Elixir Journal
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Structure of Pronephros and Development of Mesonephric Kidney in Larvae of Russian Sturgeon, Acipenser Gueldenstaedtii Brandt (Acipenseridae)
Zoologica5 PRONEPHROS Poloniae-AND (2012)-MESONEPHRIC 57/1-4: 5-20-KIDNEY-IN-LARVAE-OF-A.-GUELDENSTAEDTII 5 DOI: 10.2478/v10049-012-0001-6 STRUCTURE OF PRONEPHROS AND DEVELOPMENT OF MESONEPHRIC KIDNEY IN LARVAE OF RUSSIAN STURGEON, ACIPENSER GUELDENSTAEDTII BRANDT (ACIPENSERIDAE) L.S. KRAYUSHKINA*1, A.A. GERASIMOV1, A.A. KIRSANOV1, M.V. MOSYAGINA1, A. OGORZA£EK2 1Department of Ichthyology and Hydrobiology, St. Petersburg State University, 16-th Line 29, 199178, St. Petersburg, Russia, [email protected] 2 Department of Animal Developmental Biology, Zoological Institute, University of Wroclaw, Sienkiewicza 21, 50-335 Wroclaw, Poland. *Corresponding author Abstract. The structure of the pronephros and development of mesonephric kidney in Russian sturgeon larvae, Acipenser gueldenstaedtii Brandt at different stages of early postembryonic development (from hatching to 14 days), were studied with histological and electronic microscopy methods. The larval pronephros is represented by the system of bilaterally located pronephric tubules with ciliated nephrostomes and funnels and exog- enous single glomus, which is not integrated directly into pronephric tubules and located in the pronephric chamber. The glomus is positioned below the dorsal aorta and vascular- ized by its capillaries. The glomus has the same features of the thin structure that are typical of and necessary for the function of a filtering organ. The structure of the prone- phros in acipenserids is discussed and compared with teleosts and amphibians. Histogen- esis of the mesonephric kidney is observed during the period of pronephros functioning; it is complete by the time the larvae transfer to exogenous feeding. At this moment, the pronephros undergoes significant structural degradation. -

Stem Cells in the Embryonic Kidney R Nishinakamura1
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector http://www.kidney-international.org mini review & 2008 International Society of Nephrology Stem cells in the embryonic kidney R Nishinakamura1 1Division of Integrative Cell Biology, Institute of Molecular Embryology and Genetics, Kumamoto University, 2-2-1 Honjo, Kumamoto, Japan The mammalian kidney, the metanephros, is formed by a STRATEGY TOWARD KIDNEY RECONSTITUTION USING reciprocally inductive interaction between two precursor PROGENITOR CELLS tissues, the metanephric mesenchyme and the ureteric bud. Stem cells are defined by two criteria: self-renewal and The ureteric bud induces the metanephric mesenchyme to multipotency. Few reports in the kidney field have addressed differentiate into the epithelia of glomeruli and renal tubules. both of these criteria at a clonal level, so it is better to use the Multipotent renal progenitors that form colonies upon Wnt4 term ‘progenitor’ rather than ‘stem cells.’ In this review, renal stimulation and strongly express Sall1 exist in the progenitors in the embryonic kidney, not those in the adult metanephric mesenchyme; these cells can partially kidney, from the viewpoint of developmental biology and reconstitute a three-dimensional structure in an organ stem/progenitor cell biology will be discussed. To generate culture setting. Six2 maintains this mesenchymal progenitor multiple cell lineages for kidney regeneration, the identifica- population by opposing Wnt4-mediated epithelialization. tion of renal progenitors is a prerequisite. Furthermore, there Upon epithelial tube formation, Notch2 is required for the exist three obstacles to be overcome: (1) derivation of the differentiation of proximal nephron structures (podocyte and renal progenitors; (2) expansion of the renal progenitors; and proximal tubules). -

