, Russia 2 Institute of Marine Sciences (ICM), CSIC, Passeig Marítim de la Barceloneta, 37-49, ES08003 Barcelona, Spain; [email protected] 3 Institute for the Oceans and Fisheries, University of British Columbia, 2202 Main Mall, Vancouver, BC V6T 1Z4, Canada * Correspondence: [email protected] (N.V.A.); [email protected] (R.L.) Received: 26 February 2020; Accepted: 7 April 2020; Published: 9 April 2020 Abstract: Ancient lakes are among the most interesting models for evolution studies because their biodiversity is the result of a complex combination of migration and speciation. Here, we investigate the origin of single celled planktonic eukaryotes from the oldest lake in the world—Lake Baikal (Russia). By using 18S rDNA metabarcoding, we recovered 1414 Operational Taxonomic Units (OTUs) belonging to protists populating surface waters (1–50 m) and representing pico/nano-sized cells. The recovered communities resembled other lacustrine freshwater assemblages found elsewhere, especially the taxonomically unclassified protists. However, our results suggest that a fraction of Baikal protists could belong to glacial relicts and have close relationships with marine/brackish species. Moreover, our results suggest that rapid radiation may have occurred among some protist taxa, partially mirroring what was already shown for multicellular organisms in Lake Baikal. We found 16% of the OTUs belonging to potential species flocks in Stramenopiles, Alveolata, Opisthokonta, Archaeplastida, Rhizaria, and Hacrobia. -

Assessing the Efficiency of Molecular Markers for the Species Identification of Gregarines Isolated from the Mealworm and Super Worm Midgut
Microorganisms 2018, 6, 119 1 of 20 Article Assessing the Efficiency of Molecular Markers for the Species Identification of Gregarines Isolated from the Mealworm and Super Worm Midgut Chiara Nocciolini 1, Claudio Cucini 2, Chiara Leo 2, Valeria Francardi 3, Elena Dreassi 4 and Antonio Carapelli 2,* 1 Polo d’Innovazione di Genomica Genetica e Biologia, 53100 Siena, Italy; [email protected] 2 Department of Life Sciences, University of Siena, 53100 Siena, Italy; [email protected] (C.C.); [email protected] (C.L.) 3 Consiglio per la ricerca in agricoltura e analisi dell’economia agraria – Centro di Difesa e Certificazione CREA-DC) Research Centre for Plant Protection and Certification, via di Lanciola 12/A, 50125 Firenze, Italy; [email protected] 4 Department of Biotechnology, Chemistry and Pharmacy, University of Siena, 53100 Siena, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-0577-234-410 Received: 28 September 2018; Accepted: 23 November 2018; Published: 27 November 2018 Abstract: Protozoa, of the taxon Gregarinasina, are a heterogeneous group of Apicomplexa that includes ~1600 species. They are parasites of a large variety of both marine and terrestrial invertebrates, mainly annelids, arthropods and mollusks. Unlike coccidians and heamosporidians, gregarines have not proven to have a negative effect on human welfare; thus, they have been poorly investigated. This study focuses on the molecular identification and phylogeny of the gregarine species found in the midgut of two insect species that are considered as an alternative source of animal proteins for the human diet: the mealworm Tenebrio molitor, and the super-worm Zophobas atratus (Coleoptera: Tenebrionidae). -

(Haemosporida: Haemoproteidae), with Report of in Vitro Ookinetes of Haemoproteus Hirundi
Chagas et al. Parasites Vectors (2019) 12:422 https://doi.org/10.1186/s13071-019-3679-1 Parasites & Vectors RESEARCH Open Access Sporogony of four Haemoproteus species (Haemosporida: Haemoproteidae), with report of in vitro ookinetes of Haemoproteus hirundinis: phylogenetic inference indicates patterns of haemosporidian parasite ookinete development Carolina Romeiro Fernandes Chagas* , Dovilė Bukauskaitė, Mikas Ilgūnas, Rasa Bernotienė, Tatjana Iezhova and Gediminas Valkiūnas Abstract Background: Haemoproteus (Parahaemoproteus) species (Haemoproteidae) are widespread blood parasites that can cause disease in birds, but information about their vector species, sporogonic development and transmission remain fragmentary. This study aimed to investigate the complete sporogonic development of four Haemoproteus species in Culicoides nubeculosus and to test if phylogenies based on the cytochrome b gene (cytb) refect patterns of ookinete development in haemosporidian parasites. Additionally, one cytb lineage of Haemoproteus was identifed to the spe- cies level and the in vitro gametogenesis and ookinete development of Haemoproteus hirundinis was characterised. Methods: Laboratory-reared C. nubeculosus were exposed by allowing them to take blood meals on naturally infected birds harbouring single infections of Haemoproteus belopolskyi (cytb lineage hHIICT1), Haemoproteus hirun- dinis (hDELURB2), Haemoproteus nucleocondensus (hGRW01) and Haemoproteus lanii (hRB1). Infected insects were dissected at intervals in order to detect sporogonic stages. In vitro exfagellation, gametogenesis and ookinete development of H. hirundinis were also investigated. Microscopic examination and PCR-based methods were used to confrm species identity. Bayesian phylogenetic inference was applied to study the relationships among Haemopro- teus lineages. Results: All studied parasites completed sporogony in C. nubeculosus. Ookinetes and sporozoites were found and described. Development of H. hirundinis ookinetes was similar both in vivo and in vitro. -

