Fishes of Archbold Biological Station
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Atheriniformes : Atherinidae
Atheriniformes: Atherinidae 2111 Atheriniformes: Atherinidae Order ATHERINIFORMES ATHERINIDAE Silversides by L. Tito de Morais, IRD/LEMAR, University of Brest, Plouzané, France; M. Sylla, Centre de Recherches Océanographiques de Dakar-Thiaroye (CRODT), Senegal and W. Ivantsoff (retired), Biology Science, Macquarie University NSW 2109, North Ryde, Australia iagnostic characters: Small, elongate fish, rarely exceeding 15 cm in length. Body elongate and Dsomewhat compressed. Short head, generally flattened dorsally, large eyes, sharp nose, mouth small, oblique and in terminal position, jaws subequal, reaching or slightly exceeding the anterior margin of the eye; premaxilla with ascending process of variable length, with lateral process present or absent; ramus of dentary bone elevated posteriorly or indistinct from anterior part of lower jaw; fine, small and sharp teeth on the jaws, on the roof of mouth (vomer, palatine, pterygoid) or on outside of mouth; 10 to 26 gill rakers long and slender on lower arm of first gill arch. Two well-separated dorsal fins, the first with 6 to 10 thin, flexible spines, located approximately in the middle of the body; the second dorsal and anal fins with a single small weak spine, 1 unbranched soft ray and a variable number of soft rays. Anal fin always originating slightly in advance of second dorsal fin; pectoral fins inserted high on the flanks, directly behind posterior rim of gill cover, with spine greatly reduced and first ray much thicker than those following. Abdomninal pelvic fins with 1 spine and 5 soft rays; forked caudal fin; anus away from the origin of the anal fin. Relatively large scales, cycloid (smooth). -

Phylogeny Classification Additional Readings Clupeomorpha and Ostariophysi
Teleostei - AccessScience from McGraw-Hill Education http://www.accessscience.com/content/teleostei/680400 (http://www.accessscience.com/) Article by: Boschung, Herbert Department of Biological Sciences, University of Alabama, Tuscaloosa, Alabama. Gardiner, Brian Linnean Society of London, Burlington House, Piccadilly, London, United Kingdom. Publication year: 2014 DOI: http://dx.doi.org/10.1036/1097-8542.680400 (http://dx.doi.org/10.1036/1097-8542.680400) Content Morphology Euteleostei Bibliography Phylogeny Classification Additional Readings Clupeomorpha and Ostariophysi The most recent group of actinopterygians (rayfin fishes), first appearing in the Upper Triassic (Fig. 1). About 26,840 species are contained within the Teleostei, accounting for more than half of all living vertebrates and over 96% of all living fishes. Teleosts comprise 517 families, of which 69 are extinct, leaving 448 extant families; of these, about 43% have no fossil record. See also: Actinopterygii (/content/actinopterygii/009100); Osteichthyes (/content/osteichthyes/478500) Fig. 1 Cladogram showing the relationships of the extant teleosts with the other extant actinopterygians. (J. S. Nelson, Fishes of the World, 4th ed., Wiley, New York, 2006) 1 of 9 10/7/2015 1:07 PM Teleostei - AccessScience from McGraw-Hill Education http://www.accessscience.com/content/teleostei/680400 Morphology Much of the evidence for teleost monophyly (evolving from a common ancestral form) and relationships comes from the caudal skeleton and concomitant acquisition of a homocercal tail (upper and lower lobes of the caudal fin are symmetrical). This type of tail primitively results from an ontogenetic fusion of centra (bodies of vertebrae) and the possession of paired bracing bones located bilaterally along the dorsal region of the caudal skeleton, derived ontogenetically from the neural arches (uroneurals) of the ural (tail) centra. -

Age, Growth and Body Condition of Big-Scale Sand Smelt Atherina Boyeri Risso, 1810 Inhabiting a Freshwater Environment: Lake Trasimeno (Italy)
Knowledge and Management of Aquatic Ecosystems (2015) 416, 09 http://www.kmae-journal.org c ONEMA, 2015 DOI: 10.1051/kmae/2015005 Age, growth and body condition of big-scale sand smelt Atherina boyeri Risso, 1810 inhabiting a freshwater environment: Lake Trasimeno (Italy) M. Lorenzoni(1), D. Giannetto(2),,A.Carosi(1), R. Dolciami(3), L. Ghetti(4), L. Pompei(1) Received September 24, 2014 Revised January 29, 2015 Accepted January 29, 2015 ABSTRACT Key-words: The age, growth and body condition of the big-scale sand smelt (Athe- Population rina boyeri) population of Lake Trasimeno were investigated. In total, dynamics, 3998 specimens were collected during the study and five age classes Lee’s (from 0+ to 4+) were identified. From a subsample of 1017 specimens, phenomenon, there were 583 females, 411 males and 23 juveniles. The equations = − fishery between total length (TL) and weight (W) were: log10 W 2.326 + = − management, 3.139 log10 TL for males and log10 W 2.366 + 3.168 log10 TL for fe- introduced males. There were highly significant differences between the sexes and species, for both sexes the value of b (slope of the log (TL-W regression) was Lake Trasimeno greater than 3 (3.139 for males and 3.168 for females), indicating positive allometric growth. The parameters of the theoretical growth curve were: −1 TLt = 10.03 cm; k = 0.18 yr , t0 = −0.443 yr and Φ = 1.65. Monthly trends of overall condition and the gonadosomatic index (GSI) indicated that the reproductive period occurred from March to September. Analy- sis of back-calculated lengths indicated the occurrence of a reverse Lee’s phenomenon. -
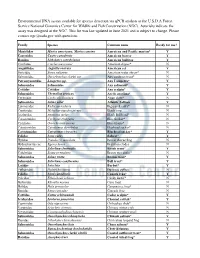
Edna Assay Development
Environmental DNA assays available for species detection via qPCR analysis at the U.S.D.A Forest Service National Genomics Center for Wildlife and Fish Conservation (NGC). Asterisks indicate the assay was designed at the NGC. This list was last updated in June 2021 and is subject to change. Please contact [email protected] with questions. Family Species Common name Ready for use? Mustelidae Martes americana, Martes caurina American and Pacific marten* Y Castoridae Castor canadensis American beaver Y Ranidae Lithobates catesbeianus American bullfrog Y Cinclidae Cinclus mexicanus American dipper* N Anguillidae Anguilla rostrata American eel Y Soricidae Sorex palustris American water shrew* N Salmonidae Oncorhynchus clarkii ssp Any cutthroat trout* N Petromyzontidae Lampetra spp. Any Lampetra* Y Salmonidae Salmonidae Any salmonid* Y Cottidae Cottidae Any sculpin* Y Salmonidae Thymallus arcticus Arctic grayling* Y Cyrenidae Corbicula fluminea Asian clam* N Salmonidae Salmo salar Atlantic Salmon Y Lymnaeidae Radix auricularia Big-eared radix* N Cyprinidae Mylopharyngodon piceus Black carp N Ictaluridae Ameiurus melas Black Bullhead* N Catostomidae Cycleptus elongatus Blue Sucker* N Cichlidae Oreochromis aureus Blue tilapia* N Catostomidae Catostomus discobolus Bluehead sucker* N Catostomidae Catostomus virescens Bluehead sucker* Y Felidae Lynx rufus Bobcat* Y Hylidae Pseudocris maculata Boreal chorus frog N Hydrocharitaceae Egeria densa Brazilian elodea N Salmonidae Salvelinus fontinalis Brook trout* Y Colubridae Boiga irregularis Brown tree snake* -

Fish Inventory at Stones River National Battlefield
Fish Inventory at Stones River National Battlefield Submitted to: Department of the Interior National Park Service Cumberland Piedmont Network By Dennis Mullen Professor of Biology Department of Biology Middle Tennessee State University Murfreesboro, TN 37132 September 2006 Striped Shiner (Luxilus chrysocephalus) – nuptial male From Lytle Creek at Fortress Rosecrans Photograph by D. Mullen Table of Contents List of Tables……………………………………………………………………….iii List of Figures………………………………………………………………………iv List of Appendices…………………………………………………………………..v Executive Summary…………………………………………………………………1 Introduction…………………………………………………………………...……..2 Methods……………………………………………………………………………...3 Results……………………………………………………………………………….7 Discussion………………………………………………………………………….10 Conclusions………………………………………………………………………...14 Literature Cited…………………………………………………………………….15 ii List of Tables Table1: Location and physical characteristics (during September 2006, and only for the riverine sites) of sample sites for the STRI fish inventory………………………………17 Table 2: Biotic Integrity classes used in assessing fish communities along with general descriptions of their attributes (Karr et al. 1986) ………………………………………18 Table 3: List of fishes potentially occurring in aquatic habitats in and around Stones River National Battlefield………………………………………………………………..19 Table 4: Fish species list (by site) of aquatic habitats at STRI (October 2004 – August 2006). MF = McFadden’s Ford, KP = King Pond, RB = Redoubt Brannan, UP = Unnamed Pond at Redoubt Brannan, LC = Lytle Creek at Fortress Rosecrans……...….22 Table 5: Fish Species Richness estimates for the 3 riverine reaches of STRI and a composite estimate for STRI as a whole…………………………………………………24 Table 6: Index of Biotic Integrity (IBI) scores for three stream reaches at Stones River National Battlefield during August 2005………………………………………………...25 Table 7: Temperature and water chemistry of four of the STRI sample sites for each sampling date…………………………………………………………………………….26 Table 8 : Total length estimates of specific habitat types at each riverine sample site. -