Vocabulario De Morfoloxía, Anatomía E Citoloxía Veterinaria
Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) Servizo de Normalización Lingüística Universidade de Santiago de Compostela COLECCIÓN VOCABULARIOS TEMÁTICOS N.º 4 SERVIZO DE NORMALIZACIÓN LINGÜÍSTICA Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) 2008 UNIVERSIDADE DE SANTIAGO DE COMPOSTELA VOCABULARIO de morfoloxía, anatomía e citoloxía veterinaria : (galego-español- inglés) / coordinador Xusto A. Rodríguez Río, Servizo de Normalización Lingüística ; autores Matilde Lombardero Fernández ... [et al.]. – Santiago de Compostela : Universidade de Santiago de Compostela, Servizo de Publicacións e Intercambio Científico, 2008. – 369 p. ; 21 cm. – (Vocabularios temáticos ; 4). - D.L. C 2458-2008. – ISBN 978-84-9887-018-3 1.Medicina �������������������������������������������������������������������������veterinaria-Diccionarios�������������������������������������������������. 2.Galego (Lingua)-Glosarios, vocabularios, etc. políglotas. I.Lombardero Fernández, Matilde. II.Rodríguez Rio, Xusto A. coord. III. Universidade de Santiago de Compostela. Servizo de Normalización Lingüística, coord. IV.Universidade de Santiago de Compostela. Servizo de Publicacións e Intercambio Científico, ed. V.Serie. 591.4(038)=699=60=20 Coordinador Xusto A. Rodríguez Río (Área de Terminoloxía. Servizo de Normalización Lingüística. Universidade de Santiago de Compostela) Autoras/res Matilde Lombardero Fernández (doutora en Veterinaria e profesora do Departamento de Anatomía e Produción Animal. -

Secreted Molecules in Metanephric Induction
J Am Soc Nephrol 11: S116–S119, 2000 Secreted Molecules in Metanephric Induction THOMAS J. CARROLL and ANDREW P. McMAHON Department of Molecular and Cellular Biology, Biological Laboratories, Harvard University, Cambridge, Massachusetts. Abstract. Nearly 50 yr ago, Clifford Grobstein made the ob- the classic model of metanephric induction. The studies of the servation that the ureteric bud induced the nephrogenic mes- classic ureteric inducer performed to date have most likely enchyme to undergo tubulogenesis. Since that discovery, sci- been characterizations of a mesenchyme-specific inducer, entists have attempted to characterize the molecular nature of Wnt-4, and its role in tubulogenesis. Ureteric induction most the inducer. To date, no single molecule that is both necessary likely involves a series of distinct events that provide prolif- and sufficient for nephric induction has been identified. Be- erative, survival, and condensation signals to the mesenchyme, cause of recent insights regarding the role of several secreted integrating the growth of the ureteric system with tubulogen- molecules in tubulogenesis, it has become necessary to revise esis. The developmental biologic processes of the kidney have been logenesis. The conclusion drawn from this discovery was that the subject of intense study for more than 100 yr (for review, the ureteric bud induces tubulogenesis within the surrounding see reference (1). All three vertebrate kidney types (pro- mesenchyme. During further investigation, it was discovered nephros, mesonephros, and metanephros) are derivatives of a that a number of tissues, including, most notably, a dorsal region of the embryo known as the intermediate mesoderm. In portion of the embryonic spinal cord, are able to substitute for mice, a portion of the mesonephric duct, known as the meta- the ureter in this inductive interaction. -

Glomus Specification and Induction in Xenopus
Development 126, 5847-5856 (1999) 5847 Printed in Great Britain © The Company of Biologists Limited 1999 DEV6378 The specification and growth factor inducibility of the pronephric glomus in Xenopus laevis Hannah C. Brennan, Sarbjit Nijjar and Elizabeth A. Jones* Cell and Molecular Development Group, Department of Biological Sciences, Warwick University, Coventry, CV4 7AL, UK *Author for correspondence (e-mail: [email protected]) Accepted 23 September; published on WWW 24 November 1999 SUMMARY We report a study on the specification of the glomus, the Furthermore, we have analysed the growth factor filtration device of the amphibian pronephric kidney, using inducibility of the glomus in the presence or absence of an explant culturing strategy in Xenopus laevis. Explants of retinoic acid (RA) by RT-PCR. We define for the first time presumptive pronephric mesoderm were dissected from the conditions under which these growth factors induce embryos of mid-gastrula to swimming tadpole stages. These glomus tissue in animal cap tissue. Activin together with explants were cultured within ectodermal wraps and high concentrations of RA can induce glomus tissue from analysed by RT-PCR for the presence of the Wilm’s Tumour- animal cap ectoderm. Unlike the pronephric tubules, the 1 gene, xWT1, a marker specific for the glomus at the stages glomus can also be induced by FGF and RA. analysed, together with other mesodermal markers. We show that the glomus is specified at stage 12.5, the same stage at which pronephric tubules are specified. We have previously shown that pronephric duct is specified somewhat later, at stage 14. -

Urinary System Intermediate Mesoderm
Urinary System Intermediate mesoderm lateral mesoderm: somite ectoderm neural NOTE: Intermediate mesoderm splanchnic groove somatic is situated between somites and lateral mesoderm (somatic and splanchnic mesoderm bordering the coelom). All mesoderm is derived from the primary mesen- intermediate mesoderm endoderm chyme that migrated through the notochord coelom (becomes urogenital ridge) primitive streak. Intermediate mesoderm (plus adjacent mesothelium lining the coelom) forms a urogenital ridge, which consists of a laterally-positioned nephrogenic cord (that forms kidneys & ureter) and a medially-positioned gonadal ridge (for ovary/testis & female/male genital tract formation). Thus urinary & genital systems have a common embryonic origin; also, they share common ducts. NOTE: Urine production essentially requires an increased capillary surface area (glomeruli), epithelial tubules to collect plasma filtrate and extract desirable constituents, and a duct system to convey urine away from the body. Kidneys Bilateraly, three kid- mesonephric duct neys develop from the neph- metanephros pronephros rogenic cord. They develop mesonephric tubules chronologically in cranial- mesonephros caudal sequence, and are designated pro—, meso—, Nephrogenic Cord (left) and meta—, respectively. cloaca The pronephros and mesonephros develop similarly: the nephrogenic cord undergoes seg- mentation, segments become tubules, tubules drain into a duct & eventually tubules disintegrate. spinal ganglion 1] Pronephros—consists of (7-8) primitive tubules and a pronephric duct that grows caudally and terminates in the cloaca. The tubules soon degenerate, but the pronephric duct persists as the neural tube mesonephric duct. (The pronephros is not functional, somite except in sheep.) notochord mesonephric NOTE tubule The mesonephros is the functional kidney for fish and am- aorta phibians. The metanephros is the functional kidney body of reptiles, birds, & mammals. -

The Derivatives of Three-Layered Embryo (Germ Layers)
HUMANHUMAN EMBRYOLOGYEMBRYOLOGY Department of Histology and Embryology Jilin University ChapterChapter 22 GeneralGeneral EmbryologyEmbryology FourthFourth week:week: TheThe derivativesderivatives ofof trilaminartrilaminar germgerm discdisc Dorsal side of the germ disc. At the beginning of the third week of development, the ectodermal germ layer has the shape of a disc that is broader in the cephalic than the caudal region. Cross section shows formation of trilaminar germ disc Primitive pit Drawing of a sagittal section through a 17-day embryo. The most cranial portion of the definitive notochord has formed. ectoderm Schematic view showing the definitive notochord. horizon =ectoderm hillside fields =neural plate mountain peaks =neural folds Cave sinks into mountain =neural tube valley =neural groove 7.1 Derivatives of the Ectodermal Germ Layer 1) Formation of neural tube Notochord induces the overlying ectoderm to thicken and form the neural plate. Cross section Animation of formation of neural plate When notochord is forming, primitive streak is shorten. At meanwhile, neural plate is induced to form cephalic to caudal end, following formation of notochord. By the end of 3rd week, neural folds and neural groove are formed. Neural folds fuse in the midline, beginning in cervical region and Cross section proceeding cranially and caudally. Neural tube is formed & invade into the embryo body. A. Dorsal view of a human embryo at approximately day 22. B. Dorsal view of a human embryo at approximately day 23. The nervous system is in connection with the amniotic cavity through the cranial and caudal neuropores. Cranial/anterior neuropore Neural fold heart Neural groove endoderm caudal/posterior neuropore A. -

