Fritz Haber, the Damned Scientist
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Goverdhan Mehta Chemistry - a 21St Century Science for Global Sustainability: Is It Future Ready?
Goverdhan Mehta Chemistry - A 21st Century Science for Global Sustainability: Is it future ready? Goverdhan Mehta A ‘selfie’ with theSchool chemical of Chemistry world…… University of Hyderabad National Geophysical Research Institute, CSIR Foundation Day, Sept. 27, 2019 Introducing Chemistry through the Lens of Earth's Systems: What Role Can Systems Thinking Play in Developing Chemically and Environmentally Literate Citizens? J. Kornfeld, S. Stokoe. J. Chemical Education 2019, 96, 2910-2917 A bouquet of ‘matters’ that matter New symbols Passion Sustainability Legacies Ethics & values Responsible Connections Systems Directions Humility Ideas & icons Inspirations Un mélange de beaucoup de choses “Chemistry ought not to be for chemists alone” - Miguel de Unamuno ‘…Life, Universe and Everything’ Chemistry – a source of happiness…. Chem -Connectome ‘...I feel sorry for people who don’t know anything about chemistry. They are missing an important source of happiness....’ - Linus Pauling 1901-1994 S.A. Matlin, G. Mehta, H. Hopf. Chemistry Embraced by All. Science 2015, 347, 1179 Chemistry is in everything. and everything is in it, it is the basis of life, without it we wouldn't exist. Green tea has ~ 200 chemicals Coffee has ~ 1000 chemicals Wine has >1000 chemicals Light cigarette ~ 4000 chemicals Chemistry is ubiquitous/omnipresent Chemistry – Tracing the roots and to the present BCE Art & craft of mixing substances A giant knowledge leap Alchemy to modern science Evidence based science Discipline in a Table - systematization Mendeleev’s Periodic Law ‘Molecularization’ of chemical matter 20th Century A century of evolutionary march of chemistry 20h Century Value added products from almost anything “Utility science” and everything Molecular understanding of life processes and “Core Science” chemical matter Interdisciplinarity in forefront “Integrative Science” Resource stressed planet “Sustainability Science” 21st Century S. -

George De Hevesy in America
Journal of Nuclear Medicine, published on July 13, 2019 as doi:10.2967/jnumed.119.233254 George de Hevesy in America George de Hevesy was a Hungarian radiochemist who was awarded the Nobel Prize in Chemistry in 1943 for the discovery of the radiotracer principle (1). As the radiotracer principle is the foundation of all diagnostic and therapeutic nuclear medicine procedures, Hevesy is widely considered the father of nuclear medicine (1). Although it is well-known that he spent time at a number of European institutions, it is not widely known that he also spent six weeks at Cornell University in Ithaca, NY, in the fall of 1930 as that year’s Baker Lecturer in the Department of Chemistry (2-6). “[T]he Baker Lecturer gave two formal presentations per week, to a large and diverse audience and provided an informal seminar weekly for students and faculty members interested in the subject. The lecturer had an office in Baker Laboratory and was available to faculty and students for further discussion.” (7) There is also evidence that, “…Hevesy visited Harvard [University, Cambridge, MA] as a Baker Lecturer at Cornell in 1930…” (8). Neither of the authors of this Note/Letter was aware of Hevesy’s association with Cornell University despite our longstanding ties to Cornell until one of us (WCK) noticed the association in Hevesy’s biographical page on the official Nobel website (6). WCK obtained both his undergraduate degree and medical degree from Cornell in Ithaca and New York City, respectively, and spent his career in nuclear medicine. JRO did his nuclear medicine training at Columbia University and has subsequently been a faculty member of Weill Cornell Medical College for the last eleven years (with a brief tenure at Memorial Sloan Kettering Cancer Center (affiliated with Cornell)), and is now the program director of the Nuclear Medicine residency and Chief of the Molecular Imaging and Therapeutics Section. -

