Community-Based Upper Gastrointestinal Cancer Screening in a Randomized Controlled Trial
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Schistosomiasis in a Migrating Population in the Lake Region of China and Its Potential Impact on Control Operation
Acta Tropica 145 (2015) 88–92 Contents lists available at ScienceDirect Acta Tropica journal homepage: www.elsevier.com/locate/actatropica Schistosomiasis in a migrating population in the lake region of China and its potential impact on control operation Chun-li Cao a,∗∗, Zi-ping Bao a, Shi-zhu Li a, Wang-yuan Wei b, Ping Yi b, Qing Yu a, Hong-qing Zhu a, Jing Xu a, Jia-gang Guo a, Zheng Feng a,∗ a National Institute of Parasitic Diseases, Chinese Center for Disease Control and Prevention, WHO Collaborating Center for Malaria, Schistosomiasis and Filariasis, The Key laboratory of Parasite and Vector Biology, Ministry of Health, China, 207 Rui Jin Er Road, Shanghai 200025, People’s Republic of China b Hunan Institute of Schistosomiasis Control, Hunan, People’s Republic of China article info abstract Article history: Coverage of migrating people in schistosomiasis control program is a growing concern in China. Schis- Received 17 March 2014 tosomiasis caused by Schistosoma japonicum is still one of the major infectious diseases of public health Received in revised form 2 February 2015 importance in China though tremendous efforts have been made to control the transmission over the past Accepted 8 February 2015 decades. Along with the rapid social-economic development, migrant population has been remarkably Available online 18 February 2015 increasing across the country. The infected migrants may introduce a new souse of infection to endemic areas or the areas where the transmission had been controlled or interrupted but the intermediate host Keywords: Oncomelania snail is still present. Preliminary studies for surveillance on schistosomiasis prevalence in Schistosomiasis japonica Migrant population migrants were reported, but there is little basic information provided. -

Appendix Iii Property Valuation Report
THIS DOCUMENT IS IN DRAFT FORM, INCOMPLETE AND SUBJECT TO CHANGE AND THAT THE INFORMATION MUST BE READ IN CONJUNCTION WITH THE SECTION “WARNING” ON THE COVER OF THIS DOCUMENT. APPENDIX III PROPERTY VALUATION REPORT The following is the text of a letter and summary disclosure of values, prepared for the purpose of incorporation in this document received from Jones Lang LaSalle Corporate Appraisal and Advisory Limited, an independent valuer, in connection with its valuation as at 31 March 2019 of the selected property interests held by Zhongliang Holdings Group Company Limited. As described in section “Documents Available for Inspection” in Appendix III, a copy of full property valuation report will be made available for public inspection. Jones Lang LaSalle Corporate Appraisal and Advisory Limited 7/F One Taikoo Place 979 King’s Road Hong Kong tel +852 2846 5000 fax +852 2169 6001 Company Licence No.: C-030171 27 June 2019 The Board of Directors Zhongliang Holdings Group Company Limited 20/F, No.3 Shanghai Convention & Exhibition Center of International Sourcing 235 Yunling East Road Putuo District, Shanghai China Dear Sirs, In accordance with your instructions to value the selected property interests held by Zhongliang Holdings Group Company Limited (the “Company”) and its subsidiaries (hereinafter together referred to as the “Group”) in the People’s Republic of China (the “PRC”), we confirm that we have carried out inspections, made relevant enquiries and searches and obtained such further information as we consider necessary for the purpose of providing you with our opinion on the market values of the property interests as at 31 March 2019 (the “valuation date”). -

