Pharmacology-2/ Dr
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Package Leaflet: Information for the Patient Desogestrel Rowex
Package leaflet: Information for the patient Desogestrel Rowex 75 microgram Film-coated tablets desogestrel Read all of this leaflet carefully before you start taking this medicine because it contains important information for you. - Keep this leaflet. You may need to read it again. - If you have any further questions, ask your doctor or pharmacist. - This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours. - If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4. What is in this leaflet 1. What Desogestrel Rowex is and what it is used for 2. What you need to know before you take Desogestrel Rowex 4. Possible side effects 5. How to store Desogestrel Rowex 6. Contents of the pack and other information 1. What Desogestrel Rowex is and what it is used for Desogestrel Rowex used to prevent pregnancy. There are 2 main kinds of hormone contraceptive. - The combined pill, "The Pill", which contains 2 types of female sex hormone an oestrogen and a progestogen, - The progestogen-only pill, POP, which doesn't contain an oestrogen. Desogestrel Rowex is a progestogen-only-pill (POP). Desogestrel Rowex contains a small amount of one type of female sex hormone, the progestogen desogestrel. Most POPs work primarily by preventing the sperm cells from entering the womb but do not always prevent the egg cell from ripening, which is the main way that combined pills work. -

Compositions Comprising Drospirenone Molecularly
(19) & (11) EP 1 748 756 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61K 9/00 (2006.01) A61K 9/107 (2006.01) 29.04.2009 Bulletin 2009/18 A61K 9/48 (2006.01) A61K 9/16 (2006.01) A61K 9/20 (2006.01) A61K 9/14 (2006.01) (2006.01) (2006.01) (21) Application number: 05708751.2 A61K 9/70 A61K 31/565 (22) Date of filing: 10.03.2005 (86) International application number: PCT/IB2005/000665 (87) International publication number: WO 2005/087194 (22.09.2005 Gazette 2005/38) (54) COMPOSITIONS COMPRISING DROSPIRENONE MOLECULARLY DISPERSED ZUSAMMENSETZUNGEN AUS DROSPIRENON IN MOLEKULARER DISPERSION DES COMPOSITIONS CONTENANT DROSPIRENONE A DISPERSION MOLECULAIRE (84) Designated Contracting States: (72) Inventors: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR • FUNKE, Adrian HU IE IS IT LI LT LU MC NL PL PT RO SE SI SK TR D-14055 Berlin (DE) Designated Extension States: • WAGNER, Torsten AL BA HR MK YU 13127 Berlin (DE) (30) Priority: 10.03.2004 EP 04075713 (74) Representative: Wagner, Kim 10.03.2004 US 551355 P Plougmann & Vingtoft a/s Sundkrogsgade 9 (43) Date of publication of application: P.O. Box 831 07.02.2007 Bulletin 2007/06 2100 Copenhagen Ø (DK) (60) Divisional application: (56) References cited: 08011577.7 / 1 980 242 EP-A- 1 260 225 EP-A- 1 380 301 WO-A-01/52857 WO-A-20/04022065 (73) Proprietor: Bayer Schering Pharma WO-A-20/04041289 US-A- 5 569 652 Aktiengesellschaft US-A- 5 656 622 US-A- 5 789 442 13353 Berlin (DE) Note: Within nine months of the publication of the mention of the grant of the European patent in the European Patent Bulletin, any person may give notice to the European Patent Office of opposition to that patent, in accordance with the Implementing Regulations. -

