What Is the Difference Between Classical Physics and Quantum Physics?
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Glossary Physics (I-Introduction)
1 Glossary Physics (I-introduction) - Efficiency: The percent of the work put into a machine that is converted into useful work output; = work done / energy used [-]. = eta In machines: The work output of any machine cannot exceed the work input (<=100%); in an ideal machine, where no energy is transformed into heat: work(input) = work(output), =100%. Energy: The property of a system that enables it to do work. Conservation o. E.: Energy cannot be created or destroyed; it may be transformed from one form into another, but the total amount of energy never changes. Equilibrium: The state of an object when not acted upon by a net force or net torque; an object in equilibrium may be at rest or moving at uniform velocity - not accelerating. Mechanical E.: The state of an object or system of objects for which any impressed forces cancels to zero and no acceleration occurs. Dynamic E.: Object is moving without experiencing acceleration. Static E.: Object is at rest.F Force: The influence that can cause an object to be accelerated or retarded; is always in the direction of the net force, hence a vector quantity; the four elementary forces are: Electromagnetic F.: Is an attraction or repulsion G, gravit. const.6.672E-11[Nm2/kg2] between electric charges: d, distance [m] 2 2 2 2 F = 1/(40) (q1q2/d ) [(CC/m )(Nm /C )] = [N] m,M, mass [kg] Gravitational F.: Is a mutual attraction between all masses: q, charge [As] [C] 2 2 2 2 F = GmM/d [Nm /kg kg 1/m ] = [N] 0, dielectric constant Strong F.: (nuclear force) Acts within the nuclei of atoms: 8.854E-12 [C2/Nm2] [F/m] 2 2 2 2 2 F = 1/(40) (e /d ) [(CC/m )(Nm /C )] = [N] , 3.14 [-] Weak F.: Manifests itself in special reactions among elementary e, 1.60210 E-19 [As] [C] particles, such as the reaction that occur in radioactive decay. -

Path Integrals in Quantum Mechanics
Path Integrals in Quantum Mechanics Dennis V. Perepelitsa MIT Department of Physics 70 Amherst Ave. Cambridge, MA 02142 Abstract We present the path integral formulation of quantum mechanics and demon- strate its equivalence to the Schr¨odinger picture. We apply the method to the free particle and quantum harmonic oscillator, investigate the Euclidean path integral, and discuss other applications. 1 Introduction A fundamental question in quantum mechanics is how does the state of a particle evolve with time? That is, the determination the time-evolution ψ(t) of some initial | i state ψ(t ) . Quantum mechanics is fully predictive [3] in the sense that initial | 0 i conditions and knowledge of the potential occupied by the particle is enough to fully specify the state of the particle for all future times.1 In the early twentieth century, Erwin Schr¨odinger derived an equation specifies how the instantaneous change in the wavefunction d ψ(t) depends on the system dt | i inhabited by the state in the form of the Hamiltonian. In this formulation, the eigenstates of the Hamiltonian play an important role, since their time-evolution is easy to calculate (i.e. they are stationary). A well-established method of solution, after the entire eigenspectrum of Hˆ is known, is to decompose the initial state into this eigenbasis, apply time evolution to each and then reassemble the eigenstates. That is, 1In the analysis below, we consider only the position of a particle, and not any other quantum property such as spin. 2 D.V. Perepelitsa n=∞ ψ(t) = exp [ iE t/~] n ψ(t ) n (1) | i − n h | 0 i| i n=0 X This (Hamiltonian) formulation works in many cases. -

Key Concepts for Future QIS Learners Workshop Output Published Online May 13, 2020
Key Concepts for Future QIS Learners Workshop output published online May 13, 2020 Background and Overview On behalf of the Interagency Working Group on Workforce, Industry and Infrastructure, under the NSTC Subcommittee on Quantum Information Science (QIS), the National Science Foundation invited 25 researchers and educators to come together to deliberate on defining a core set of key concepts for future QIS learners that could provide a starting point for further curricular and educator development activities. The deliberative group included university and industry researchers, secondary school and college educators, and representatives from educational and professional organizations. The workshop participants focused on identifying concepts that could, with additional supporting resources, help prepare secondary school students to engage with QIS and provide possible pathways for broader public engagement. This workshop report identifies a set of nine Key Concepts. Each Concept is introduced with a concise overall statement, followed by a few important fundamentals. Connections to current and future technologies are included, providing relevance and context. The first Key Concept defines the field as a whole. Concepts 2-6 introduce ideas that are necessary for building an understanding of quantum information science and its applications. Concepts 7-9 provide short explanations of critical areas of study within QIS: quantum computing, quantum communication and quantum sensing. The Key Concepts are not intended to be an introductory guide to quantum information science, but rather provide a framework for future expansion and adaptation for students at different levels in computer science, mathematics, physics, and chemistry courses. As such, it is expected that educators and other community stakeholders may not yet have a working knowledge of content covered in the Key Concepts. -

