The Impacts of a Stem Boring Weevil, Mecinus Janthinus , On
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Role of a State Insectary in Weed Biocontrol
The Role of a State Insectary in Weed Biocontrol Dan Bean, Director Palisade Insectary Colorado Department of Agriculture Conservation Services Commissioner’s Office 7 Divisions Plant Industry Brands Inspection and Consumer Services Conservation Services Markets State Fair Animal Industries Commissioner’s Office 7 Divisions Conservation Services Commissioner’s Office 7 Divisions Conservation Services 4 Programs Commissioner’s Office 7 Divisions Conservation Services 4 Programs Conservation Districts Commissioner’s Office 7 Divisions Conservation Services 4 Programs Groundwater Conservation Districts Commissioner’s Office 7 Divisions Conservation Services 4 Programs Noxious Weeds Groundwater Conservation Districts Commissioner’s Office 7 Divisions Conservation Services 4 Programs Biological Pest Control Noxious Weeds Groundwater Conservation Districts Commissioner’s Office 7 Divisions Conservation Services 4 Programs Biological Pest Control Palisade Insectary Palisade, Colorado Palisade Insectary . Began in the 1940’s to fight Oriental fruit moth, a project that helped peach farmers and is still going . Moved to new 14,000 sq ft facility in 1992 . Distributes over 30 biocontrol agents for the control of insect pests and weeds . Is a partner in pest management on a local, state, tribal and federal level The Program mission is to provide biological control agents and expertise to the citizens of Colorado in order to assist in achieving their land and resource management objectives Leafy spurge infestation near Pine, CO Eric Lane, Director -

3.7.10 Curculioninae Latreille, 1802 Jetzt Beschriebenen Palaearctischen Ceuthor- Rhynchinen
Curculioninae Latreille, 1802 305 Schultze, A. (1902): Kritisches Verzeichniss der bis 3.7.10 Curculioninae Latreille, 1802 jetzt beschriebenen palaearctischen Ceuthor- rhynchinen. – Deutsche Entomologische Zeitschrift Roberto Caldara , Nico M. Franz, and Rolf 1902: 193 – 226. G. Oberprieler Schwarz, E. A. (1894): A “ parasitic ” scolytid. – Pro- ceedings of the Entomological Society of Washington 3: Distribution. The subfamily as here composed (see 15 – 17. Phylogeny and Taxonomy below) includes approx- Scudder, S. H. (1893): Tertiary Rhynchophorous Coleo- ptera of the United States. xii + 206 pp. US Geological imately 350 genera and 4500 species (O ’ Brien & Survey, Washington, DC. Wibmer 1978; Thompson 1992; Alonso-Zarazaga Stierlin, G. (1886): Fauna insectorum Helvetiae. Coleo- & Lyal 1999; Oberprieler et al. 2007), provisionally ptera helvetiae , Volume 2. 662 pp. Rothermel & Cie., divided into 34 tribes. These are geographically Schaffhausen. generally restricted to a lesser or larger degree, only Thompson, R. T. (1973): Preliminary studies on the two – Curculionini and Rhamphini – being virtually taxonomy and distribution of the melon weevil, cosmopolitan in distribution and Anthonomini , Acythopeus curvirostris (Boheman) (including Baris and Tychiini only absent from the Australo-Pacifi c granulipennis (Tournier)) (Coleoptera, Curculion- region. Acalyptini , Cionini , Ellescini , Mecinini , idae). – Bulletin of Entomological Research 63: 31 – 48. and Smicronychini occur mainly in the Old World, – (1992): Observations on the morphology and clas- from Africa to the Palaearctic and Oriental regions, sifi cation of weevils (Coleoptera, Curculionidae) with Ellescini, Acalyptini, and Smicronychini also with a key to major groups. – Journal of Natural His- extending into the Nearctic region and at least tory 26: 835 – 891. the latter two also into the Australian one. -