Drospirenone-Containing Birth Control Pills (Yaz, Yasmin, Ocella, Gianvi and Their Generics)
Drospirenone-Containing Birth Control Pills (Yaz, Yasmin, Ocella, Gianvi and their generics) Drospirenone is a progestin component of some birth control pills that does some blocking of testosterone (the "male hormone" that women have, too, and the hormone that can be a part of making acne worse). This is why drospirenone-containing birth control pills are especially good at helping acne (and why they can theoretically help hirsutism and female-pattern scalp hair loss as well). Drospirenone is also a mild diuretic and is why these birth control pills do not cause the water weight gain that some birth control pills can. Any birth control pill can theoretically help acne just by evening out hormonal fluctuations, but for many birth control pills, the effects on acne is very minimal. Which birth control pills are most likely to help acne? Some birth control pills are more like testosterone and some are less like testosterone. The ones that are less like testosterone can theoretically help acne more. The drospirenone-containing birth control pills go one step further by blocking testosterone, and may show the most benefit on clearing acne. Drospirenone is a progestin that is derived from 17-alpha-spironolactone and is chemically related to the diuretic spironolactone. Spironolactone is a diuretic (used for high blood pressure) that is sometimes used off-label for the treatment of acne, hirsutism or female-pattern hair loss. Often people say that a drospirenone-containing birth control pill may be equivalent to 25mg of spironolactone. Risks : Any estrogen-containing birth control pill increases the risk of a high blood pressure, stroke, or other life-threatening blood clot, and one study showed possibly a slightly higher risk with a drospirenone-containing birth control pill. -

Ocps) Used in the NM Family Planning Program (FPP
The following slides are intended to familiarize nurses and clinicians with oral contraceptive pills (OCPs) used in the NM Family Planning Program (FPP). The FPP does not intend for this information to supersede the Family Planning Protocol particularly on the requirement for Public Health Nurses in the Public Health Offices to consult a clinician as stated in the Protocol when in doubt or if it is necessary to switch the client’s OCP type. 1 2 Combined oral contraceptives (COCs) contain two hormones; estrogen and progestin. In general, any combined OCP is good for most women who are eligible to take estrogen according to the CDC U.S. Medical Eligibility Criteria (MEC). Once again, refer to the US MEC chart to find out if OCP is a suitable choice for clients with specific health conditions. To learn a little bit about what each hormone does, the FPP is providing the following summary: Estrogen: provides endometrial stability = menstrual cycle control. A higher estrogen dose increases the venous thromboembolism (VTE) or clot risk but OCP clot risk is still less harmful than the clot risk related to pregnancy and giving birth. Progestin: provides most of the contraceptive effect by ‐Preventing luteinizing hormone (LH) surge /ovulation ‐Thickening the cervical mucus to prevent sperm entry. Two major OCP formations are available. Monophasic: There is only one dose of estrogen and progestin in each active pill in the packet; and Multiphasic: There are varying doses of hormones, particularly progestin in the active pills. 3 Section 3 of the FPP Protocol contains the OCP Substitute Table, which groups OCPs into 6 classes according to the estrogen dosage, the type of progestin and the formulations. -

Download PDF File
Ginekologia Polska 2019, vol. 90, no. 9, 520–526 Copyright © 2019 Via Medica ORIGINAL PAPER / GYNECologY ISSN 0017–0011 DOI: 10.5603/GP.2019.0091 Anti-androgenic therapy in young patients and its impact on intensity of hirsutism, acne, menstrual pain intensity and sexuality — a preliminary study Anna Fuchs, Aleksandra Matonog, Paulina Sieradzka, Joanna Pilarska, Aleksandra Hauzer, Iwona Czech, Agnieszka Drosdzol-Cop Department of Pregnancy Pathology, Department of Woman’s Health, School of Health Sciences in Katowice, Medical University of Silesia, Katowice, Poland ABSTRACT Objectives: Using anti-androgenic contraception is one of the methods of birth control. It also has a significant, non-con- traceptive impact on women’s body. These drugs can be used in various endocrinological disorders, because of their ability to reduce the level of male hormones. The aim of our study is to establish a correlation between taking different types of anti-androgenic drugs and intensity of hirsutism, acne, menstrual pain intensity and sexuality . Material and methods: 570 women in childbearing age that had been using oral contraception for at least three months took part in our research. We examined women and asked them about quality of life, health, direct causes and effects of that treatment, intensity of acne and menstrual pain before and after. Our research group has been divided according to the type of gestagen contained in the contraceptive pill: dienogest, cyproterone, chlormadynone and drospirenone. Ad- ditionally, the control group consisted of women taking oral contraceptives without antiandrogenic component. Results: The mean age of the studied group was 23 years ± 3.23. 225 of 570 women complained of hirsutism. -