Department of Physics College of Arts and Sciences Physics
DEPARTMENT OF PHYSICS COLLEGE OF ARTS AND SCIENCES PHYSICS Faculty I. Major in Physics—38 hours William Nettles (2006). Professor of Physics, Department A. Physics 231-232, 311, 313, 314, 420, 424(1-3 Chair, and Associate Dean of the College of Arts and hours), 430, 498—28–30 hours Sciences. B.S., Mississippi College; M.S., and Ph.D., B. Select three or more courses: PHY 262, 325, 350, Vanderbilt University. 360, 395-6-7*, 400, 410, 417, 425 (1-2 hours**), 495* Ildefonso Guilaran (2008). Associate Professor of Physics. C. Prerequisites: MAT 211, 212, 213, 314 B.S., Western Kentucky University; M.S. and Ph.D., *Must be approved Special/Independent Studies Florida State University. **Maximum 3 hours from 424 and 425 apply to major. Geoffrey Poore (2010). Assistant Professor of Physics. B.A., II. Major in Physical Science—44 hours Wheaton College; M.S. and Ph.D., University of Illinois. A. CHE 111, 112, 113, 211, 221—15 hours David A. Ward (1992, 1999). Professor of Physics, B.S. B. PHY 112, 231-32, 311, 310 or 301—22 hours and M.A., University of South Florida; Ph.D., North C. Upper Level Electives from CHE and PHY—7 Carolina State University. hours; maximum 1 hour from 424 and 1 from 498 III. Minor in Physics—24 semester hours Staff Physics 231-232, 311, + 10 hours of Physics electives Christine Rowland (2006). Academic Secretary— except PHY 111, 112, 301, 310 Engineering, Physics, Math, and Computer Science. IV. Teacher Licensure in Physics (Grades 6–12) A. Complete the requirements shown above for the Physics or Physical Science major. -

Fundamentals of Particle Physics
Fundamentals of Par0cle Physics Particle Physics Masterclass Emmanuel Olaiya 1 The Universe u The universe is 15 billion years old u Around 150 billion galaxies (150,000,000,000) u Each galaxy has around 300 billion stars (300,000,000,000) u 150 billion x 300 billion stars (that is a lot of stars!) u That is a huge amount of material u That is an unimaginable amount of particles u How do we even begin to understand all of matter? 2 How many elementary particles does it take to describe the matter around us? 3 We can describe the material around us using just 3 particles . 3 Matter Particles +2/3 U Point like elementary particles that protons and neutrons are made from. Quarks Hence we can construct all nuclei using these two particles -1/3 d -1 Electrons orbit the nuclei and are help to e form molecules. These are also point like elementary particles Leptons We can build the world around us with these 3 particles. But how do they interact. To understand their interactions we have to introduce forces! Force carriers g1 g2 g3 g4 g5 g6 g7 g8 The gluon, of which there are 8 is the force carrier for nuclear forces Consider 2 forces: nuclear forces, and electromagnetism The photon, ie light is the force carrier when experiencing forces such and electricity and magnetism γ SOME FAMILAR THE ATOM PARTICLES ≈10-10m electron (-) 0.511 MeV A Fundamental (“pointlike”) Particle THE NUCLEUS proton (+) 938.3 MeV neutron (0) 939.6 MeV E=mc2. Einstein’s equation tells us mass and energy are equivalent Wave/Particle Duality (Quantum Mechanics) Einstein E -