Acanthopterygii, Bone, Eurypterygii, Osteology, Percomprpha
Research in Zoology 2014, 4(2): 29-42 DOI: 10.5923/j.zoology.20140402.01 Comparative Osteology of the Jaws in Representatives of the Eurypterygian Fishes Yazdan Keivany Department of Natural Resources (Fisheries Division), Isfahan University of Technology, Isfahan, 84156-83111, Iran Abstract The osteology of the jaws in representatives of 49 genera in 40 families of eurypterygian fishes, including: Aulopiformes, Myctophiformes, Lampridiformes, Polymixiiformes, Percopsiformes, Mugiliformes, Atheriniformes, Beloniformes, Cyprinodontiformes, Stephanoberyciformes, Beryciformes, Zeiformes, Gasterosteiformes, Synbranchiformes, Scorpaeniformes (including Dactylopteridae), and Perciformes (including Elassomatidae) were studied. Generally, in this group, the upper jaw consists of the premaxilla, maxilla, and supramaxilla. The lower jaw consists of the dentary, anguloarticular, retroarticular, and sesamoid articular. In higher taxa, the premaxilla bears ascending, articular, and postmaxillary processes. The maxilla usually bears a ventral and a dorsal articular process. The supramaxilla is present only in some taxa. The dentary is usually toothed and bears coronoid and posteroventral processes. The retroarticular is small and located at the posteroventral corner of the anguloarticular. Keywords Acanthopterygii, Bone, Eurypterygii, Osteology, Percomprpha following method for clearing and staining bone and 1. Introduction cartilage provided in reference [18]. A camera lucida attached to a Wild M5 dissecting stereomicroscope was used Despite the introduction of modern techniques such as to prepare the drawings. The bones in the first figure of each DNA sequencing and barcoding, osteology, due to its anatomical section are arbitrarily shaded and labeled and in reliability, still plays an important role in the systematic the others are shaded in a consistent manner (dark, medium, study of fishes and comprises a major percent of today’s and clear) to facilitate comparison among the taxa. -

Strong Reproductive Skew Among Males in the Multiply Mated Swordtail Xiphophorus Multilineatus (Teleostei)
Journal of Heredity 2005:96(4):346–355 ª The American Genetic Association. 2005. All rights reserved. doi:10.1093/jhered/esi042 For Permissions, please email: [email protected]. Advance Access publication March 2, 2005 Strong Reproductive Skew Among Males in the Multiply Mated Swordtail Xiphophorus multilineatus (Teleostei) J. LUO,M.SANETRA,M.SCHARTL, AND A. MEYER From Fachbereich Biologie, Universita¨t Konstanz, 78457 Konstanz, Germany (Luo, Sanetra, and Meyer); and Physiologische Chemie I, Biozentrum der Universita¨t, Am Hubland, 97074 Wu¨rzburg, Germany (Schartl). Address correspondence to Axel Meyer, Fachbereich Biologie, Universita¨t Konstanz, Fach M617, Universita¨tsstrasse 10, 78457 Konstanz, Germany, or e-mail: [email protected]. Abstract Male swordtails in the genus Xiphophorus display a conspicuous ventral elongation of the caudal fin, the sword, which arose through sexual selection due to female preference. Females mate regularly and are able to store sperm for at least 6 months. If multiple mating is frequent, this would raise the intriguing question about the role of female choice and male-male competition in shaping the mating system of these fishes. Size-dependent alternate mating strategies occur in Xiphophorus; one such strategy is courtship with a sigmoid display by large dominant males, while the other is gonopodial thrusting, in which small subordinate males sneak copulations. Using microsatellite markers, we observed a frequency of multiple paternity in wild-caught Xiphophorus multilineatus in 28% of families analyzed, but the actual frequency of multiple mating suggested by the correction factor PrDM was 33%. The number of fathers contributing genetically to the brood ranged from one to three. -