Embryology of the Kidney Rizaldy Paz Scott | Yoshiro Maezawa | Jordan Kreidberg | Susan E
1 Embryology of the Kidney Rizaldy Paz Scott | Yoshiro Maezawa | Jordan Kreidberg | Susan E. Quaggin CHAPTER OUTLINE MAMMALIAN KIDNEY DEVELOPMENT, 2 MOLECULAR GENETICS OF MODEL SYSTEMS TO STUDY KIDNEY NEPHROGENESIS, 22 DEVELOPMENT, 8 GENETIC ANALYSIS OF MAMMALIAN KIDNEY DEVELOPMENT, 15 KEY POINTS • The development of the kidney relies on reciprocal signaling and inductive interactions between neighboring cells. • Epithelial cells that comprise the tubular structures of the kidney are derived from two distinct cell lineages: the ureteric epithelia lineage that branches and gives rise to collecting ducts and the nephrogenic mesenchyme lineage that undergoes mesenchyme to epithelial transition to form connecting tubules, distal tubules, the loop of Henle, proximal tubules, parietal epithelial cells, and podocytes. • Nephrogenesis and nephron endowment requires an epigenetically regulated balance between nephron progenitor self-renewal and epithelial differentiation. • The timing of incorporation of nephron progenitor cells into nascent nephrons predicts their positional identity within the highly patterned mature nephron. • Stromal cells and their derivatives coregulate ureteric branching morphogenesis, nephrogenesis, and vascular development. • Endothelial cells track the development of the ureteric epithelia and establish the renal vasculature through a combination of vasculogenic and angiogenic processes. • Collecting duct epithelia have an inherent plasticity enabling them to switch between principal and intercalated cell identities. MAMMALIAN KIDNEY DEVELOPMENT The filtration function of the kidneys is accomplished by basic units called nephrons (Fig. 1.1). Humans on average have 1 million nephrons per adult kidney but the range of ANATOMIC OVERVIEW OF THE 4 MAMMALIAN KIDNEY total nephrons is highly variable across human populations. Each mouse kidney may contain up to 12,000–16,000 nephrons The kidney is a sophisticated, highly vascularized organ that depending on the strain.5 This wide range in nephron number plays a central role in overall body homeostasis. -
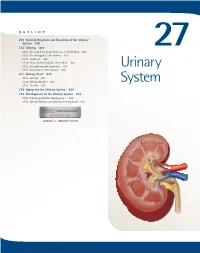
Urinary System
OUTLINE 27.1 General Structure and Functions of the Urinary System 818 27.2 Kidneys 820 27 27.2a Gross and Sectional Anatomy of the Kidney 820 27.2b Blood Supply to the Kidney 821 27.2c Nephrons 824 27.2d How Tubular Fluid Becomes Urine 828 27.2e Juxtaglomerular Apparatus 828 Urinary 27.2f Innervation of the Kidney 828 27.3 Urinary Tract 829 27.3a Ureters 829 27.3b Urinary Bladder 830 System 27.3c Urethra 833 27.4 Aging and the Urinary System 834 27.5 Development of the Urinary System 835 27.5a Kidney and Ureter Development 835 27.5b Urinary Bladder and Urethra Development 835 MODULE 13: URINARY SYSTEM mck78097_ch27_817-841.indd 817 2/25/11 2:24 PM 818 Chapter Twenty-Seven Urinary System n the course of carrying out their specific functions, the cells Besides removing waste products from the bloodstream, the uri- I of all body systems produce waste products, and these waste nary system performs many other functions, including the following: products end up in the bloodstream. In this case, the bloodstream is ■ Storage of urine. Urine is produced continuously, but analogous to a river that supplies drinking water to a nearby town. it would be quite inconvenient if we were constantly The river water may become polluted with sediment, animal waste, excreting urine. The urinary bladder is an expandable, and motorboat fuel—but the town has a water treatment plant that muscular sac that can store as much as 1 liter of urine. removes these waste products and makes the water safe to drink. -

Equine Placenta – Marvelous Organ and a Lethal Weapon
Equine placenta – marvelous organ and a lethal weapon Malgorzata Pozor, DVM, PhD, Diplomate ACT Introduction Placenta has been defined as: „an apposition between parent (usually maternal) and fetal tissue in order to establish physiological exchange” (1). Another definition of this important organ was proposed by Steven and Morris: „a device consisting of one or more transport epithelia located between fetal and maternal blood supply” (2). The main function of placenta is to provide an interface between the dam and the the fetus and to allow the metabolic exchange of the the nutrients, oxygen and waste material. The maternal circulation is brought into a close apposition to the fetal circulation, while a separation of these two circulatory systems remain separated (3). A degree and complexity of this „intimate relationship” varies greately between species mostly due to the structural diversity of the extraembryonic membranes of the vertebrates. The early feto-maternal exchange in the equine pregnancy is established as early as on day 22 after fertilization. The fetal and choriovitellin circulations are already present, the capsule ruptures and the allantois is already visible (4). The allantois starts expanding by day 32 and vascularizes approximately 90% of the chorion and fuses with it to form chorioallantois by day 38 of gestation (5). The equine placenta continues increasing its complexity till approximately day 150 of gestation. Equids have epitheliochorial placenta, there are six leyers separating maternal and fetal circulation, and there are no erosion of the luminal, maternal epithelium, like in ruminants (6). Thousands of small chorionic microvilli develop and penetrate into endometrial invaginations. -