A Brief History and Philosophy of Mechanics
A Brief History and Philosophy of Mechanics S. A. Gadsden McMaster University, [email protected] 1. Introduction During this period, several clever inventions were created. Some of these were Archimedes screw, Hero of Mechanics is a branch of physics that deals with the Alexandria’s aeolipile (steam engine), the catapult and interaction of forces on a physical body and its trebuchet, the crossbow, pulley systems, odometer, environment. Many scholars consider the field to be a clockwork, and a rolling-element bearing (used in precursor to modern physics. The laws of mechanics Roman ships). [5] Although mechanical inventions apply to many different microscopic and macroscopic were being made at an accelerated rate, it wasn’t until objects—ranging from the motion of electrons to the after the Middle Ages when classical mechanics orbital patterns of galaxies. [1] developed. Attempting to understand the underlying principles of 3. Classical Period (1500 – 1900 AD) the universe is certainly a daunting task. It is therefore no surprise that the development of ‘idea and thought’ Classical mechanics had many contributors, although took many centuries, and bordered many different the most notable ones were Galileo, Huygens, Kepler, fields—atomism, metaphysics, religion, mathematics, Descartes and Newton. “They showed that objects and mechanics. The history of mechanics can be move according to certain rules, and these rules were divided into three main periods: antiquity, classical, and stated in the forms of laws of motion.” [1] quantum. The most famous of these laws were Newton’s Laws of 2. Period of Antiquity (800 BC – 500 AD) Motion, which accurately describe the relationships between force, velocity, and acceleration on a body. -

Classical Mechanics
Classical Mechanics Hyoungsoon Choi Spring, 2014 Contents 1 Introduction4 1.1 Kinematics and Kinetics . .5 1.2 Kinematics: Watching Wallace and Gromit ............6 1.3 Inertia and Inertial Frame . .8 2 Newton's Laws of Motion 10 2.1 The First Law: The Law of Inertia . 10 2.2 The Second Law: The Equation of Motion . 11 2.3 The Third Law: The Law of Action and Reaction . 12 3 Laws of Conservation 14 3.1 Conservation of Momentum . 14 3.2 Conservation of Angular Momentum . 15 3.3 Conservation of Energy . 17 3.3.1 Kinetic energy . 17 3.3.2 Potential energy . 18 3.3.3 Mechanical energy conservation . 19 4 Solving Equation of Motions 20 4.1 Force-Free Motion . 21 4.2 Constant Force Motion . 22 4.2.1 Constant force motion in one dimension . 22 4.2.2 Constant force motion in two dimensions . 23 4.3 Varying Force Motion . 25 4.3.1 Drag force . 25 4.3.2 Harmonic oscillator . 29 5 Lagrangian Mechanics 30 5.1 Configuration Space . 30 5.2 Lagrangian Equations of Motion . 32 5.3 Generalized Coordinates . 34 5.4 Lagrangian Mechanics . 36 5.5 D'Alembert's Principle . 37 5.6 Conjugate Variables . 39 1 CONTENTS 2 6 Hamiltonian Mechanics 40 6.1 Legendre Transformation: From Lagrangian to Hamiltonian . 40 6.2 Hamilton's Equations . 41 6.3 Configuration Space and Phase Space . 43 6.4 Hamiltonian and Energy . 45 7 Central Force Motion 47 7.1 Conservation Laws in Central Force Field . 47 7.2 The Path Equation . -

Quantum Field Theory*
Quantum Field Theory y Frank Wilczek Institute for Advanced Study, School of Natural Science, Olden Lane, Princeton, NJ 08540 I discuss the general principles underlying quantum eld theory, and attempt to identify its most profound consequences. The deep est of these consequences result from the in nite number of degrees of freedom invoked to implement lo cality.Imention a few of its most striking successes, b oth achieved and prosp ective. Possible limitation s of quantum eld theory are viewed in the light of its history. I. SURVEY Quantum eld theory is the framework in which the regnant theories of the electroweak and strong interactions, which together form the Standard Mo del, are formulated. Quantum electro dynamics (QED), b esides providing a com- plete foundation for atomic physics and chemistry, has supp orted calculations of physical quantities with unparalleled precision. The exp erimentally measured value of the magnetic dip ole moment of the muon, 11 (g 2) = 233 184 600 (1680) 10 ; (1) exp: for example, should b e compared with the theoretical prediction 11 (g 2) = 233 183 478 (308) 10 : (2) theor: In quantum chromo dynamics (QCD) we cannot, for the forseeable future, aspire to to comparable accuracy.Yet QCD provides di erent, and at least equally impressive, evidence for the validity of the basic principles of quantum eld theory. Indeed, b ecause in QCD the interactions are stronger, QCD manifests a wider variety of phenomena characteristic of quantum eld theory. These include esp ecially running of the e ective coupling with distance or energy scale and the phenomenon of con nement. -