Tennessee Fish Species
The Angler’s Guide To TennesseeIncluding Aquatic Nuisance SpeciesFish Published by the Tennessee Wildlife Resources Agency Cover photograph Paul Shaw Graphics Designer Raleigh Holtam Thanks to the TWRA Fisheries Staff for their review and contributions to this publication. Special thanks to those that provided pictures for use in this publication. Partial funding of this publication was provided by a grant from the United States Fish & Wildlife Service through the Aquatic Nuisance Species Task Force. Tennessee Wildlife Resources Agency Authorization No. 328898, 58,500 copies, January, 2012. This public document was promulgated at a cost of $.42 per copy. Equal opportunity to participate in and benefit from programs of the Tennessee Wildlife Resources Agency is available to all persons without regard to their race, color, national origin, sex, age, dis- ability, or military service. TWRA is also an equal opportunity/equal access employer. Questions should be directed to TWRA, Human Resources Office, P.O. Box 40747, Nashville, TN 37204, (615) 781-6594 (TDD 781-6691), or to the U.S. Fish and Wildlife Service, Office for Human Resources, 4401 N. Fairfax Dr., Arlington, VA 22203. Contents Introduction ...............................................................................1 About Fish ..................................................................................2 Black Bass ...................................................................................3 Crappie ........................................................................................7 -

Ecology and Conservation of Mudminnow Species Worldwide
FEATURE Ecology and Conservation of Mudminnow Species Worldwide Lauren M. Kuehne Ecología y conservación a nivel mundial School of Aquatic and Fishery Sciences, University of Washington, Seattle, WA 98195 de los lucios RESUMEN: en este trabajo, se revisa y resume la ecología Julian D. Olden y estado de conservación del grupo de peces comúnmente School of Aquatic and Fishery Sciences, University of Washington, Box conocido como “lucios” (anteriormente conocidos como 355020, Seattle, WA 98195, and Australian Rivers Institute, Griffith Uni- la familia Umbridae, pero recientemente reclasificados en versity, QLD, 4111, Australia. E-mail: [email protected] la Esocidae) los cuales se constituyen de sólo cinco espe- cies distribuidas en tres continentes. Estos peces de cuerpo ABSTRACT: We review and summarize the ecology and con- pequeño —que viven en hábitats de agua dulce y presentan servation status of the group of fishes commonly known as movilidad limitada— suelen presentar poblaciones aisla- “mudminnows” (formerly known as the family Umbridae but das a lo largo de distintos paisajes y son sujetos a las típi- recently reclassified as Esocidae), consisting of only five species cas amenazas que enfrentan las especies endémicas que distributed on three continents. These small-bodied fish—resid- se encuentran en contacto directo con los impactos antro- ing in freshwater habitats and exhibiting limited mobility—often pogénicos como la contaminación, alteración de hábitat occur in isolated populations across landscapes and are subject e introducción de especies no nativas. Aquí se resume el to conservation threats common to highly endemic species in conocimiento actual acerca de la distribución, relaciones close contact with anthropogenic impacts, such as pollution, filogenéticas, ecología y estado de conservación de cada habitat alteration, and nonnative species introductions. -

Luth Wfu 0248D 10922.Pdf
SCALE-DEPENDENT VARIATION IN MOLECULAR AND ECOLOGICAL PATTERNS OF INFECTION FOR ENDOHELMINTHS FROM CENTRARCHID FISHES BY KYLE E. LUTH A Dissertation Submitted to the Graduate Faculty of WAKE FOREST UNIVERSITY GRADAUTE SCHOOL OF ARTS AND SCIENCES in Partial Fulfillment of the Requirements for the Degree of DOCTOR OF PHILOSOPHY Biology May 2016 Winston-Salem, North Carolina Approved By: Gerald W. Esch, Ph.D., Advisor Michael V. K. Sukhdeo, Ph.D., Chair T. Michael Anderson, Ph.D. Herman E. Eure, Ph.D. Erik C. Johnson, Ph.D. Clifford W. Zeyl, Ph.D. ACKNOWLEDGEMENTS First and foremost, I would like to thank my PI, Dr. Gerald Esch, for all of the insight, all of the discussions, all of the critiques (not criticisms) of my works, and for the rides to campus when the North Carolina weather decided to drop rain on my stubborn head. The numerous lively debates, exchanges of ideas, voicing of opinions (whether solicited or not), and unerring support, even in the face of my somewhat atypical balance of service work and dissertation work, will not soon be forgotten. I would also like to acknowledge and thank the former Master, and now Doctor, Michael Zimmermann; friend, lab mate, and collecting trip shotgun rider extraordinaire. Although his need of SPF 100 sunscreen often put our collecting trips over budget, I could not have asked for a more enjoyable, easy-going, and hard-working person to spend nearly 2 months and 25,000 miles of fishing filled days and raccoon, gnat, and entrail-filled nights. You are a welcome camping guest any time, especially if you do as good of a job attracting scorpions and ants to yourself (and away from me) as you did on our trips. -

Esox Lucius) Ecological Risk Screening Summary
Northern Pike (Esox lucius) Ecological Risk Screening Summary U.S. Fish & Wildlife Service, February 2019 Web Version, 8/26/2019 Photo: Ryan Hagerty/USFWS. Public Domain – Government Work. Available: https://digitalmedia.fws.gov/digital/collection/natdiglib/id/26990/rec/22. (February 1, 2019). 1 Native Range and Status in the United States Native Range From Froese and Pauly (2019a): “Circumpolar in fresh water. North America: Atlantic, Arctic, Pacific, Great Lakes, and Mississippi River basins from Labrador to Alaska and south to Pennsylvania and Nebraska, USA [Page and Burr 2011]. Eurasia: Caspian, Black, Baltic, White, Barents, Arctic, North and Aral Seas and Atlantic basins, southwest to Adour drainage; Mediterranean basin in Rhône drainage and northern Italy. Widely distributed in central Asia and Siberia easward [sic] to Anadyr drainage (Bering Sea basin). Historically absent from Iberian Peninsula, Mediterranean France, central Italy, southern and western Greece, eastern Adriatic basin, Iceland, western Norway and northern Scotland.” Froese and Pauly (2019a) list Esox lucius as native in Armenia, Azerbaijan, China, Georgia, Iran, Kazakhstan, Mongolia, Turkey, Turkmenistan, Uzbekistan, Albania, Austria, Belgium, Bosnia Herzegovina, Bulgaria, Croatia, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Ireland, Italy, Latvia, Lithuania, Luxembourg, Macedonia, Moldova, Monaco, 1 Netherlands, Norway, Poland, Romania, Russia, Serbia, Slovakia, Slovenia, Sweden, Switzerland, United Kingdom, Ukraine, Canada, and the United States (including Alaska). From Froese and Pauly (2019a): “Occurs in Erqishi river and Ulungur lake [in China].” “Known from the Selenge drainage [in Mongolia] [Kottelat 2006].” “[In Turkey:] Known from the European Black Sea watersheds, Anatolian Black Sea watersheds, Central and Western Anatolian lake watersheds, and Gulf watersheds (Firat Nehri, Dicle Nehri). -

Summary Report of Freshwater Nonindigenous Aquatic Species in U.S
Summary Report of Freshwater Nonindigenous Aquatic Species in U.S. Fish and Wildlife Service Region 4—An Update April 2013 Prepared by: Pam L. Fuller, Amy J. Benson, and Matthew J. Cannister U.S. Geological Survey Southeast Ecological Science Center Gainesville, Florida Prepared for: U.S. Fish and Wildlife Service Southeast Region Atlanta, Georgia Cover Photos: Silver Carp, Hypophthalmichthys molitrix – Auburn University Giant Applesnail, Pomacea maculata – David Knott Straightedge Crayfish, Procambarus hayi – U.S. Forest Service i Table of Contents Table of Contents ...................................................................................................................................... ii List of Figures ............................................................................................................................................ v List of Tables ............................................................................................................................................ vi INTRODUCTION ............................................................................................................................................. 1 Overview of Region 4 Introductions Since 2000 ....................................................................................... 1 Format of Species Accounts ...................................................................................................................... 2 Explanation of Maps ................................................................................................................................