Comparing the Effects of Combined Oral Contraceptives Containing Progestins with Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis In
JMIR RESEARCH PROTOCOLS Amiri et al Review Comparing the Effects of Combined Oral Contraceptives Containing Progestins With Low Androgenic and Antiandrogenic Activities on the Hypothalamic-Pituitary-Gonadal Axis in Patients With Polycystic Ovary Syndrome: Systematic Review and Meta-Analysis Mina Amiri1,2, PhD, Postdoc; Fahimeh Ramezani Tehrani2, MD; Fatemeh Nahidi3, PhD; Ali Kabir4, MD, MPH, PhD; Fereidoun Azizi5, MD 1Students Research Committee, School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 2Reproductive Endocrinology Research Center, Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 3School of Nursing and Midwifery, Department of Midwifery and Reproductive Health, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran 4Minimally Invasive Surgery Research Center, Iran University of Medical Sciences, Tehran, Islamic Republic Of Iran 5Endocrine Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Islamic Republic Of Iran Corresponding Author: Fahimeh Ramezani Tehrani, MD Reproductive Endocrinology Research Center Research Institute for Endocrine Sciences Shahid Beheshti University of Medical Sciences 24 Parvaneh Yaman Street, Velenjak, PO Box 19395-4763 Tehran, 1985717413 Islamic Republic Of Iran Phone: 98 21 22432500 Email: [email protected] Abstract Background: Different products of combined oral contraceptives (COCs) can improve clinical and biochemical findings in patients with polycystic ovary syndrome (PCOS) through suppression of the hypothalamic-pituitary-gonadal (HPG) axis. Objective: This systematic review and meta-analysis aimed to compare the effects of COCs containing progestins with low androgenic and antiandrogenic activities on the HPG axis in patients with PCOS. -
PMBJP Product.Pdf
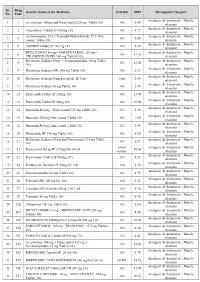
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

Attenuation of Antepartum Relaxin Surge and Induction of Parturition by Antiprogesterone RU 486 in Sheep O
Attenuation of antepartum relaxin surge and induction of parturition by antiprogesterone RU 486 in sheep O. S. Gazal, Y. Li, C. Schwabe and L. L. Anderson 1 Department of Animal Science, Iowa State University, Ames, LA 50011, USA; and 2Department of Biochemistry, Medical University of South Carolina, Charleston, SC 29459, USA Pregnant ewes were injected with either the antiprogesterone, RU 486 (4 mg kg\m=-\1body weight, i.m.; n = 5), 3000 iu relaxin (i.m.; n = 9), or diluent (n = 8) at 12:00 h on days 144 and 145, to determine its effect on progesterone and relaxin secretion, and on induction of lambing. RU 486 induced earlier lambing (P < 0.01) compared with diluent treatment, but relaxin treatment did not significantly reduce the interval to parturition. Mean injection\p=n-\ lambing intervals were 31 \m=+-\2, 109 \m=+-\23 and 121 \m=+-\27 h for the RU 486, relaxin and diluent groups, respectively. There was no incidence of difficult birth (dystocia); all lambs were vigorous at birth; and placenta delivery was rapid (within 207 min) with RU 486 and relaxin treatments compared with diluent treated controls. Plasma progesterone concen- trations averaged 11 ng ml\m=-\1during the pretreatment period for all animals. RU 486 had a biphasic effect on progesterone concentrations, causing an initial increase (P < 0.05) within 2 h, and then an abrupt drop (P < 0.01) to 6 ng ml\m=-\1by 18:00 h on day 145. Progesterone concentrations remained consistently lower (P < 0.05) in relaxin-treated ewes than in diluent\x=req-\ treated controls from days 144 to 147 and then began a steady decrease to 4 ng ml\m=-\1on the day of parturition (days 149 and 150) in both groups. -