Engineering Physics I Syllabus COURSE IDENTIFICATION Course
Engineering Physics I Syllabus COURSE IDENTIFICATION Course Prefix/Number PHYS 104 Course Title Engineering Physics I Division Applied Science Division Program Physics Credit Hours 4 credit hours Revision Date Fall 2010 Assessment Goal per Outcome(s) 70% INSTRUCTION CLASSIFICATION Academic COURSE DESCRIPTION This course is the first semester of a calculus-based physics course primarily intended for engineering and science majors. Course work includes studying forces and motion, and the properties of matter and heat. Topics will include motion in one, two, and three dimensions, mechanical equilibrium, momentum, energy, rotational motion and dynamics, periodic motion, and conservation laws. The laboratory (taken concurrently) presents exercises that are designed to reinforce the concepts presented and discussed during the lectures. PREREQUISITES AND/OR CO-RECQUISITES MATH 150 Analytic Geometry and Calculus I The engineering student should also be proficient in algebra and trigonometry. Concurrent with Phys. 140 Engineering Physics I Laboratory COURSE TEXT *The official list of textbooks and materials for this course are found on Inside NC. • COLLEGE PHYSICS, 2nd Ed. By Giambattista, Richardson, and Richardson, McGraw-Hill, 2007. • Additionally, the student must have a scientific calculator with trigonometric functions. COURSE OUTCOMES • To understand and be able to apply the principles of classical Newtonian mechanics. • To effectively communicate classical mechanics concepts and solutions to problems, both in written English and through mathematics. • To be able to apply critical thinking and problem solving skills in the application of classical mechanics. To demonstrate successfully accomplishing the course outcomes, the student should be able to: 1) Demonstrate knowledge of physical concepts by their application in problem solving. -
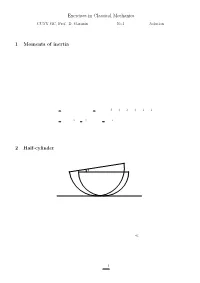
Exercises in Classical Mechanics 1 Moments of Inertia 2 Half-Cylinder
Exercises in Classical Mechanics CUNY GC, Prof. D. Garanin No.1 Solution ||||||||||||||||||||||||||||||||||||||||| 1 Moments of inertia (5 points) Calculate tensors of inertia with respect to the principal axes of the following bodies: a) Hollow sphere of mass M and radius R: b) Cone of the height h and radius of the base R; both with respect to the apex and to the center of mass. c) Body of a box shape with sides a; b; and c: Solution: a) By symmetry for the sphere I®¯ = I±®¯: (1) One can ¯nd I easily noticing that for the hollow sphere is 1 1 X ³ ´ I = (I + I + I ) = m y2 + z2 + z2 + x2 + x2 + y2 3 xx yy zz 3 i i i i i i i i 2 X 2 X 2 = m r2 = R2 m = MR2: (2) 3 i i 3 i 3 i i b) and c): Standard solutions 2 Half-cylinder ϕ (10 points) Consider a half-cylinder of mass M and radius R on a horizontal plane. a) Find the position of its center of mass (CM) and the moment of inertia with respect to CM. b) Write down the Lagrange function in terms of the angle ' (see Fig.) c) Find the frequency of cylinder's oscillations in the linear regime, ' ¿ 1. Solution: (a) First we ¯nd the distance a between the CM and the geometrical center of the cylinder. With σ being the density for the cross-sectional surface, so that ¼R2 M = σ ; (3) 2 1 one obtains Z Z 2 2σ R p σ R q a = dx x R2 ¡ x2 = dy R2 ¡ y M 0 M 0 ¯ 2 ³ ´3=2¯R σ 2 2 ¯ σ 2 3 4 = ¡ R ¡ y ¯ = R = R: (4) M 3 0 M 3 3¼ The moment of inertia of the half-cylinder with respect to the geometrical center is the same as that of the cylinder, 1 I0 = MR2: (5) 2 The moment of inertia with respect to the CM I can be found from the relation I0 = I + Ma2 (6) that yields " µ ¶ # " # 1 4 2 1 32 I = I0 ¡ Ma2 = MR2 ¡ = MR2 1 ¡ ' 0:3199MR2: (7) 2 3¼ 2 (3¼)2 (b) The Lagrange function L is given by L('; '_ ) = T ('; '_ ) ¡ U('): (8) The potential energy U of the half-cylinder is due to the elevation of its CM resulting from the deviation of ' from zero. -