The Place of Zinc, Cadmium, and Mercury in the Periodic Table
Information • Textbooks • Media • Resources The Place of Zinc, Cadmium, and Mercury in the Periodic Table William B. Jensen Department of Chemistry, University of Cincinnati, Cincinnati, OH 45221-0172; [email protected] One of the few facts that I can remember from my un- a quarter of the more recent introductory inorganic texts. In dergraduate inorganic course was my instructor’s insistence all cases, the Zn group was incorrectly labeled as being a mem- that zinc, cadmium, and mercury should be classified as main- ber of the d block or transition block. Those introductory block elements rather than as transition-block or d-block el- inorganic texts that presented some sort of systematic survey ements. Though I have always assumed that the evidence for of descriptive chemistry usually contradicted this assignment this statement was unambiguous, I have also noticed the ap- in their later discussions of the chemistry of these elements. pearance over the last decade of an increasing number of gen- On the other hand, the surveys of descriptive chemistry found eral chemistry texts, inorganic texts, and advanced inorganic in most of the general chemistry texts were so superficial that monographs that either explicitly or implicitly contradict this the existence of this inconsistency seldom became explicit. assignment. The inorganic textbook by Cotton and In light of these trends, I thought it might be of interest Wilkinson, which has served as the American standard for to summarize the evidence relating to the proper placement nearly 40 years, has always been firm in its treatment of the of the Zn group within the periodic table. -

Europe's Favorite Chemists?
ability and on uncertainty evaluation of chemical mea- surement results. Participants can take this expertise back to their home countries and/or become regional coordinators for IMEP in their respective countries. In collaboration with EUROMET and CCQM, IMEP samples are offered for the organization of EUROMET key or supplementary comparisons and, where appro- priate, for the organization of CCQM key comparisons or pilot studies. In its role as a neutral, impartial international evalu- ation program, IMEP displays existing problems in chemical measurement. IRMM is dedicated to tackling this problem and will, where possible, collaborate with international bodies, education and accreditation au- thorities, and NMIs to achieve more reliable measure- ments and contribute to setting up an internationally structured measurement system. Ellen Poulsen (Denmark) preparing graphs for the IMEP-9 participants’ report on trace elements in water. Europe’s Favorite Chemists? Choosing Europe’s Top 100 Chemists: birth of Christ is quite forgotten in general. An addi- A Difficult Task tional irony lies in the fact that recent evidence from history, archaeology, and astronomy suggests a birthdate This article by Prof. Colin Russell (Department of His- about seven years earlier, so the real millennium came tory of Science and Technology, Open University, and went unnoticed in the early 1990s. However that Milton Keynes, England MK6 7AA, UK) was com- may be, the grand spirit of revelry and bonhomie can- missioned by Chemistry in Britain and published by not be quenched by such mundane considerations, and that magazine in Vol. 36, pp. 50–52, February 2000. celebration there shall be. Nor are societies to be left The list of Europe’s 100 distinguished chemists was behind in the general euphoria. -

Stainless Steels in Ammonia Production
Stainless Steels in Ammonia Production Committee of Stainless Steel Producers American Iron and Steel Institute Washington, D.C. CONTENTS INTRODUCTION ...................... 4 PROCESS DESCRIPTION ....... 5 CORROSIVES IN AMMONIA PROCESSES .......... 5 CONSIDERATIONS FOR SELECTING STAINLESS STEELS .................................... 6 Desulfurization of Natural Gas ................. 6 Catalytic Steam Reforming of Natural Gas ................. 6 Carbon Monoxide Shift ......... 8 Removal of Carbon Dioxide . 10 Methanation .......................... 11 Synthesis of Ammonia ......... 11 Turbine-Driven Centrifugal Compression Trains ........ 14 STAINLESS STEELS ................ 15 Metallurgical Structure ......... 16 High-Temperature Mechanical Properties ..... 16 Thermal Conductivity ........... 20 Oxidation Resistance ........... 21 Sulfidation Resistance .......... 22 REFERENCES .......................... 22 The material presented in this booklet has been prepared for the general information of the reader. It should not be used without first securing competent advice with respect to its suitability for any given application. While the material is believed to be technically correct, neither the Committee of Stainless Steel Pro- ducers nor the companies represented on the The Committee of Stainless Steel Producers acknowledges the help Committee warrant its suitability for any gen- by Gregory Kobrin, Senior Consultant, Materials Engineering, E.I. eral or particular use. DuPont de Nemours & Company in assembling data for this booklet. single-train plants came increasing demands on materials of construc- INTRODUCTION tion. The process for making am- monia is considered to be only Approximately 12 elements are es- moderately corrosive, so considera- sential to plant growth. Of these, nit- ble use is made of carbon and low- rogen is the main nutrient and is re- alloy steels for vessels and piping. quired in much larger amounts than However, numerous applications any other element. -