Curculionidae (Except Scolytinae and Platypodinae) in Latvian Fauna, Taxonomical Structure, Biogeography and Forecasted Species
Acta Biol. Univ. Daugavp. 12 (4) 2012 ISSN 1407 - 8953 CURCULIONIDAE (EXCEPT SCOLYTINAE AND PLATYPODINAE) IN LATVIAN FAUNA, TAXONOMICAL STRUCTURE, BIOGEOGRAPHY AND FORECASTED SPECIES Maksims Balalaikins Balalaikins M. 2012. Curculionidae (except Scolytinae and Platypodinae) in Latvian fauna, taxonomical structure, biogeography and forecasted species. Acta Biol. Univ. Daugavp., 12 (4): 67 – 83. This paper presents analysis of taxonomical structure of Latvian Curculionidae (except Scolytinae and Platypodinae) and comparison with the neighboring countries (Lithuania and Estonia) weevil’s fauna. Range of chorotypes and biogeography analysis of Latvian Curculionidae (except Scolytinae and Platypodinae) is presented in current paper. List of forecasted weevils species of Latvian fauna is compilled. Key words: Coleoptera, Curculionidae, Latvia, fauna, taxonomical structure, chorotypes, forecasted species. Maksims Balalaikins Institute of Systematic Biology, Daugavpils University, Vienības 13, Daugavpils, LV-5401, Latvia; e-mail: [email protected] INTRODUCTION The current paper is a continuation of studies on the Latvian fauna of Curculionidae (Balalaikins Worldwide, the Curculionidae is one of the largest 2011a, 2011b, 2012a, 2012b, 2012c, 2012d, in families of the order Coleoptera, represented by press; Balalaikins & Bukejs 2009, 2010, 2011a, 4600 genera and 51000 species (Alonso-Zarazaga 2011b, 2012), Balalaikins & Telnov 2012. The and Lyal 1999, Oberprieler et al. 2007). This aim of this work is to summarize and analyse family -

Biology and Biological Control of Dalmatian and Y Ellow T Oadflax
Forest Health Technology Enterprise Team TECHNOLOGY TRANSFER Biological Control BIOLOGY AND BIOLOGICAL CONTROL OF DALMATIAN AND Y ELLOW T OADFLAX LINDA M. WILSON, SHARLENE E. SING, GARY L. PIPER, RICHARD W. H ANSEN, ROSEMARIE DE CLERCK-FLOATE, DANIEL K. MACKINNON, AND CAROL BELL RANDALL Forest Health Technology Enterprise Team—Morgantown FHTET-2005-13 U.S. Department Forest September 2005 of Agriculture Service he Forest Health Technology Enterprise Team (FHTET) was created in 1995 Tby the Deputy Chief for State and Private Forestry, USDA, Forest Service, to develop and deliver technologies to protect and improve the health of American forests. This book was published by FHTET as part of the technology transfer series. http://www.fs.fed.us/foresthealth/technology/ Cover photos: Toadflax (UGA1416053)—Linda Wilson, Beetles (UGA14160033-top, UGA1416054-bottom)—Bob Richard All photographs in this publication can be accessed and viewed on-line at www.forestryimages.org, sponsored by the University of Georgia. You will find reference codes (UGA000000) in the captions for each figure in this publication. To access them, point your browser at http://www.forestryimages.org, and enter the reference code at the search prompt. How to cite this publication: Wilson, L. M., S. E. Sing, G. L. Piper, R. W. Hansen, R. De Clerck- Floate, D. K. MacKinnon, and C. Randall. 2005. Biology and Biological Control of Dalmatian and Yellow Toadflax. USDA Forest Service, FHTET-05-13. The U.S. Department of Agriculture (USDA) prohibits discrimination in all its programs and activities on the basis of race, color, national origin, sex, religion, age, disability, political beliefs, sexual orientation, or marital or family status. -

