What Is a Chemical Formula? Empirical Formula
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
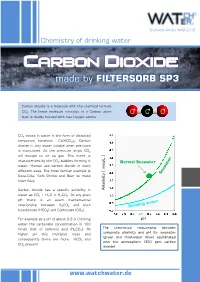
Carbon Dioxide - Made by FILTERSORB SP3
Technical Article MAR 2015 Chemistry of drinking water Carbon dioxide - made by FILTERSORB SP3 Carbon dioxide is a molecule with the chemical formula CO2. The linear molecule consists of a Carbon atom O C O that is doubly bonded with two Oxygen atoms. CO2 exists in water in the form of dissolved temporary hardness Ca(HCO3)2. Carbon dioxide is only water soluble when pressure is maintained. As the pressure drops CO2 will escape to air as gas. This event is characterized by the CO2 bubbles forming in /L ] Normal Seawater water. Human use carbon dioxide in many meq different ways. The most familiar example is Coca-Cola, Soft Drinks and Beer to make them fizzy. Carbon dioxide has a specific solubility in [ Alkalinity water as CO2 + H2O ⇌ H2CO3. At any given pH there is an exact mathematical relationship between H2CO3 and both bicarbonate (HCO3) and Carbonate (CO3). For example at a pH of about 9.3 in Drinking pH water the carbonate concentration is 100 The theoretical relationship between times that of carbonic acid (H2CO3). At carbonate alkalinity and pH for seawater higher pH this multiplier rises and (green and freshwater (blue) equilibrated consequently there are more HCO and 3 with the atmosphere (350 ppm carbon CO present 3 dioxide) www.watchwater.de Water ® Technology FILTERSORB SP3 WATCH WATER & Chemicals Making the healthiest water How SP3 Water functions in human body ? Answer: Carbon dioxide is a waste product of the around these hollow spaces will spasm and respiratory system, and of several other constrict. chemical reactions in the body such as the Transportation of oxygen to the tissues: Oxygen creation of ATP. -

A Complete Guide to Benzene
A Complete Guide to Benzene Benzene is an important organic chemical compound with the chemical formula C6H6. The benzene molecule is composed of six carbon atoms joined in a ring with one hydrogen atom attached to each. As it contains only carbon and hydrogen atoms, benzene is classed as a hydrocarbon. Benzene is a natural constituent of crude oil and is one of the elementary petrochemicals. Due to the cyclic continuous pi bond between the carbon atoms, benzene is classed as an aromatic hydrocarbon, the second [n]- annulene ([6]-annulene). It is sometimes abbreviated Ph–H. Benzene is a colourless and highly flammable liquid with a sweet smell. Source: Wikipedia Protecting people and the environment Protecting both people and the environment whilst meeting the operational needs of your business is very important and, if you have operations in the UK you will be well aware of the requirements of the CoSHH Regulations1 and likewise the Code of Federal Regulations (CFR) in the US2. Similar legislation exists worldwide, the common theme being an onus on hazard identification, risk assessment and the provision of appropriate control measures (bearing in mind the hierarchy of controls) as well as health surveillance in most cases. And whilst toxic gasses such as hydrogen sulphide and carbon monoxide are a major concern because they pose an immediate (acute) danger to life, long term exposure to relatively low level concentrations of other gasses or vapours such as volatile organic compounds (VOC) are of equal importance because of the chronic illnesses that can result from that ongoing exposure. Benzene, a common VOC Organic means the chemistry of carbon based compounds, which are substances that results from a combination of two or more different chemical elements. -