Einstein and Physics Hundred Years Ago∗
Vol. 37 (2006) ACTA PHYSICA POLONICA B No 1 EINSTEIN AND PHYSICS HUNDRED YEARS AGO∗ Andrzej K. Wróblewski Physics Department, Warsaw University Hoża 69, 00-681 Warszawa, Poland [email protected] (Received November 15, 2005) In 1905 Albert Einstein published four papers which revolutionized physics. Einstein’s ideas concerning energy quanta and electrodynamics of moving bodies were received with scepticism which only very slowly went away in spite of their solid experimental confirmation. PACS numbers: 01.65.+g 1. Physics around 1900 At the turn of the XX century most scientists regarded physics as an almost completed science which was able to explain all known physical phe- nomena. It appeared to be a magnificent structure supported by the three mighty pillars: Newton’s mechanics, Maxwell’s electrodynamics, and ther- modynamics. For the celebrated French chemist Marcellin Berthelot there were no major unsolved problems left in science and the world was without mystery. Le monde est aujourd’hui sans mystère— he confidently wrote in 1885 [1]. Albert A. Michelson was of the opinion that “The more important fundamen- tal laws and facts of physical science have all been discovered, and these are now so firmly established that the possibility of their ever being supplanted in consequence of new discoveries is exceedingly remote . Our future dis- coveries must be looked for in the sixth place of decimals” [2]. Physics was not only effective but also perfect and beautiful. Henri Poincaré maintained that “The theory of light based on the works of Fresnel and his successors is the most perfect of all the theories of physics” [3]. -

Otto Sackur's Pioneering Exploits in the Quantum Theory Of
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Catalogo dei prodotti della ricerca Chapter 3 Putting the Quantum to Work: Otto Sackur’s Pioneering Exploits in the Quantum Theory of Gases Massimiliano Badino and Bretislav Friedrich After its appearance in the context of radiation theory, the quantum hypothesis rapidly diffused into other fields. By 1910, the crisis of classical traditions of physics and chemistry—while taking the quantum into account—became increas- ingly evident. The First Solvay Conference in 1911 pushed quantum theory to the fore, and many leading physicists responded by embracing the quantum hypoth- esis as a way to solve outstanding problems in the theory of matter. Until about 1910, quantum physics had drawn much of its inspiration from two sources. The first was the complex formal machinery connected with Max Planck’s theory of radiation and, above all, its close relationship with probabilis- tic arguments and statistical mechanics. The fledgling 1900–1901 version of this theory hinged on the application of Ludwig Boltzmann’s 1877 combinatorial pro- cedure to determine the state of maximum probability for a set of oscillators. In his 1906 book on heat radiation, Planck made the connection with gas theory even tighter. To illustrate the use of the procedure Boltzmann originally developed for an ideal gas, Planck showed how to extend the analysis of the phase space, com- monplace among practitioners of statistical mechanics, to electromagnetic oscil- lators (Planck 1906, 140–148). In doing so, Planck identified a crucial difference between the phase space of the gas molecules and that of oscillators used in quan- tum theory. -

EINSTEIN and NAZI PHYSICS When Science Meets Ideology and Prejudice
MONOGRAPH Mètode Science Studies Journal, 10 (2020): 147-155. University of Valencia. DOI: 10.7203/metode.10.13472 ISSN: 2174-3487. eISSN: 2174-9221. Submitted: 29/11/2018. Approved: 23/05/2019. EINSTEIN AND NAZI PHYSICS When science meets ideology and prejudice PHILIP BALL In the 1920s and 30s, in a Germany with widespread and growing anti-Semitism, and later with the rise of Nazism, Albert Einstein’s physics faced hostility and was attacked on racial grounds. That assault was orchestrated by two Nobel laureates in physics, who asserted that stereotypical racial features are exhibited in scientific thinking. Their actions show how ideology can infect and inflect science. Reviewing this episode in the current context remains an instructive and cautionary tale. Keywords: Albert Einstein, Nazism, anti-Semitism, science and ideology. It was German society, Einstein said, that revealed from epidemiology and research into disease (the to him his Jewishness. «This discovery was brought connection of smoking to cancer, and of HIV to home to me by non-Jews rather than Jews», he wrote AIDS) to climate change, this idea perhaps should in 1929 (cited in Folsing, 1998, p. 488). come as no surprise. But it is for that very reason that Shortly after the boycott of Jewish businesses at the the hostility Einstein’s physics sometimes encountered start of April 1933, the German Students Association, in Germany in the 1920s and 30s remains an emboldened by Hitler’s rise to total power, declared instructive and cautionary tale. that literature should be cleansed of the «un-German spirit». The result, on 10 May, was a ritualistic ■ AGAINST RELATIVITY burning of tens of thousands of books «marred» by Jewish intellectualism. -