Impact of MBL and MASP-2 Gene Polymorphism and Its Interaction On
Chen et al. BMC Infectious Diseases (2015) 15:151 DOI 10.1186/s12879-015-0879-y RESEARCH ARTICLE Open Access Impact of MBL and MASP-2 gene polymorphism and its interaction on susceptibility to tuberculosis Mengshi Chen1,2, Ying Liang1,3, Wufei Li4, Mian Wang1,LiHu1,5, Benjamin Kwaku Abuaku1,6, Xin Huang1, Hongzhuan Tan1* and Shi Wu Wen1,7 Abstract Background: Mannose-binding lectin (MBL) and MBL-associated serine proteases 2 (MASP-2) are important proteins in the lectin pathway of the immune system. Polymorphism of MBL and MASP-2 genes may affect the serum concentration of MBL and MASP-2. This study explores the association between MBL and MASP-2 gene polymorphism and their interactions and the susceptibility to tuberculosis (TB). Method: A total of 503 patients with TB and 419 healthy controls were recruited to participate in this case-control study. PCR-SSP technology was applied to genotype rs7096206 of MBL genes and rs2273346 and rs6695096 of MASP-2 genes. Demographic data and some exposure informationwerealsoobtainedfromstudyparticipants. Unconditional logistic regression analysis was used to identify association between the various factors and TB whilst Marginal Structural Linear Odds Models were used to estimate the interactions. Results: Both genotype GC at rs7096206 of MBL genes and genotype TC at rs2273346 and rs6695096 of MASP-2 genes were more prevalent in the TB patient group than the healthy control group (P < 0.05, OR 1.393, 1.302 and 1.426 respectively). The relative excess risk of interaction (RERI) between rs7096206 of MBL genes and rs2273346 and rs6695096 of MASP-2 genes was 0.897 (95% CI: 0.282, 1.513) and 1.142 (95% CI: 0.755, 1.530) respectively (P < 0.05). -
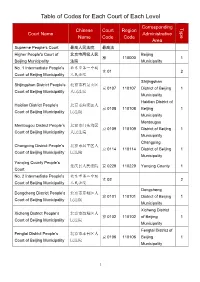
Table of Codes for Each Court of Each Level
Table of Codes for Each Court of Each Level Corresponding Type Chinese Court Region Court Name Administrative Name Code Code Area Supreme People’s Court 最高人民法院 最高法 Higher People's Court of 北京市高级人民 Beijing 京 110000 1 Beijing Municipality 法院 Municipality No. 1 Intermediate People's 北京市第一中级 京 01 2 Court of Beijing Municipality 人民法院 Shijingshan Shijingshan District People’s 北京市石景山区 京 0107 110107 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Haidian District of Haidian District People’s 北京市海淀区人 京 0108 110108 Beijing 1 Court of Beijing Municipality 民法院 Municipality Mentougou Mentougou District People’s 北京市门头沟区 京 0109 110109 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Changping Changping District People’s 北京市昌平区人 京 0114 110114 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Yanqing County People’s 延庆县人民法院 京 0229 110229 Yanqing County 1 Court No. 2 Intermediate People's 北京市第二中级 京 02 2 Court of Beijing Municipality 人民法院 Dongcheng Dongcheng District People’s 北京市东城区人 京 0101 110101 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Xicheng District Xicheng District People’s 北京市西城区人 京 0102 110102 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Fengtai District of Fengtai District People’s 北京市丰台区人 京 0106 110106 Beijing 1 Court of Beijing Municipality 民法院 Municipality 1 Fangshan District Fangshan District People’s 北京市房山区人 京 0111 110111 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Daxing District of Daxing District People’s 北京市大兴区人 京 0115 -