Chemical Formula
Chemical Formula Jean Brainard, Ph.D. Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) AUTHOR Jean Brainard, Ph.D. To access a customizable version of this book, as well as other interactive content, visit www.ck12.org CK-12 Foundation is a non-profit organization with a mission to reduce the cost of textbook materials for the K-12 market both in the U.S. and worldwide. Using an open-content, web-based collaborative model termed the FlexBook®, CK-12 intends to pioneer the generation and distribution of high-quality educational content that will serve both as core text as well as provide an adaptive environment for learning, powered through the FlexBook Platform®. Copyright © 2013 CK-12 Foundation, www.ck12.org The names “CK-12” and “CK12” and associated logos and the terms “FlexBook®” and “FlexBook Platform®” (collectively “CK-12 Marks”) are trademarks and service marks of CK-12 Foundation and are protected by federal, state, and international laws. Any form of reproduction of this book in any format or medium, in whole or in sections must include the referral attribution link http://www.ck12.org/saythanks (placed in a visible location) in addition to the following terms. Except as otherwise noted, all CK-12 Content (including CK-12 Curriculum Material) is made available to Users in accordance with the Creative Commons Attribution-Non-Commercial 3.0 Unported (CC BY-NC 3.0) License (http://creativecommons.org/ licenses/by-nc/3.0/), as amended and updated by Creative Com- mons from time to time (the “CC License”), which is incorporated herein by this reference. -

Introduction, Formulas and Spectroscopy Overview Problem 1
Chapter 1 – Introduction, Formulas and Spectroscopy Overview Problem 1 (p 12) - Helpful equations: c = ()() and = (1 / ) so c = () / ( ) c = 3.00 x 108 m/sec = 3.00 x 1010 cm/sec a. Which photon of electromagnetic radiation below would have the longer wavelength? Convert both values to meters. -1 = 3500 cm = 4 x 1014 Hz = (1/) = (c / ) = (1/ ) = (3.0 x 108 m/s) / (4 x 1014 s-1) = (1/ cm-1)(1 m/100 cm) = 2.9 x 10-6 m = 7.5 x 10-7 m = longer wavelength = lower energy b. Which photon of electromagnetic radiation below would have the higher frequency? Convert both values to Hz. = 400 cm-1 = 300 nm = (1/ ) = 1/ [(400 cm-1)(100 cm / 1 m)] = (c/) 8 -5 9 = 2.5 x 10-5 m = (3.0 x 10 m/s) / (2.5 x 10 nm)(1m / 10 nm) v = (3.0 x 108 m/s) / (300 nm)(1m / 109 nm) = (c/) v = 1.0 x x 1015 s-1 = (3.0 x 108 m/s) / (2.5 x 10-5 m) v = 1.2 x 1013 s-1 higher frequency c. Which photon of electromagnetic radiation below would have the smaller wavenumber? Convert both values to cm-1. = 1 m = 6 x 1010 Hz = (1 / ) -1 = ( / c) = (1 / 1m) = 1 m-1 100 cm = 100 cm-1 100 cm-1 -1 = (6 x 1010 s-1 / 3.0 x 108 m/s) = 200 m-1 -1 1 m -1 = 20,000 cm smaller wavenumber 1 m Problem 2 (p 14) - Order the following photons from lowest to highest energy (first convert each value to kjoules/mole). -

Third-Generation Synchrotron X-Ray Diffraction of 6- M Crystal of Raite, Na
Proc. Natl. Acad. Sci. USA Vol. 94, pp. 12263–12267, November 1997 Geology Third-generation synchrotron x-ray diffraction of 6-mm crystal of raite, 'Na3Mn3Ti0.25Si8O20(OH)2z10H2O, opens up new chemistry and physics of low-temperature minerals (crystal structureymicrocrystalyphyllosilicate) JOSEPH J. PLUTH*, JOSEPH V. SMITH*†,DMITRY Y. PUSHCHAROVSKY‡,EUGENII I. SEMENOV§,ANDREAS BRAM¶, CHRISTIAN RIEKEL¶,HANS-PETER WEBER¶, AND ROBERT W. BROACHi *Department of Geophysical Sciences, Center for Advanced Radiation Sources, GeologicalySoilyEnvironmental, and Materials Research Science and Engineering Center, 5734 South Ellis Avenue, University of Chicago, Chicago, IL 60637; ‡Department of Geology, Moscow State University, Moscow, 119899, Russia; §Fersman Mineralogical Museum, Russian Academy of Sciences, Moscow, 117071, Russia; ¶European Synchrotron Radiation Facility, BP 220, 38043, Grenoble, France; and UOP Research Center, Des Plaines, IL 60017 Contributed by Joseph V. Smith, September 3, 1997 ABSTRACT The crystal structure of raite was solved and the energy and metal industries, hydrology, and geobiology. refined from data collected at Beamline Insertion Device 13 at Raite lies in the chemical cooling sequence of exotic hyperal- the European Synchrotron Radiation Facility, using a 3 3 3 3 kaline rocks of the Kola Peninsula, Russia, and the 65 mm single crystal. The refined lattice constants of the Monteregian Hills, Canada (2). This hydrated sodium- monoclinic unit cell are a 5 15.1(1) Å; b 5 17.6(1) Å; c 5 manganese silicate extends the already wide range of manga- 5.290(4) Å; b 5 100.5(2)°; space group C2ym. The structure, nese crystal chemistry (3), which includes various complex including all reflections, refined to a final R 5 0.07. -