4 Extraembryonic Membranes
Implantation, Extraembryonic Membranes, Placental Structure and Classification A t t a c h m e n t and Implantation Implantation is the first stage in development of the placenta. In most cases, implantation is preceded by a close interaction of embryonic trophoblast and endometrial epithelial cells that is known as adhesion or attachment. Implantation also is known as the stage where the blastocyst embeds itself in the endometrium, the inner membrane of the uterus. This usually occurs near the top of the uterus and on the posterior wall. Among other things, attachment involves a tight intertwining of microvilli on the maternal and embryonic cells. Following attachment, the blastocyst is no longer easily flushed from the lumen of the uterus. In species that carry multiple offspring, attachment is preceeded by a remarkably even spacing of embryos through the uterus. This process appears to result from uterine contractions and in some cases involves migration of embryos from one uterine horn to another (transuterine migration). The effect of implantation in all cases is to obtain very close apposition between embryonic and maternal tissues. There are, however, substantial differences among species in the process of implantation, particularly with regard to "invasiveness," or how much the embryo erodes into maternal tissue. In species like horses and pigs, attachment and implantation are essentially equivalent. In contrast, implantation in humans involves the embryo eroding deeply into the substance of the uterus. •Centric: the embryo expands to a large size before implantation, then remains in the center of the uterus. Examples include carnivores, ruminants, horses, and pigs. •Eccentric: The blastocyst is small and implants within the endometrium on the side of the uterus, usually opposite to the mesometrium. -
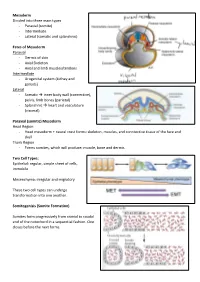
Mesoderm Divided Into Three Main Types - Paraxial (Somite) - Intermediate - Lateral (Somatic and Splanchnic)
Mesoderm Divided into three main types - Paraxial (somite) - Intermediate - Lateral (somatic and splanchnic) Fates of Mesoderm Paraxial - Dermis of skin - Axial Skeleton - Axial and limb muscles/tendons Intermediate - Urogenital system (kidney and gonads) Lateral - Somatic inner body wall (connective), pelvis, limb bones (parietal) - Splanchnic heart and vasculature (visceral) Paraxial (somitic) Mesoderm Head Region - Head mesoderm + neural crest forms: skeleton, muscles, and conntective tissue of the face and skull Trunk Region - Forms somites, which will produce: muscle, bone and dermis Two Cell Types: Epithelial: regular, simple sheet of cells, immobile Mesenchyma: irregular and migratory These two cell types can undergo transformation into one another. Somitogenisis (Somite Formation) Somites form progressively from cranial to caudal end of the notochord in a sequential fashion. One closes before the next forms. Somite Differentiation The somite splits into the epithelial dermamyotome (dermis/muscle) and the messenchymal sclerotome (skeletal). The somite is all paraxial mesoderm. Somite location determines the fate of its associates derma/myo/sclerotomes. Intermediate Mesoderm Urogenital system: - Kidneys - Gonads - Reproductive Duct Systems Runs alongside the paraxial mesoderm. Urogenital System Along mesonephric duct: - Pronephros, mesonephros, and metanephros - Pronephros fall away as gonad develops on ventral-medial side of mesonephros. - Metanephrogenic mesenchyme gives rise to kidney. The mesonephric duct will become the Wolffian duct forming at the nephric bud. The Mullerian duct forms via an invagination on the dorsal side of the nephric duct. The gonad will degenerate one of the two ducts depending on the hormones it produces. XX degenerates Wolffian duct – no testosterone, anti-Mullerian hormone (AMH) not produced, and Mullerian duct can develop in addition to female reproductive organs (ovaries, vagina) XY degenerates Mullerian duct – testosterone, AMH produced, Wolffian duct continues as male reproductive organs (testes, penis) develop.