Annual Report 2019
HAITONG SECURITIES CO., LTD. 海通證券股份有限公司 Annual Report 2019 2019 年度報告 2019 年度報告 Annual Report CONTENTS Section I DEFINITIONS AND MATERIAL RISK WARNINGS 4 Section II COMPANY PROFILE AND KEY FINANCIAL INDICATORS 8 Section III SUMMARY OF THE COMPANY’S BUSINESS 25 Section IV REPORT OF THE BOARD OF DIRECTORS 33 Section V SIGNIFICANT EVENTS 85 Section VI CHANGES IN ORDINARY SHARES AND PARTICULARS ABOUT SHAREHOLDERS 123 Section VII PREFERENCE SHARES 134 Section VIII DIRECTORS, SUPERVISORS, SENIOR MANAGEMENT AND EMPLOYEES 135 Section IX CORPORATE GOVERNANCE 191 Section X CORPORATE BONDS 233 Section XI FINANCIAL REPORT 242 Section XII DOCUMENTS AVAILABLE FOR INSPECTION 243 Section XIII INFORMATION DISCLOSURES OF SECURITIES COMPANY 244 IMPORTANT NOTICE The Board, the Supervisory Committee, Directors, Supervisors and senior management of the Company warrant the truthfulness, accuracy and completeness of contents of this annual report (the “Report”) and that there is no false representation, misleading statement contained herein or material omission from this Report, for which they will assume joint and several liabilities. This Report was considered and approved at the seventh meeting of the seventh session of the Board. All the Directors of the Company attended the Board meeting. None of the Directors or Supervisors has made any objection to this Report. Deloitte Touche Tohmatsu (Deloitte Touche Tohmatsu and Deloitte Touche Tohmatsu Certified Public Accountants LLP (Special General Partnership)) have audited the annual financial reports of the Company prepared in accordance with PRC GAAP and IFRS respectively, and issued a standard and unqualified audit report of the Company. All financial data in this Report are denominated in RMB unless otherwise indicated. -

Annual Report 2019 Annual Report
Annual Report 2019 Annual Report 2019 For more information, please refer to : CONTENTS DEFINITIONS 2 Section I Important Notes 5 Section II Company Profile and Major Financial Information 6 Section III Company Business Overview 18 Section IV Discussion and Analysis on Operation 22 Section V Directors’ Report 61 Section VI Other Significant Events 76 Section VII Changes in Shares and Information on Shareholders 93 Section VIII Directors, Supervisors, Senior Management and Staff 99 Section IX Corporate Governance Report 119 Section X Independent Auditor’s Report 145 Section XI Consolidated Financial Statements 151 Appendix I Information on Securities Branches 276 Appendix II Information on Branch Offices 306 China Galaxy Securities Co., Ltd. Annual Report 2019 1 DEFINITIONS “A Share(s)” domestic shares in the share capital of the Company with a nominal value of RMB1.00 each, which is (are) listed on the SSE, subscribed for and traded in Renminbi “Articles of Association” the articles of association of the Company (as amended from time to time) “Board” or “Board of Directors” the board of Directors of the Company “CG Code” Corporate Governance Code and Corporate Governance Report set out in Appendix 14 to the Stock Exchange Listing Rules “Company”, “we” or “us” China Galaxy Securities Co., Ltd.(中國銀河證券股份有限公司), a joint stock limited company incorporated in the PRC on 26 January 2007, whose H Shares are listed on the Hong Kong Stock Exchange (Stock Code: 06881), the A Shares of which are listed on the SSE (Stock Code: 601881) “Company Law” -

Baseline Results in a Non-High-Incidence Area
Published OnlineFirst December 23, 2019; DOI: 10.1158/1940-6207.CAPR-19-0422 CANCER PREVENTION RESEARCH | RESEARCH ARTICLE Community-Based Upper Gastrointestinal Cancer Screening in a Randomized Controlled Trial: Baseline Results in a Non-high-incidence Area Hai-Fan Xiao1, Shi-Peng Yan1, Yan-fang Chen2, Zhaohui Shi1, Yan-Hua Zou1, Song-lin Zhu1, Ke-Kui Xu1, Ting Song2, and Xian-Zhen Liao1 ABSTRACT ◥ A cluster-randomized controlled trial (RCT) was con- arms, respectively. The participation rate was 74.3%. In the ducted to evaluate to the effectiveness of reducing mor- screening arm, 5,242 individuals (50.3%) were estimated tality of upper gastrointestinal cancer (UGC) and feasi- to be high-risk. Among them, 2,388 (45.6%) underwent bility of screening through a questionnaire combined with endoscopic screening. Age and household income were endoscopy in non-high-incidence urban areas in China. associated with undergoing endoscopy. Three early esoph- The trial design, recruitment performance, and prelimi- ageal cancer (0.13%), one gastric cancer (0.04%), 29 pre- nary results from baseline endoscopy are reported. Sev- cancerous esophageal lesions (1.21%), and 53 precancer- enty-five communities in two urban cities with a non- ous gastric lesions (2.22%) were detected. Age, sex, high-incidence of UGC were randomized to a screening a family history of cancer, intake of meat–egg–milk endoscopy arm (n ¼ 38) or a control arm (n ¼ 37). In the frequently, superficial gastritis, and clinical symptoms screening arm, individuals at high risk of UGC underwent of gastric cancer were associated with the presence of endoscopic screening. -