Chemistry: Empirical & Molecular Formulas
Chemistry: Empirical & Molecular Formulas Name______________________ 1) What is the empirical formula for C15H30O5H5? 2) A compound has an empirical formula of C2H4O3N. Its molar mass is 359 g. What is the molecular formula? 3) A compound contains 12.7 g iron & 7.29 g oxygen. Its molar mass is 87.85 g. What is its empirical formula? What is its molecular formula? 4) A compound contains 65.6% carbon, 9.45% hydrogen & 25.0% oxygen. Its molar mass is 1922.85 g. What is its empirical formula? What is its molecular formula? 5) A compound contains 92.2% carbon & 7.75% hydrogen. It has a molar mass of 118 g. What is its empirical formula? What is its molecular formula? 6) A compound has an empirical formula of C2H5O3N. Its molar mass is 454 g. What is its molecular formula? 7) What is the empirical formula for C20H32O4N4? 8) A compound contains 69.6% carbon, 9.76% hydrogen & 20.6% oxygen. Its molar mass is 465.72 g. What is its empirical formula? What is its molecular formula? 9) A compound contains 42.044 g carbon & 7.956 g hydrogen. Its molar mass is 114.26 g. What is its empirical formula? What is its molecular formula? 10) A compound contains 30.79 g copper & 19.31 g oxygen. Its molar mass is 207 g. What is its empirical formula? What is its molecular formula? 11) A compound contains 40.9% carbon, 4.58% hydrogen & 54.5% oxygen. Its molar mass is 176.1 g. What is its empirical formula? What is its molecular formula? 12) A compound has an empirical formula of CH2O & a gram molecular mass of 180.16 g. -

11.2 Alkanes
11.2 Alkanes A large number of carbon compounds are possible because the covalent bond between carbon atoms, such as those in hexane, C6H14, are very strong. Learning Goal Write the IUPAC names and draw the condensed structural formulas and skeletal formulas for alkanes and cycloalkanes. Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc. Naming Alkanes Alkanes • are hydrocarbons that contain only C—C and C—H bonds • are formed by a continuous chain of carbon atoms • are named using the IUPAC (International Union of Pure and Applied Chemistry) system • have names that end in ane • use Greek prefixes to name carbon chains with five or more carbon atoms Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc. IUPAC Naming of First Ten Alkanes Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc. 1 Condensed Structural Formulas In a condensed structural formula, • each carbon atom and its attached hydrogen atoms are written as a group • a subscript indicates the number of hydrogen atoms bonded to each carbon atom The condensed structural formula of butane has four carbon atoms. CH3—CH2—CH2—CH3 butane Core Chemistry Skill Naming and Drawing Alkanes Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc. Condensed Structural Formulas Alkanes are written with structural formulas that are • expanded to show each bond • condensed to show each carbon atom and its attached hydrogen atoms Expanded Condensed Expanded Condensed Chemistry: An Introduction to General, Organic, and Biological Chemistry, Twelfth Edition © 2015 Pearson Education, Inc. -