Contraception and Misconceptions
CONTRACEPTION AND MISCONCEPTIONS CONTRACEPTION IN WOMEN WITH MENTAL ILLNESS OVERVIEW Hormones and mood How mental illness impacts on contraceptive choice Ideal contraception Pro and cons of contraceptive methods in women with mental illness Hormonal contraception and mood ESTROGENS anti-inflammatory neuroprotective effects of estradiol modulation of the limbic processing memory of emotionally-relevant information. estradiol “beneficially” modulates pathways implicated in the pathophysiology of depression, including serotonin and norepinephrine pathways PROGESTROGENS Previously thought to be anxiogenic, depressogenic Breakdown products Depression – alpha hydroxy progestrogen breakdown products pro – inflammatory, anxiogenic HORMONES AND MOODS – COMPLEX INTERPLAY MENTAL ILLNESS AND CONTRACEPTIVE CHOICE Effect of mental illness on contraceptive choice Effect of contraceptive choice on mental illness Drug interactions EFFECT MENTAL ILLNESS ON CONTRACEPTIVE CHOICE Impulsive Poor planning Cognitive and problem judgement Poor adherence IMPULSIVITY COGNITION POOR JUDGEMENT LETS NOT FORGET SUBSTANCES…… TYPES OF CONTRACEPTION No method Non hormonal Condom/Femidom Diaphragm Non hormone containing IUCD (Copper T) Hormonal COC POP Mirena Evra Patch Nuva Ring Implant ORAL CONTRACEPTIVES POP – avoid Same time every day COC Pros Effective Reduction PMS – monophasic estrogen dominant pill eg Femodene, Yaz, Nordette Cons Drug interactions Pill burden Daily dose EVRA PATCH Weekly patch Transdermal system: 150 mcg/day -

ORTHO TRI-CYCLEN® TABLETS ORTHO-CYCLEN® TABLETS (Norgestimate/Ethinyl Estradiol)
PHYSICIANS' PACKAGE INSERT ORTHO TRI-CYCLEN® TABLETS ORTHO-CYCLEN® TABLETS (norgestimate/ethinyl estradiol) Patients should be counseled that this product does not protect against HIV infection (AIDS) and other sexually transmitted diseases. DESCRIPTION Each of the following products is a combination oral contraceptive containing the progestational compound norgestimate and the estrogenic compound ethinyl estradiol. ORTHO TRI-CYCLEN 21 Tablets and ORTHO TRI-CYCLEN 28 Tablets. Each white tablet contains 0.180 mg of the progestational compound, norgestimate (18,19-Dinor- 17-pregn-4-en-20-yn-3-one,17-(acetyloxy)-13-ethyl-, oxime,(17α)-(+)-) and 0.035 mg of the estrogenic compound, ethinyl estradiol (19-nor-17α-pregna,1,3,5(10)-trien-20-yne-3,17-diol). Inactive ingredients include lactose, magnesium stearate, and pregelatinized starch. Each light blue tablet contains 0.215 mg of the progestational compound norgestimate (18,19- Dinor-17-pregn-4-en-20-yn-3-one,17-(acetyloxy)-13-ethyl-,oxime,(17α)-(+)-) and 0.035 mg of the estrogenic compound, ethinyl estradiol (19-nor-17α-pregna,1,3,5(10)-trien-20-yne-3,17-diol). Inactive ingredients include FD & C Blue No. 2 Aluminum Lake, lactose, magnesium stearate, and pregelatinized starch. Each blue tablet contains 0.250 mg of the progestational compound norgestimate (18,19-Dinor-17- pregn-4-en-20-yn-3-one, 17-(acetyloxy)-13-ethyl-,oxime,(17α)-(+)-) and 0.035 mg of the estrogenic compound, ethinyl estradiol (19-nor-17α-pregna,1,3,5(10)-trien-20-yne-3,17-diol). Inactive ingredients include FD & C Blue No. 2 Aluminum Lake, lactose, magnesium stearate, and pregelatinized starch. -