Bugs and Diseases Newsletter December 2015
Vol. 26 No. 3 December 2015 It’s the Thought That Counts... It’s the time of year to look forward to spending time with family and showing each other how much we care by giving the perfect gift. This isn’t limited to just humans though, insects exhibit this kind of behaviour too! Family time in ambrosia beetles: The majority of ambrosia beetle species tunnel Alberta’s eye into the heartwood of weakened trees, where they cultivate fungus as a food source. Like termites, bees, and ants, many ambrosia beetles are eusocial; they on forest live in colonies with many individuals and overlapping generations. Just like health human families, every individual in the colony has their job. The brood are respon- sible for enlarging the gallery and sweeping up the poop (frass). Adults protect the brood and shuffle the frass balls out of the gallery, while everyone helps take care Issue highlights: of the fungus garden. And as with many human families, the mother runs things: The matriarch of the ambrosia beetle colony is the sole egg-layer, while the other Insect Gifting 1 females care for the offspring. Once the colony is thriving, subordinate females strike out on their own to start their own colonies and become matriarchs in their What’s the BUZZ— 2 Moth of the Mtns own right. Gypsy Moth Detec- 3 Gift-giving in dance flies: Insects are some of the best gift-givers because they tion in NE AB give such a wide variety of gifts in many different ways. But here’s where it gets a bit racythese are usually nuptial gifts; gifts that male insects give to females Spruce Beetle Update 5 in RMH Area during courtship and mating, often to convince the female that he’s Mr. -

Chapter 12. Biocontrol Arthropods: New Denizens of Canada's
291 Chapter 12 Biocontrol Arthropods: New Denizens of Canada’s Grassland Agroecosystems Rosemarie De Clerck-Floate and Héctor Cárcamo Agriculture and Agri-Food Canada, Lethbridge Research Centre 5403 - 1 Avenue South, P.O. Box 3000 Lethbridge, Alberta, Canada T1J 4B1 Abstract. Canada’s grassland ecosystems have undergone major changes since the arrival of European agriculture, ranging from near-complete replacement of native biodiversity with annual crops to the effects of overgrazing by cattle on remnant native grasslands. The majority of the “agroecosystems” that have replaced the historical native grasslands now encompass completely new associations of plants and arthropods in what is typically a mix of introduced and native species. Some of these species are pests of crops and pastures and were accidentally introduced. Other species are natural enemies of these pests and were deliberately introduced as classical biological control (biocontrol) agents to control these pests. To control weeds, 76 arthropod species have been released against 24 target species in Canada since 1951, all of which also have been released in western Canada. Of these released species, 53 (70%) have become established, with 18 estimated to be reducing target weed populations. The biocontrol programs for leafy spurge in the prairie provinces and knapweeds in British Columbia have been the largest, each responsible for the establishment of 10 new arthropod species on rangelands. This chapter summarizes the ecological highlights of these programs and those for miscellaneous weeds. Compared with weed biocontrol on rangelands, classical biocontrol of arthropod crop pests by using arthropods lags far behind, mostly because of a preference to manage crop pests with chemicals. -

Operational Field Guide
BRITISH COLUMBIA MINISTRY OF FORESTS AND RANGE OPERATIONAL FIELD GUIDE TO THE PROPAGATION AND ESTABLISHMENT OF THE BIOCONTROL AGENT RHINUSA ANTIRRHINI (TOADFLAX SEED-FEEDER) May 2008 The contents of this Field Guide may not be cited in whole or in part without the advance written approval of the Director, Forest Practices Branch, Ministry of Forests and Range, Victoria, British Columbia. Information contained in this Field Guide is comprised of fact and field observations as of September 2007. Site specific experiences may vary. OPERATIONAL FIELD GUIDE TO THE PROPAGATION AND ESTABLISHMENT OF THE BIOCONTROL AGENT RHINUSA ANTIRRHINI (TOADFLAX SEED-FEEDER) May 2008 Forest Practices Branch Integrated Resources Section Biocontrol Development Program British Columbia Ministry of Forests and Range PREPARED BY: SUSAN TURNER BIOCONTROL DEVELOPMENT PROGRAM FOREST PRACTICES BRANCH MINISTRY OF FORESTS AND RANGE EDITED BY: DR. ROSE DECLERCK‐FLOATE RESEARCH SCIENTIST WEED BIOCONTROL AGRICULTURE AND AGRI‐FOOD CANADA INFORMATION CONTRIBUTED BY: SANDY CESSELLI MARSHA DEWOLF BIOCONTROL DEVELOPMENT PROGRAM FOREST PRACTICES BRANCH MINISTRY OF FORESTS AND RANGE Rhinusa antirrhini (Toadflax seed-feeding weevil) - Operational Field Guide TABLE OF CONTENTS 1. PURPOSE....................................................................................................................................................... 1 2. INTRODUCTION............................................................................................................................................ -