Plasmodium Asexual Growth and Sexual Development in the Haematopoietic Niche of the Host
REVIEWS Plasmodium asexual growth and sexual development in the haematopoietic niche of the host Kannan Venugopal 1, Franziska Hentzschel1, Gediminas Valkiūnas2 and Matthias Marti 1* Abstract | Plasmodium spp. parasites are the causative agents of malaria in humans and animals, and they are exceptionally diverse in their morphology and life cycles. They grow and develop in a wide range of host environments, both within blood- feeding mosquitoes, their definitive hosts, and in vertebrates, which are intermediate hosts. This diversity is testament to their exceptional adaptability and poses a major challenge for developing effective strategies to reduce the disease burden and transmission. Following one asexual amplification cycle in the liver, parasites reach high burdens by rounds of asexual replication within red blood cells. A few of these blood- stage parasites make a developmental switch into the sexual stage (or gametocyte), which is essential for transmission. The bone marrow, in particular the haematopoietic niche (in rodents, also the spleen), is a major site of parasite growth and sexual development. This Review focuses on our current understanding of blood-stage parasite development and vascular and tissue sequestration, which is responsible for disease symptoms and complications, and when involving the bone marrow, provides a niche for asexual replication and gametocyte development. Understanding these processes provides an opportunity for novel therapies and interventions. Gametogenesis Malaria is one of the major life- threatening infectious Malaria parasites have a complex life cycle marked Maturation of male and female diseases in humans and is particularly prevalent in trop- by successive rounds of asexual replication across gametes. ical and subtropical low- income regions of the world. -

(PAD) and Post-Translational Protein Deimination—Novel Insights Into Alveolata Metabolism, Epigenetic Regulation and Host–Pathogen Interactions
biology Article Peptidylarginine Deiminase (PAD) and Post-Translational Protein Deimination—Novel Insights into Alveolata Metabolism, Epigenetic Regulation and Host–Pathogen Interactions Árni Kristmundsson 1,*, Ásthildur Erlingsdóttir 1 and Sigrun Lange 2,* 1 Institute for Experimental Pathology at Keldur, University of Iceland, Keldnavegur 3, 112 Reykjavik, Iceland; [email protected] 2 Tissue Architecture and Regeneration Research Group, School of Life Sciences, University of Westminster, London W1W 6UW, UK * Correspondence: [email protected] (Á.K.); [email protected] (S.L.) Simple Summary: Alveolates are a major group of free living and parasitic organisms; some of which are serious pathogens of animals and humans. Apicomplexans and chromerids are two phyla belonging to the alveolates. Apicomplexans are obligate intracellular parasites; that cannot complete their life cycle without exploiting a suitable host. Chromerids are mostly photoautotrophs as they can obtain energy from sunlight; and are considered ancestors of the apicomplexans. The pathogenicity and life cycle strategies differ significantly between parasitic alveolates; with some causing major losses in host populations while others seem harmless to the host. As the life cycles of Citation: Kristmundsson, Á.; Erlingsdóttir, Á.; Lange, S. some are still poorly understood, a better understanding of the factors which can affect the parasitic Peptidylarginine Deiminase (PAD) alveolates’ life cycles and survival is of great importance and may aid in new biomarker discovery. and Post-Translational Protein This study assessed new mechanisms relating to changes in protein structure and function (so-called Deimination—Novel Insights into “deimination” or “citrullination”) in two key parasites—an apicomplexan and a chromerid—to Alveolata Metabolism, Epigenetic assess the pathways affected by this protein modification.