Pp375-430-Annex 1.Qxd
ANNEX 1 CHEMICAL AND PHYSICAL DATA ON COMPOUNDS USED IN COMBINED ESTROGEN–PROGESTOGEN CONTRACEPTIVES AND HORMONAL MENOPAUSAL THERAPY Annex 1 describes the chemical and physical data, technical products, trends in produc- tion by region and uses of estrogens and progestogens in combined estrogen–progestogen contraceptives and hormonal menopausal therapy. Estrogens and progestogens are listed separately in alphabetical order. Trade names for these compounds alone and in combination are given in Annexes 2–4. Sales are listed according to the regions designated by WHO. These are: Africa: Algeria, Angola, Benin, Botswana, Burkina Faso, Burundi, Cameroon, Cape Verde, Central African Republic, Chad, Comoros, Congo, Côte d'Ivoire, Democratic Republic of the Congo, Equatorial Guinea, Eritrea, Ethiopia, Gabon, Gambia, Ghana, Guinea, Guinea-Bissau, Kenya, Lesotho, Liberia, Madagascar, Malawi, Mali, Mauritania, Mauritius, Mozambique, Namibia, Niger, Nigeria, Rwanda, Sao Tome and Principe, Senegal, Seychelles, Sierra Leone, South Africa, Swaziland, Togo, Uganda, United Republic of Tanzania, Zambia and Zimbabwe America (North): Canada, Central America (Antigua and Barbuda, Bahamas, Barbados, Belize, Costa Rica, Cuba, Dominica, El Salvador, Grenada, Guatemala, Haiti, Honduras, Jamaica, Mexico, Nicaragua, Panama, Puerto Rico, Saint Kitts and Nevis, Saint Lucia, Saint Vincent and the Grenadines, Suriname, Trinidad and Tobago), United States of America America (South): Argentina, Bolivia, Brazil, Chile, Colombia, Dominican Republic, Ecuador, Guyana, Paraguay, -

209627Orig1s000
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 209627Orig1s000 MULTI-DISCIPLINE REVIEW Summary Review Office Director Cross Discipline Team Leader Review Clinical Review Non-Clinical Review Statistical Review Clinical Pharmacology Review Reviewers of Multi-Disciplinary Review and Evaluation SECTIONS OFFICE/ AUTHORED/ ACKNOWLEDGED/ DISCIPLINE REVIEWER DIVISION APPROVED Mark Seggel, Ph.D. OPQ/ONDP/DNDP2 Authored: Section 4.2 Digitally signed by Mark R. Seggel -S CMC Lead DN: c=US, o=U.S. Government, ou=HHS, ou=FDA, ou=People, cn=Mark R. Signature: Mark R. Seggel -S Seggel -S, 0.9.2342.19200300.100.1.1=1300071539 Date: 2018.08.08 16:29:15 -04'00' Frederic Moulin, DVM, PhD OND/ODE3/DBRUP Authored: Section 5 Pharmacology/ Digitally signed by Frederic Moulin -S Toxicology DN: c=US, o=U.S. Government, ou=HHS, ou=FDA, ou=People, Reviewer Signature: Frederic Moulin -S 0.9.2342.19200300.100.1.1=2001708658, cn=Frederic Moulin -S Date: 2018.08.08 15:26:57 -04'00' Kimberly Hatfield, PhD OND/ODE3/DBRUP Approved: Section 5 Pharmacology/ Toxicology Digitally signed by Kimberly P. Hatfield -S DN: c=US, o=U.S. Government, ou=HHS, ou=FDA, ou=People, Team Leader Signature: Kimberly P. Hatfield -S 0.9.2342.19200300.100.1.1=1300387215, cn=Kimberly P. Hatfield -S Date: 2018.08.08 14:56:10 -04'00' Li Li, Ph.D. OCP/DCP3 Authored: Sections 6 and 17.3 Clinical Pharmacology Dig ta ly signed by Li Li S DN c=US o=U S Government ou=HHS ou=FDA ou=People Reviewer cn=Li Li S Signature: Li Li -S 0 9 2342 19200300 100 1 1=20005 08577 Date 2018 08 08 15 39 23 04'00' Doanh Tran, Ph.D.