Analysis of Nonlinear Dynamics in a Classical Transmon Circuit
Analysis of Nonlinear Dynamics in a Classical Transmon Circuit Sasu Tuohino B. Sc. Thesis Department of Physical Sciences Theoretical Physics University of Oulu 2017 Contents 1 Introduction2 2 Classical network theory4 2.1 From electromagnetic fields to circuit elements.........4 2.2 Generalized flux and charge....................6 2.3 Node variables as degrees of freedom...............7 3 Hamiltonians for electric circuits8 3.1 LC Circuit and DC voltage source................8 3.2 Cooper-Pair Box.......................... 10 3.2.1 Josephson junction.................... 10 3.2.2 Dynamics of the Cooper-pair box............. 11 3.3 Transmon qubit.......................... 12 3.3.1 Cavity resonator...................... 12 3.3.2 Shunt capacitance CB .................. 12 3.3.3 Transmon Lagrangian................... 13 3.3.4 Matrix notation in the Legendre transformation..... 14 3.3.5 Hamiltonian of transmon................. 15 4 Classical dynamics of transmon qubit 16 4.1 Equations of motion for transmon................ 16 4.1.1 Relations with voltages.................. 17 4.1.2 Shunt resistances..................... 17 4.1.3 Linearized Josephson inductance............. 18 4.1.4 Relation with currents................... 18 4.2 Control and read-out signals................... 18 4.2.1 Transmission line model.................. 18 4.2.2 Equations of motion for coupled transmission line.... 20 4.3 Quantum notation......................... 22 5 Numerical solutions for equations of motion 23 5.1 Design parameters of the transmon................ 23 5.2 Resonance shift at nonlinear regime............... 24 6 Conclusions 27 1 Abstract The focus of this thesis is on classical dynamics of a transmon qubit. First we introduce the basic concepts of the classical circuit analysis and use this knowledge to derive the Lagrangians and Hamiltonians of an LC circuit, a Cooper-pair box, and ultimately we derive Hamiltonian for a transmon qubit. -

Wave-Particle Duality Relation with a Quantum Which-Path Detector
entropy Article Wave-Particle Duality Relation with a Quantum Which-Path Detector Dongyang Wang , Junjie Wu * , Jiangfang Ding, Yingwen Liu, Anqi Huang and Xuejun Yang Institute for Quantum Information & State Key Laboratory of High Performance Computing, College of Computer Science and Technology, National University of Defense Technology, Changsha 410073, China; [email protected] (D.W.); [email protected] (J.D.); [email protected] (Y.L.); [email protected] (A.H.); [email protected] (X.Y.) * Correspondence: [email protected] Abstract: According to the relevant theories on duality relation, the summation of the extractable information of a quanton’s wave and particle properties, which are characterized by interference visibility V and path distinguishability D, respectively, is limited. However, this relation is violated upon quantum superposition between the wave-state and particle-state of the quanton, which is caused by the quantum beamsplitter (QBS). Along another line, recent studies have considered quantum coherence C in the l1-norm measure as a candidate for the wave property. In this study, we propose an interferometer with a quantum which-path detector (QWPD) and examine the generalized duality relation based on C. We find that this relationship still holds under such a circumstance, but the interference between these two properties causes the full-particle property to be observed when the QWPD system is partially present. Using a pair of polarization-entangled photons, we experimentally verify our analysis in the two-path case. This study extends the duality relation between coherence and path information to the quantum case and reveals the effect of quantum superposition on the duality relation.