The Impacts of a Stem Boring Weevil, Mecinus Janthinus, on Dalmatian
THE IMPACTS OF A STEM BORING WEEVIL, MECINUS JANTHINUS , ON DALMATIAN TOADFLAX, LINARIA DALMATICA by Marjolein Schat A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Ecology and Environmental Sciences MONTANA STATE UNIVERSITY Bozeman, Montana December 2008 ©COPYRIGHT by Marjolein Schat 2008 All Rights Reserved ii APPROVAL of a dissertation submitted by Marjolein Schat This dissertation has been read by each member of the dissertation committee and has been found to be satisfactory regarding content, English usage, format, citation, bibliographic style, and consistency, and is ready for submission to the Division of Graduate Education. Robert K.D. Peterson Fabian Menalled Approved for the Department of Land Resources and Environmental Sciences Bruce D. Maxwell Approved for the Division of Graduate Education Dr. Carl A. Fox iii STATEMENT OF PERMISSION TO USE In presenting this dissertation in partial fulfillment of the requirements for a doctoral degree at Montana State University, I agree that the Library shall make it available to borrowers under rules of the Library. I further agree that copying of this dissertation is allowable only for scholarly purposes, consistent with “fair use” as prescribed in the U.S. Copyright Law. Requests for extensive copying or reproduction of this dissertation should be referred to ProQuest Information and Learning, 300 North Zeeb Road, Ann Arbor, Michigan 48106, to whom I have granted “the exclusive right to reproduce and distribute my dissertation in and from microform along with the non- exclusive right to reproduce and distribute my abstract in any format in whole or in part.” Marjolein Schat December 2008 iv ACKNOWLEDGEMENTS I would never have embarked on this journey without the example and encouragement provided by my father, K.A. -

Invasive Vegetation Management Plan Environmental Assessment February 21, 2013
National Park Service U.S. Department of the Interior Yellowstone National Park Invasive Vegetation Management Plan Environmental Assessment February 21, 2013 ii Invasive Vegetation Management Plan / Environmental Assessment Executive Summary Yellowstone National Park (YNP) has developed a plan to address terrestrial invasive nonnative plants parkwide. (As used here, the term “invasive nonnative plant” has a similar meaning as alien, exotic, or noxious plant or weed.) This Invasive Vegetation Management Plan and Environmental Assessment (EA) provides a comprehensive approach toward invasive vegetation management to preserve, protect, and restore the diversity, ecological integrity, and processes associated with native plant communities in the park. The purpose of the plan is to expand current invasive plant management efforts and implement a parkwide strategy to prevent the establishment and spread of invasive nonnative plant species and to restore, as needed, native plant communities in the park. The plan proposes to achieve these goals by accomplishing the following objectives: 1. Prevent the entry and establishment of new invasive plants 2. Control existing populations of invasive plants by eradicating them, reducing their abundance and density, and containing their spread 3. Restore native plant communities when they have been disrupted or replaced by invasive nonnative plant populations. This environmental assessment evaluates a No-Action Alternative and one action alternative: • Alternative A – The No-Action Alternative represents -

Phylogeny and Sequestration of Iridoid Glycosides In
Phylogeny and sequestration of iridoid glycosides in selected genera of the Mecininae (Coleoptera, Curculionidae) with particular focus on their host plant relationship Dissertation Zur Erlangung der Würde des Doktors der Naturwissenschaften des Fachbereichs Biologie, der Fakultät für Mathematik, Informatik und Naturwissenschaften, der Universität Hamburg vorgelegt von Christian Ulrich Baden aus Hamburg Hamburg, im August 2011 April 18, 2011 To Whom It May Concern: I have read over the Ph.D. thesis of Christian Baden entitled, 'Phylogeny and sequestration of iridoidglycosides in selected genera of the Mecininae (Coleoptera, Curculionidae) with particular focus on their host plant relationship'. As a native speaker of English, I attest to the quality of Mr. Baden's English. Sincerely, Scott T. Kelley Associate Professor Department of Biology San Diego State University 5500 Campanile Drive San Diego, CA 92182-4614 5500 Campanile Drive, San Diego, CA 92182-4614 Tel (619) 594-5371 FAX (619) 594-5676 Aufführung der in Anspruch genommenen fremden Hilfen In den Kapiteln 2 und 3 hat Herr Dr. Stephan Franke die GC, GC/MS und HPLC/MS mit mir zusammen und teilweise alleine bedient. Des Weiteren hat er bei der Zuordnung der chemischen Stoffe geholfen. In Kapitel 4 hat Dr. Ralph Peters mir bei der Realisierung des Olfaktometers und bei der statistischen Auswertung geholfen. In Kapitel 5 hat mich Viola Boxberger an einigen Tagen bei den Arbeiten im Feld unterstützt. Wenn Käfer nicht von mir gesammelt wurden, so ist dies innerhalb der Arbeit vermerkt. -