2020 Stainless Steels in Ammonia Production
STAINLESS STEELS IN AMMONIA PRODUCTION A DESIGNERS’ HANDBOOK SERIES NO 9013 Produced by Distributed by AMERICAN IRON NICKEL AND STEEL INSTITUTE INSTITUTE STAINLESS STEELS IN AMMONIA PRODUCTION A DESIGNERS’ HANDBOOK SERIES NO 9013 Originally, this handbook was published in 1978 by the Committee of Stainless Steel Producers, American Iron and Steel Institute. The Nickel Institute republished the handbook in 2020. Despite the age of this publication the information herein is considered to be generally valid. Material presented in the handbook has been prepared for the general information of the reader and should not be used or relied on for specific applications without first securing competent advice. The Nickel Institute, the American Iron and Steel Institute, their members, staff and consultants do not represent or warrant its suitability for any general or specific use and assume no liability or responsibility of any kind in connection with the information herein. Nickel Institute [email protected] www.nickelinstitute.org CONTENTS INTRODUCTION ............................ 4 PROCESS DESCRIPTION ............ 5 CORROSIVES IN AMMONIA PROCESSES ............... 5 CONSIDERATIONS FOR SELECTING STAINLESS STEELS .......................................... 6 Desulfurization of Natural Gas ....................... 6 Catalytic Steam Reforming of Natural Gas ....................... 6 Carbon Monoxide Shift .............. 8 Removal of Carbon Dioxide . 10 Methanation ............................. 11 Synthesis of Ammonia ............. 11 -

THEODOR Ft5rster 1910
THEODOR Ft5RSTER 1910 - 19714 Journal of Luminescence 12/13 (1976) 8—12 © North-Holland Publishing Company MEMORIAL FOR THE LATE PROFESSOR THEODOR FORSTER A. WELLER Max-Planck-Institut für Biophysikalische Chemie Göttingen, Fed. Rep. Germany On the 20th of May 1974 Theodor Forster died of a heart attack at the age of 64 while he was on his way to the Institute of Physical Chemistry at the University of Stuttgart whose director he had been for 23 years. In view of his numerous important and fundamental contributions to almost all aspects of luminescence, it is indeed appropriate to pay honour to the man and his work today. Theodor Forster was born on May 15, 1910, in Frankfurt, Germany. He went to school there (the same school which about 30 years before Otto Hahn had attended), and after completing his Abitur in 1929, he became a student of physics and mathe- matics at Frankfurt University. At that time there was very little red tape at German Universities. It was for in- stance possible to study physics and/or mathematics without an examination except for the final oral examination after having finished the PhD thesis. As a consequence the good student, in general, was able to finish the University in surprisingly short time; and so in 1933 at the age of 23 Theodor Förster was promoted to Dr. phil. with a thesis entitled “ZurPolarisation von Elektronen durch Reflexion” under the tutelage of E. Madelung. Still before having finished his thesis he had become an Assistant (scientific co- worker) of ProfessorKarl-Friedrich Bonhoeffer. -

Otto Hahn Otto Hahn
R.N. 70269/98 Postal Registration No.: DL-SW-1/4082/15-17 ISSN : 0972-169X Date of posting: 26-27 of advance month Date of publication: 24 of advance month May 2017 Vol. 19 No. 8 Rs. 5.00 Otto Hahn Discoverer of Nuclear Fission Editorial: Consolidating 35 science communication activities in our country Otto Hahn: Discoverer of 34 Nuclear Fission Keep Your Eyes Healthy 31 Phenol: A Serious 30 Environmental Threat Accidental Discoveries in 28 Medical Science Cures for haemorrhoids— 24 Simple treatments and Surgeries Recent developments 21 in science and technology 36 Editorial Consolidating science communication activities in our country Dr. R. Gopichandran It is well known that the National Council of Science Museums of India’s leadership in science technology and innovation (STI) across the Ministry of Culture, Government of India, the National Institute the bilateral and multilateral framework also. The news feature service of Science Communication and Information Resources (NISCAIR) and the portal activity have well defined action plans to reach out to of CSIR, the National Council for Science and Technology fellow institutions and citizens with suitably embellished platform Communication (NCSTC) of the Department of Science and and opportunities for all to deliver together. Technology (DST), Government of India and Vigyan Prasar, also While these are interesting and extremely important, especially of DST, have been carrying out excellent science communication because they respond to the call to upscale and value add science activities over the years. It cannot be denied that the reach has been and technology communication, it is equally important to document quite significant collectively. -
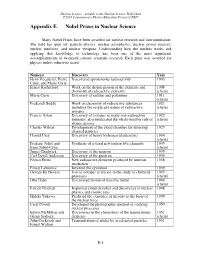
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence