Selective Androgen Receptor Modulators: the Future of Androgen Therapy?
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The 2021 List of Pharmacological Classes of Doping Agents and Doping Methods
BGBl. III - Ausgegeben am 8. Jänner 2021 - Nr. 1 1 von 23 The 2021 list of pharmacological classes of doping agents and doping methods www.ris.bka.gv.at BGBl. III - Ausgegeben am 8. Jänner 2021 - Nr. 1 2 von 23 www.ris.bka.gv.at BGBl. III - Ausgegeben am 8. Jänner 2021 - Nr. 1 3 von 23 THE 2021 PROHIBITED LIST WORLD ANTI-DOPING CODE DATE OF ENTRY INTO FORCE 1 January 2021 Introduction The Prohibited List is a mandatory International Standard as part of the World Anti-Doping Program. The List is updated annually following an extensive consultation process facilitated by WADA. The effective date of the List is 1 January 2021. The official text of the Prohibited List shall be maintained by WADA and shall be published in English and French. In the event of any conflict between the English and French versions, the English version shall prevail. Below are some terms used in this List of Prohibited Substances and Prohibited Methods. Prohibited In-Competition Subject to a different period having been approved by WADA for a given sport, the In- Competition period shall in principle be the period commencing just before midnight (at 11:59 p.m.) on the day before a Competition in which the Athlete is scheduled to participate until the end of the Competition and the Sample collection process. Prohibited at all times This means that the substance or method is prohibited In- and Out-of-Competition as defined in the Code. Specified and non-Specified As per Article 4.2.2 of the World Anti-Doping Code, “for purposes of the application of Article 10, all Prohibited Substances shall be Specified Substances except as identified on the Prohibited List. -

Sarms in Dietary Supplements and Other Products
SARMs in Dietary Supplements and Other Products Selective androgen receptor modulators, or SARMs, are syn- damage.” They’re also prohibited for use in sport. For exam- thetic drugs designed to mimic the effects of testosterone. Al- ple, a 2017 FDA Safety Alert describes andarine and ostarine though these drugs are still in the research and testing stages as “unapproved drugs and anabolic steroid-like substances.” of development, and they are classified as drugs, yet most are In addition, some experimental drugs that are not SARMs are readily available online both as ingredients in dietary supple- being marketed as or along with SARMs, but they are equally ment products and as chemicals available for “research only.” illegal in dietary supplements and prohibited for sport. As a Some websites marketing SARMs for “research use” include result, the most common of these are also included on the next descriptions with language clearly targeting use for bodybuild- page. In addition, there are dietary supplements marketed as ing. However, according to FDA “Body-building products that contain selective androgen receptor modulators, or SARMs, SARMs that do not contain any of the drugs listed here. The have not been approved by the FDA and are associated with following list will help you recognize these drugs, which often serious safety concerns, including potential to increase the risk go by various names, as well as currently available products of heart attack or stroke and life threatening reactions like liver that contain them. SARMs • LGD-3033 -

UFC PROHIBITED LIST Effective June 1, 2021 the UFC PROHIBITED LIST
UFC PROHIBITED LIST Effective June 1, 2021 THE UFC PROHIBITED LIST UFC PROHIBITED LIST Effective June 1, 2021 PART 1. Except as provided otherwise in PART 2 below, the UFC Prohibited List shall incorporate the most current Prohibited List published by WADA, as well as any WADA Technical Documents establishing decision limits or reporting levels, and, unless otherwise modified by the UFC Prohibited List or the UFC Anti-Doping Policy, Prohibited Substances, Prohibited Methods, Specified or Non-Specified Substances and Specified or Non-Specified Methods shall be as identified as such on the WADA Prohibited List or WADA Technical Documents. PART 2. Notwithstanding the WADA Prohibited List and any otherwise applicable WADA Technical Documents, the following modifications shall be in full force and effect: 1. Decision Concentration Levels. Adverse Analytical Findings reported at a concentration below the following Decision Concentration Levels shall be managed by USADA as Atypical Findings. • Cannabinoids: natural or synthetic delta-9-tetrahydrocannabinol (THC) or Cannabimimetics (e.g., “Spice,” JWH-018, JWH-073, HU-210): any level • Clomiphene: 0.1 ng/mL1 • Dehydrochloromethyltestosterone (DHCMT) long-term metabolite (M3): 0.1 ng/mL • Selective Androgen Receptor Modulators (SARMs): 0.1 ng/mL2 • GW-1516 (GW-501516) metabolites: 0.1 ng/mL • Epitrenbolone (Trenbolone metabolite): 0.2 ng/mL 2. SARMs/GW-1516: Adverse Analytical Findings reported at a concentration at or above the applicable Decision Concentration Level but under 1 ng/mL shall be managed by USADA as Specified Substances. 3. Higenamine: Higenamine shall be a Prohibited Substance under the UFC Anti-Doping Policy only In-Competition (and not Out-of- Competition). -

Enhanced UHPLC-MS/MS Screening of Selective Androgen Receptor Modulators Following Urine Hydrolysis
MethodsX 7 (2020) 100926 Contents lists available at ScienceDirect MethodsX j o u r n a l h o m e p a g e: w w w . e l s e v i e r . c o m / l o c a t e / m e x Method Article Enhanced UHPLC-MS/MS screening of selective androgen receptor modulators following urine hydrolysis ∗ ∗ Anna Gadaj a,b, , Emiliano Ventura a, , Jim Healy c,d, Francesco Botrè e, Saskia S. Sterk f, Tom Buckley g, Mark H. Mooney a a Institute for Global Food Security, School of Biological Sciences, Queen’s University Belfast, BT9 5DL, United Kingdom b Chemical and Immunodiagnostic Sciences Branch, Veterinary Sciences Division, Agri-Food & Biosciences Institute (AFBI), Stoney Road, Belfast BT4 3SD, United Kingdom c Laboratory, Irish Greyhound Board, Limerick Greyhound Stadium, Ireland d Applied Science Department, Limerick Institute of Technology, Moylish, Limerick, Ireland e Laboratorio Antidoping, Federazione Medico Sportiva Italiana, Italy f Wageningen Food Safety Research, Wageningen University & Research, European Union Reference Laboratory, Wageningen, the Netherlands g Irish Diagnostic Laboratory Services Ltd., Johnstown, W91 RH93, Co. Kildare, Ireland a b s t r a c t Selective androgen receptor modulators (SARMs) represent non-steroidal agents commonly abused in human and animal (i.e. equine, canine) sports, with potential for further misuse as growth promoting agents in livestock- based farming. As a direct response to the real and possible implications of illicit application in both sport as well as food production systems, this study incorporated enzymatic hydrolysis ( β-glucuronidase/arylsulfatase) into a previously established protocol while maintaining the minimal volume (200 μL) of urine sample required to detect SARMs encompassing various pharmacophores in urine from a range of species (i.e. -

RAD140 | Medchemexpress
Inhibitors Product Data Sheet RAD140 • Agonists Cat. No.: HY-14383 CAS No.: 1182367-47-0 Molecular Formula: C₂₀H₁₆ClN₅O₂ • Molecular Weight: 393.83 Screening Libraries Target: Androgen Receptor Pathway: Others Storage: Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month SOLVENT & SOLUBILITY In Vitro DMSO : ≥ 100 mg/mL (253.92 mM) * "≥" means soluble, but saturation unknown. Mass Solvent 1 mg 5 mg 10 mg Concentration Preparing 1 mM 2.5392 mL 12.6958 mL 25.3917 mL Stock Solutions 5 mM 0.5078 mL 2.5392 mL 5.0783 mL 10 mM 0.2539 mL 1.2696 mL 2.5392 mL Please refer to the solubility information to select the appropriate solvent. In Vivo 1. Add each solvent one by one: 10% DMSO >> 40% PEG300 >> 5% Tween-80 >> 45% saline Solubility: ≥ 2.5 mg/mL (6.35 mM); Clear solution 2. Add each solvent one by one: 10% DMSO >> 90% (20% SBE-β-CD in saline) Solubility: 2.5 mg/mL (6.35 mM); Suspended solution; Need ultrasonic 3. Add each solvent one by one: 10% DMSO >> 90% corn oil Solubility: ≥ 2.5 mg/mL (6.35 mM); Clear solution BIOLOGICAL ACTIVITY Description RAD140 is a potent, orally active, nonsteroidal selective androgen receptor modulator (SARM) with a Ki of 7 nM. RAD140 shows good selectivity over other steroid hormone nuclear receptors[1]. IC₅₀ & Target Ki: 7 nM (Androgen receptor)[1] In Vitro RAD140 (0-300 nM; pretreated for 1 hour) increases neuron viability against Aβ in a concentration-dependent manner[2]. -

Banned Substances in Sports Supplements
Banned substances in sports supplements Consumer Information Sheet The Health Protection Service (HPS) is aware that sports supplements labelled as containing banned substances are being sold in the ACT primarily through retail supplement stores. Banned substances include selective androgen receptor modulators (SARMs), stenabolic, ibutamoren, cardarine, tadalafil, oxedrine, melatonin, phenibut, clomifene, l-dopa and DHEA. Most of these substances are associated with significant health and safety concerns and their long-term effects on the human body are unknown. Some substances are prescription medicines that without appropriate medical management and monitoring, pose significant risks to the individuals who take them. Some substances are prohibited substances that are associated with high risk of harm to individuals who take them and have no therapeutic purpose. This information sheet provides further details on these substances. If you are taking a supplement which is labelled as containing a banned substance and that has not been prescribed by your doctor, you should stop taking it immediately and report the name of the product and place of purchase to the Pharmaceutical Services Section of the HPS on (02) 6205 0998. If you have concerns about your health, please see your General Practitioner. Selective Androgen Receptor Modulators (SARMs) SARMs are a group of compounds which act in a similar way to anabolic steroids and cause tissue (bone and muscle) growth. Unlike anabolic steroids, SARMs are less likely to cause unfavourable side effects such as the development of male gender characteristics in females, and the development of baldness, breast tissue and testicular shrinkage in males. SARMs are associated with serious safety concerns including liver toxicity and the potential to increase the risk of heart attack and stroke.1 The long-term effects of SARMs on the human body are unknown. -

Anti-Doping Manual Revised Dec. 2018
PGA TOUR Anti-Doping Program Manual REVISED DECEMBER 2018 Table of Contents How To Use This Manual .......................................................................................................................................................2 SECTION 1: Player Guide To Anti-Doping ...........................................................................................................................3 Who is covered by the Anti-Doping Program .........................................................................................................3 What substances and methods are banned ..........................................................................................................3 Am I liable for a prohibited substance in my body even if I did not intend to take the substance ........................3 What should players know about nutritional and health products ...................................................................3 Are there supplements that have been tested/certified as free from banned substances ........................3 What about medical treatment.................................................................................................................................4 What medications are permitted .............................................................................................................................4 Who conducts the testing and who will be tested ................................................................................................4 What are the steps in -

2019 Prohibited List
THE WORLD ANTI-DOPING CODE INTERNATIONAL STANDARD PROHIBITED LIST JANUARY 2019 The official text of the Prohibited List shall be maintained by WADA and shall be published in English and French. In the event of any conflict between the English and French versions, the English version shall prevail. This List shall come into effect on 1 January 2019 SUBSTANCES & METHODS PROHIBITED AT ALL TIMES (IN- AND OUT-OF-COMPETITION) IN ACCORDANCE WITH ARTICLE 4.2.2 OF THE WORLD ANTI-DOPING CODE, ALL PROHIBITED SUBSTANCES SHALL BE CONSIDERED AS “SPECIFIED SUBSTANCES” EXCEPT SUBSTANCES IN CLASSES S1, S2, S4.4, S4.5, S6.A, AND PROHIBITED METHODS M1, M2 AND M3. PROHIBITED SUBSTANCES NON-APPROVED SUBSTANCES Mestanolone; S0 Mesterolone; Any pharmacological substance which is not Metandienone (17β-hydroxy-17α-methylandrosta-1,4-dien- addressed by any of the subsequent sections of the 3-one); List and with no current approval by any governmental Metenolone; regulatory health authority for human therapeutic use Methandriol; (e.g. drugs under pre-clinical or clinical development Methasterone (17β-hydroxy-2α,17α-dimethyl-5α- or discontinued, designer drugs, substances approved androstan-3-one); only for veterinary use) is prohibited at all times. Methyldienolone (17β-hydroxy-17α-methylestra-4,9-dien- 3-one); ANABOLIC AGENTS Methyl-1-testosterone (17β-hydroxy-17α-methyl-5α- S1 androst-1-en-3-one); Anabolic agents are prohibited. Methylnortestosterone (17β-hydroxy-17α-methylestr-4-en- 3-one); 1. ANABOLIC ANDROGENIC STEROIDS (AAS) Methyltestosterone; a. Exogenous* -

FDHT-PET to Predict Bicalutamide Efficacy in Patients with Androgen
University of Groningen Serial [F-18]-FDHT-PET to predict bicalutamide efficacy in patients with androgen receptor positive metastatic breast cancer Boers, Jorianne; Venema, Clasina M; de Vries, Erik F J; Hospers, Geke A P; Boersma, Hendrikus H; Rikhof, Bart; Dorbritz, Christine; Glaudemans, Andor W J M; Schröder, Carolina P Published in: European Journal of Cancer DOI: 10.1016/j.ejca.2020.11.008 IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document version below. Document Version Publisher's PDF, also known as Version of record Publication date: 2021 Link to publication in University of Groningen/UMCG research database Citation for published version (APA): Boers, J., Venema, C. M., de Vries, E. F. J., Hospers, G. A. P., Boersma, H. H., Rikhof, B., Dorbritz, C., Glaudemans, A. W. J. M., & Schröder, C. P. (2021). Serial [F-18]-FDHT-PET to predict bicalutamide efficacy in patients with androgen receptor positive metastatic breast cancer. European Journal of Cancer, 144, 151-161. https://doi.org/10.1016/j.ejca.2020.11.008 Copyright Other than for strictly personal use, it is not permitted to download or to forward/distribute the text or part of it without the consent of the author(s) and/or copyright holder(s), unless the work is under an open content license (like Creative Commons). Take-down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. -

Targeting the Androgen Receptor in Breast Cancer
Targeting the Androgen Receptor in Breast Cancer ______________________________________________________________________ Kee Ming Chia A thesis in fulfilment of the requirements for the degree of Doctor of Philosophy Garvan Institute of Medical Research St. Vincent’s Hospital Clinical School Faculty of Medicine December 2018 iii Thesis/Dissertation Sheet Surname/Family Name : CHIA Given Name/s : KEE MING Abbreviation for degree as given in the : PhD University calendar Faculty : Medicine School : St Vincent’s Hospital Clinical School Thesis Title : Targeting AR in breast cancer Abstract Estrogen receptor positive (ER+) breast cancer constitutes 70% of all breast cancers and anti-ER therapies such as aromatase inhibitors and tamoxifen represent the main therapeutic strategies in the treatment of this disease. Unfortunately, up to 30% of all primary ER+ tumours will ultimately develop endocrine-resistance and progress on ER-targeted therapies resulting in disease-related morbidity. As a result, there is an urgent medical need for novel therapeutic strategies capable of managing endocrine-resistant breast cancer. Androgen receptor (AR) is expressed in up to 90% of ER+ breast cancers. AR functions as a tumour suppressor in primary ER+ breast cancer and high AR positivity is strongly associated with a favourable patient outcome in the ER+ setting. However, the role of AR in endocrine-resistant breast tumours is highly controversial with data supporting both oncogenic and tumour suppressive functions reported in the literature. Here I have used different modulatory approaches on in vitro and in vivo preclinical models to dissect the functions of AR and determine the best approach to target AR in endocrine-resistant breast cancer. I use an siRNA-mediated approach to knock down AR in cell line models and discover that the basal expression of AR contributes to endocrine-resistance and that loss of AR restores classical ER signalling and reverses endocrine-resistance. -

Development and Validation of a Semi-Quantitative Ultra-High
G Model CHROMA-360196; No. of Pages 14 ARTICLE IN PRESS Journal of Chromatography A, xxx (2019) xxx–xxx Contents lists available at ScienceDirect Journal of Chromatography A j ournal homepage: www.elsevier.com/locate/chroma Development and validation of a semi-quantitative ultra-high performance liquid chromatography-tandem mass spectrometry method for screening of selective androgen receptor modulators in urine a,∗ a,∗ a a b,c Emiliano Ventura , Anna Gadaj , Gail Monteith , Alexis Ripoche , Jim Healy , d e f a Francesco Botrè , Saskia S. Sterk , Tom Buckley , Mark H. Mooney a Institute for Global Food Security, School of Biological Sciences, Queen’s University Belfast, BT9 5AG, United Kingdom b Laboratory, Irish Greyhound Board, Limerick Greyhound Stadium, Ireland c Applied Science Department, Limerick Institute of Technology, Moylish, Limerick, Ireland d Laboratorio Antidoping, Federazione Medico Sportiva Italiana, Italy e RIKILT Wageningen University & Research, European Union Reference Laboratory, Wageningen, the Netherlands f Irish Diagnostic Laboratory Services Ltd., Johnstown, Co. Kildare, Ireland a r t i c l e i n f o a b s t r a c t Article history: A semi-quantitative method was developed to monitor the misuse of 15 SARM compounds belonging Received 25 January 2019 to nine different families, in urine matrices from a range of species (equine, canine, human, bovine and Received in revised form 16 April 2019 murine). SARM residues were extracted from urine (200 L) with tert-butyl methyl ether (TBME) without Accepted 17 April 2019 further clean-up and analysed by ultra-high performance liquid chromatography coupled to tandem mass Available online xxx spectrometry (UHPLC-MS/MS). -
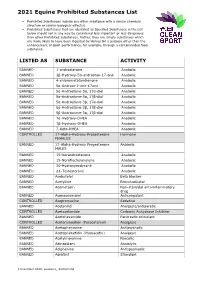
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s).