Field Guide for the Biological Control of Weeds in Eastern North America
US Department TECHNOLOGY of Agriculture TRANSFER FIELD GUIDE FOR THE BIOLOGICAL CONTROL OF WEEDS IN EASTERN NORTH AMERICA Rachel L. Winston, Carol B. Randall, Bernd Blossey, Philip W. Tipping, Ellen C. Lake, and Judy Hough-Goldstein Forest Health Technology FHTET-2016-04 Enterprise Team April 2017 The Forest Health Technology Enterprise Team (FHTET) was created in 1995 by the Deputy Chief for State and Private Forestry, USDA, Forest Service, to develop and deliver technologies to protect and improve the health of American forests. This book was published by FHTET as part of the technology transfer series. http://www.fs.fed.us/foresthealth/technology/ Cover photos: Purple loosestrife (Jennifer Andreas, Washington State University Extension), Galerucella calmariensis (David Cappaert, Michigan State University, bugwood.org), tropical soda apple ((J. Jeffrey Mullahey, University of Florida, bugwood.org), Gratiana boliviana (Rodrigo Diaz, Louisiana State University), waterhyacinth (Chris Evans, University of Illinois, bugwood.org), Megamelus scutellaris (Jason D. Stanley, USDA ARS, bugwood.org), mile-a-minute weed (Leslie J. Mehrhoff, University of Connecticut, bugwood.org), Rhinoncomimus latipes (Amy Diercks, bugwood.org) How to cite this publication: Winston, R.L., C.B. Randall, B. Blossey, P.W. Tipping, E.C. Lake, and J. Hough-Goldstein. 2017. Field Guide for the Biological Control of Weeds in Eastern North America. USDA Forest Service, Forest Health Technology Enterprise Team, Morgantown, West Virginia. FHTET-2016-04. In accordance with -
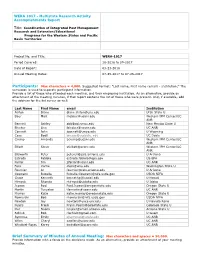
WERA1017 2017 Report.Pdf
WERA 1017 - Multistate Research Activity Accomplishments Report Title: Coordination of Integrated Pest Management Research and Extension/Educational Programs for the Western States and Pacific Basin Territories Project No. and Title: WERA-1017 Period Covered: 10-2016 to 09-2017 Date of Report: 03-23-2018 Annual Meeting Dates: 07-25-2017 to 07-26-2017 Participants: Max characters = 4,000. Suggested Format: "Last name, First name (email) - Institution;" The semicolon is used to separate participant information. Provide a list of those who attended each meeting, and their employing institution. As an alternative, provide an attachment of the meeting minutes, if that report contains the list of those who were present. And, if available, add the address for the list server as well. Last Name First Name email Institution Alston Diane [email protected] Utah State U Baur Matt [email protected] Western IPM Center/UC ANR Bennett Ashley [email protected] New Mexico State U Blecker Lisa [email protected] UC ANR Connett John [email protected] U Wyoming Cass Bodil bncass@ucdavis. edu UC Davis Crump Amanda [email protected] Western IPM Center/UC ANR Elliott Steve [email protected] Western IPM Center/UC ANR Ellsworth Peter [email protected] U Arizona Estrada Fabiola [email protected] US EPA Farrar Jim [email protected] UC ANR Foss Carrie [email protected] Washington State U Fournier Al [email protected] U Arizona Goswami Rubella [email protected] USDA NIFA Grace Kenneth [email protected] U Hawaii Hirnyck Rhonda [email protected]