Hunan~Ce Prvice
. .. ... ..... ...... Public Disclosure Authorized Hunan~CePrvice PRC . .......... - Envieronm-yn t As se oflt. P Plant Phase 2 Extension Project (2 x 300MW), Hunan Province, PRC ... -, Public Disclosure Authorized ~~~~- F~~~~~~R?S~~~~~~~~:; F ., < - .~~~~~~~~~~~~~ ' - -P"*t ,;tmpany... '.:ebruay 199 Public Disclosure Authorized 7- r_e- PowerCowuU ~ ~ -, ,N,',-.I .~~ ,,,9-~~~~Fbur Public Disclosure Authorized COMMERCIALIN CONFIDENCE This reportwas preparedby POWERGENplc for China ElectricPower Technology Import & Export Corp.Hunan Electric Power Company under Contract No. 971JBJGB/000072 GB. Thiscontract was to assistHunan ElectricPower Company (HEPC) in the productionof an EnvironmentalAssessment Reportto meet World Bank requirements,in collaborationwith HEPCand NanjingElectric Power ResearchInstitute (NEPRI). The reportis mainlybased upon information supplied by HEPCand NEPRI NeitherPOWERGEN nor any personsacting on its behalf.(a) makesany warranty,express or implied, with respectto the use of any information,apparatus, method or processdisclosed in thisreport or that suchuse may not infringethe rightsof any thirdparty, or (b) assumesany liabilitieswith respectto the use of, or for damagesresulting in any way from the use of, any information,apparatus, method or processdisclosed in the report,except in so far as any warrantyor liabilityarises out of the termsand conditionsof theabove Contract. © PowerGenpic 1998 No part of this publicationmay be reproduced,stored in a retrievalsystem or transmitted,in any form or by any meanselectronic, -

Results Announcement for the Year Ended December 31, 2020
B_table indent_3.5 mm N_table indent_3 mm Hong Kong Exchanges and Clearing Limited, The Stock Exchange of Hong Kong Limited and Hong Kong Securities Clearing Company Limited take no responsibility for the contents of this announcement, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this announcement. (A joint stock company incorporated in the People’s Republic of China with limited liability under the Chinese corporate name “東方證券股份有限公司” and carrying on business in Hong Kong as “東方證券” (in Chinese) and “DFZQ” (in English)) (Stock Code: 03958) RESULTS ANNOUNCEMENT FOR THE YEAR ENDED DECEMBER 31, 2020 The board of directors (the “Board”) of 東方證券股份有限公司 (the “Company”) hereby announces the audited results of the Company and its subsidiaries (the “Group”) for the year ended December 31, 2020. This announcement, containing the full text of the 2020 annual report of the Company, complies with the relevant requirements of the Rules Governing the Listing of Securities on The Stock Exchange of Hong Kong Limited in relation to information to accompany preliminary announcement of annual results. The Group’s final results for the year ended December 31, 2020 have been reviewed by the audit committee of the Company. PUBLICATION OF ANNUAL RESULTS ANNOUNCEMENT AND ANNUAL REPORT This results announcement will be published on the website of The Stock Exchange of Hong Kong Limited (www.hkexnews.hk) and the Company’s website (www.dfzq.com.cn). The Company’s 2020 annual report will be despatched to holders of H shares and published on the websites of the Company and The Stock Exchange of Hong Kong Limited in due course. -