WS 4.2: Empirical Formula Problems
Name: Answer Key Period: ______ Date: __________ Chem B odds for HW and evens for review WS 4.2: Empirical Formula Problems Directions: Determine the empirical formula for the following substances. If a molecular formula cannot be reduced, write “cannot be reduced in the box for empirical formula” Molecular Formula Empirical Formula Molecular Formula Empirical Formula 1) C6H6 CH 2) C10H18 C5H9 3) C8H18 C4H9 4) C7H15O2 Not reducible 5) WO2 Not reducible 6) N2H4 NH2 7) C2H6O2 CH3O 8) P3F6 PF2 9) X39Y13 X3Y 10) IF5 Not reducible 11) C4H10 C2H5 12) N2O5 Not reducible 13) C6H12O6 CH2O 14) N2O4 NO2 15) P2F4 PF2 16) SF6 Not reducible 17) SO3 Not reducible 18) N2Cl4 NCl2 19) N2Br4 NBr2 20) P2O5 Not reducible Directions: Solve the following problems. You may need to use a separate sheet of paper. 1) What is the empirical formula for a compound which 3) Barry Um has a sample of a compound which weighs 200 contains 0.0134 g of iron, 0.00769 g of sulfur and 0.0115 g of grams and contains only carbon, hydrogen, oxygen and oxygen? nitrogen. By analysis, he finds that it contains 97.56 grams of carbon, 4.878 g of hydrogen, 52.03 g of oxygen and 45.53 g of nitrogen. Find its empirical formula. 3 FeSO C5H3N2O2 2) A compound is found to contain 53.7 % iron and 46.27 % sulfur. Find its empirical formula. 4) What is the empirical formula for a compound which contains 80.3% zinc and the rest is oxygen? Fe2S3 ZnO 5) Rubbing alcohol was found to contain 60.0 % carbon, 13.4 9) Give the name and empirical formula of a compound % hydrogen, and the remaining mass was due to oxygen. -

TSCA Inventory Representation for Products Containing Two Or More
mixtures.txt TOXIC SUBSTANCES CONTROL ACT INVENTORY REPRESENTATION FOR PRODUCTS CONTAINING TWO OR MORE SUBSTANCES: FORMULATED AND STATUTORY MIXTURES I. Introduction This paper explains the conventions that are applied to listings of certain mixtures for the Chemical Substance Inventory that is maintained by the U.S. Environmental Protection Agency (EPA) under the Toxic Substances Control Act (TSCA). This paper discusses the Inventory representation of mixtures of substances that do not react together (i.e., formulated mixtures) as well as those combinations that are formed during certain manufacturing activities and are designated as mixtures by the Agency (i.e., statutory mixtures). Complex reaction products are covered in a separate paper. The Agency's goal in developing this paper is to make it easier for the users of the Inventory to interpret Inventory listings and to understand how new mixtures would be identified for Inventory inclusion. Fundamental to the Inventory as a whole is the principle that entries on the Inventory are identified as precisely as possible for the commercial chemical substance, as reported by the submitter. Substances that are chemically indistinguishable, or even identical, may be listed differently on the Inventory, depending on the degree of knowledge that the submitters possess and report about such substances, as well as how submitters intend to represent the chemical identities to the Agency and to customers. Although these chemically indistinguishable substances are named differently on the Inventory, this is not a "nomenclature" issue, but an issue of substance representation. Submitters should be aware that their choice for substance representation plays an important role in the Agency's determination of how the substance will be listed on the Inventory. -

Introduction to Chemistry
Introduction to Chemistry Author: Tracy Poulsen Digital Proofer Supported by CK-12 Foundation CK-12 Foundation is a non-profit organization with a mission to reduce the cost of textbook Introduction to Chem... materials for the K-12 market both in the U.S. and worldwide. Using an open-content, web-based Authored by Tracy Poulsen collaborative model termed the “FlexBook,” CK-12 intends to pioneer the generation and 8.5" x 11.0" (21.59 x 27.94 cm) distribution of high-quality educational content that will serve both as core text as well as provide Black & White on White paper an adaptive environment for learning. 250 pages ISBN-13: 9781478298601 Copyright © 2010, CK-12 Foundation, www.ck12.org ISBN-10: 147829860X Except as otherwise noted, all CK-12 Content (including CK-12 Curriculum Material) is made Please carefully review your Digital Proof download for formatting, available to Users in accordance with the Creative Commons Attribution/Non-Commercial/Share grammar, and design issues that may need to be corrected. Alike 3.0 Unported (CC-by-NC-SA) License (http://creativecommons.org/licenses/by-nc- sa/3.0/), as amended and updated by Creative Commons from time to time (the “CC License”), We recommend that you review your book three times, with each time focusing on a different aspect. which is incorporated herein by this reference. Specific details can be found at http://about.ck12.org/terms. Check the format, including headers, footers, page 1 numbers, spacing, table of contents, and index. 2 Review any images or graphics and captions if applicable. -