Annual Report 2017
ANNUAL REPORT 2017 * For identification purpose only CONTENTS Section I DEFINITIONS AND IMPORTANT RISK WARNINGS 3 Section II COMPANY PROFILE AND KEY FINANCIAL INDICATORS 7 Section III SUMMARY OF THE COMPANY’S BUSINESS 23 Section IV REPORT OF THE BOARD OF DIRECTORS 29 Section V SIGNIFICANT EVENTS 74 Section VI CHANGES IN ORDINARY SHARES AND PARTICULARS ABOUT SHAREHOLDERS 104 Section VII PREFERENCE SHARES 114 Section VIII PARTICULARS ABOUT DIRECTORS, SUPERVISORS, SENIOR MANAGEMENT AND EMPLOYEES 115 Section IX CORPORATE GOVERNANCE 164 Section X CORPORATE BONDS 202 Section XI FINANCIAL REPORT 212 Section XII DOCUMENTS AVAILABLE FOR INSPECTION 213 Section XIII INFORMATION DISCLOSURE OF SECURITIES COMPANY 214 HAITONG SECURITIES CO., LTD. Annual Report 2017 (H Share) 1 IMPORTANT NOTICE The Board, the Supervisory Committee, Directors, Supervisors and senior management of the Company warrant the truthfulness, accuracy and completeness of contents of this annual report (the “Report”) and that there is no false representation, misleading statement contained herein or material omission from this Report, for which they will assume joint and several liabilities. This Report was considered and approved at the 29th meeting of the sixth session of the Board. The number of Directors to attend the Board meeting should be 13 and the number of Directors who actually attended the Board meeting was 11. Ms. Yu Liping, a non-executive Director, and Mr. Feng Lun, an independent non-executive Director, were unable to attend the Board meeting in person due to business trips, and had appointed Ms. Zhang Xinmei, a non-executive Director, and Mr. Xiao Suining, an independent non-executive Director, to vote on their behalves respectively. -
2018 China Adult Tobacco Survey Report
2018 China Adult Tobacco Survey Report Contents Definition of indicators ................................................................................................................................... 5 Chapter 1 Introduction .................................................................................................................................... 6 Chapter 2 Survey methodology ...................................................................................................................... 9 2.1 Goals of study ...................................................................................................................................... 9 2.2 Target population ................................................................................................................................. 9 2.3 Inclusion criteria .................................................................................................................................. 9 2.4 Sampling design .................................................................................................................................. 9 2.5 Questionnaire survey .........................................................................................................................11 2.6 Data collection ...................................................................................................................................11 2.7 Statistical analysis .............................................................................................................................12 -

People's Procuratorate of Yueyanglou District, Yueyang City, Hunan
People's Court of Yueyanglou District, Yueyang, Hunan Judgement in a Criminal and Incidental Civil Action (2015) Lou Xing Yi Chu Zi No. 291 The public Procuratorial Organ: People's Procuratorate of Yueyanglou District, Yueyang. Plaintiff of the incidental civil action: Yueyang Municipal Forestry Bureau. Legal representative: He Zuoyun Entrusted agent: Gao Dali, Section Chief of Hunan East Dongting Lake National Nature Reserve Administration, a secondary institution under the Yueyang Municipal Forestry Bureau Defendant: He Jianqiang, nicknamed “He Laosi”, male, born on August 12, 1970, Hunan, Han Chinese, with an educational background of primary school, fisherman; detained on January 21 2015 and arrested on February 16 of the same year for illegal hunting and killing of endangered wildlife by Yueyang Forest Public Security Bureau; currently being detained at Yueyang Detention Center Attorney: Zhang Guohui, Hunan Yuezhou Law Firm. Defendant: Zhong Dejun, nicknamed “Jun Meizi”, male, born on December 22, 1965, Han Chinese, with an educational background of primary school, farmer; detained on January 21 2015 and arrested on February 16 of the same year for illegal acquiring, transporting and selling endangered wildlife and for illegal hunting by Yueyang Forest Public Security Bureau; currently being detained at Yueyang Detention Center. Defendant: Fang Jianhua, male, born on March 25, 1976, Han Chinese, with an educational background of primary school, farmer; detained on April 9 2015 and arrested on 16th of the same month for illegal acquiring,