Outline of Physical Science
Outline of physical science “Physical Science” redirects here. It is not to be confused • Astronomy – study of celestial objects (such as stars, with Physics. galaxies, planets, moons, asteroids, comets and neb- ulae), the physics, chemistry, and evolution of such Physical science is a branch of natural science that stud- objects, and phenomena that originate outside the atmosphere of Earth, including supernovae explo- ies non-living systems, in contrast to life science. It in turn has many branches, each referred to as a “physical sions, gamma ray bursts, and cosmic microwave background radiation. science”, together called the “physical sciences”. How- ever, the term “physical” creates an unintended, some- • Branches of astronomy what arbitrary distinction, since many branches of physi- cal science also study biological phenomena and branches • Chemistry – studies the composition, structure, of chemistry such as organic chemistry. properties and change of matter.[8][9] In this realm, chemistry deals with such topics as the properties of individual atoms, the manner in which atoms form 1 What is physical science? chemical bonds in the formation of compounds, the interactions of substances through intermolecular forces to give matter its general properties, and the Physical science can be described as all of the following: interactions between substances through chemical reactions to form different substances. • A branch of science (a systematic enterprise that builds and organizes knowledge in the form of • Branches of chemistry testable explanations and predictions about the • universe).[1][2][3] Earth science – all-embracing term referring to the fields of science dealing with planet Earth. Earth • A branch of natural science – natural science science is the study of how the natural environ- is a major branch of science that tries to ex- ment (ecosphere or Earth system) works and how it plain and predict nature’s phenomena, based evolved to its current state. -

H2CS) and Its Thiohydroxycarbene Isomer (HCSH
A chemical dynamics study on the gas phase formation of thioformaldehyde (H2CS) and its thiohydroxycarbene isomer (HCSH) Srinivas Doddipatlaa, Chao Hea, Ralf I. Kaisera,1, Yuheng Luoa, Rui Suna,1, Galiya R. Galimovab, Alexander M. Mebelb,1, and Tom J. Millarc,1 aDepartment of Chemistry, University of Hawai’iatManoa, Honolulu, HI 96822; bDepartment of Chemistry and Biochemistry, Florida International University, Miami, FL 33199; and cSchool of Mathematics and Physics, Queen’s University Belfast, Belfast BT7 1NN, Northern Ireland, United Kingdom Edited by Stephen J. Benkovic, The Pennsylvania State University, University Park, PA, and approved August 4, 2020 (received for review March 13, 2020) Complex organosulfur molecules are ubiquitous in interstellar molecular sulfur dioxide (SO2) (21) and sulfur (S8) (22). The second phase clouds, but their fundamental formation mechanisms have remained commences with the formation of the central protostars. Tempera- largely elusive. These processes are of critical importance in initiating a tures increase up to 300 K, and sublimation of the (sulfur-bearing) series of elementary chemical reactions, leading eventually to organo- molecules from the grains takes over (20). The subsequent gas-phase sulfur molecules—among them potential precursors to iron-sulfide chemistry exploits complex reaction networks of ion–molecule and grains and to astrobiologically important molecules, such as the amino neutral–neutral reactions (17) with models postulating that the very acid cysteine. Here, we reveal through laboratory experiments, first sulfur–carbon bonds are formed via reactions involving methyl electronic-structure theory, quasi-classical trajectory studies, and astro- radicals (CH3)andcarbene(CH2) with atomic sulfur (S) leading to chemical modeling that the organosulfur chemistry can be initiated in carbonyl monosulfide and thioformaldehyde, respectively (18).