Bio-Implant Reference Manual
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Nerve Block of Lateral Femoral Cutaneous Nerve of the Thigh
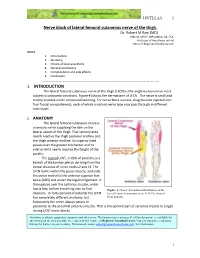
18VTLLAA 1 Nerve block of lateral femoral cutaneous nerve of the thigh. Dr. Robert M Raw (MD) . MBChB, MFGP, MPraxMed, DA, FCA. Professor of Anesthesia retired Editor of Regional-Anesthesia.Com INDEX. 1. Introduction 2. Anatomy 3. Choice of local anesthetic 4. General indications 5. Complications and side effects 6. Conclusion ------------------------------------------------------------------------------------ 1. INTRODUCTION The lateral femoral cutaneous nerve of the thigh (LFCN) is the single human nerve most subject to anatomic variations. Figure #1shows the dermatome of LFCN. The nerve is small and mostly invisible under ultrasound scanning. For nerve block success, drug must be injected into four fascial compartments, each of which a variant nerve type may pass through in different individuals. 2. ANATOMY The lateral femoral cutaneous nerve is a sensory nerve supplying the skin on the lateral aspect of the thigh. That sensory area nearly reaches the thigh posterior midline and the thigh anterior midline. Its superior limit passes over the greater trochanter and its inferior limit nearly reaches the height of the patella. The typical LCNT, in 60% of patients, is a branch of the lumbar plexus deriving from the dorsal divisions of nerve roots L2 and L3. The LFCN forms within the psoas muscle, and exits the pelvis medial to the anterior superior iliac spine (ASIS) and under the inguinal ligament. It then passes over the sartorius muscle, under fascia lata, before branching into its final Figure 1. Classic dermatomal distribution of the divisions. In forty percent of patients the LFCN lateral femoral cutaneous nerve (LFCN), derived has completely different anatomy, but from Sobotta. fortunately the nerve always passes in proximity to the proximal sartorius muscle. -

Preparatory: 1 Venous Access and Medication Administration: 4
Preparatory: 1 Venous Access and Medication Administration: 4 W4444444444444444444444444444444444444444444444444444444444444444444444444444444444444444444444444444444444444 UNIT TERMINAL OBJECTIVE 1-4 At the completion of this unit, the EMT-Critical Care Technician student will be able to safely and precisely access the venous circulation and administer medications. COGNITIVE OBJECTIVES At the completion of this unit, the EMT-Critical Care Technician student will be able to: 1-4.1 Review the specific anatomy and physiology pertinent to medication administration. (C-1) 1-4.2 Review mathematical principles. (C-1) 1-4.3 Review mathematical equivalents. (C-1) 1-4.4 Differentiate temperature readings between the Centigrade and Fahrenheit scales. (C-3) 1-4.5 Discuss formulas as a basis for performing drug calculations. (C-1) 1-4.6 Calculate oral and parenteral drug dosages for all emergency medications administered to adults, infants and children. (C-2) 1-4.7 Calculate intravenous infusion rates for adults, infants, and children. (C-2) 1-4.8 Discuss legal aspects affecting medication administration. (C-1) 1-4.9 Discuss the "six rights" of drug administration and correlate these with the principles of medication administration. (C-1) 1-4.10 Discuss medical asepsis and the differences between clean and sterile techniques. (C-1) 1-4.11 Describe use of antiseptics and disinfectants. (C-1) 1-4.12 Describe the use of universal precautions and body substance isolation (BSI) procedures when administering a medication. (C-1) 1-4.13 Describe the indications, equipment needed, techniques utilized, precautions, and general principles of peripheral venous cannulation (Including saline locks). (C-1) 1-4.14 Describe the indications, equipment needed, techniques utilized, precautions, and general principles of intraosseous needle placement and infusion. -

Handbook ESRA
TECHNIQUES HEAD & NECK 4 Intracranial surgery p. 3 Eye blocks p. 5 Face anatomy p. 16 Face particularity p. 23 Ophtalmic nerve blocks p. 27 Maxillary nerve blocks p. 33 Mandibular nerve blocks p. 46 THORAX & ABDOMEN 50 Epidural anaesthesia in Cardio-thoracic surgery p. 50 Ilioinguinal-Iliohypogastric block p. 55 Peri-umbilical & Rectus sheath block p. 57 Pudendal block p. 58 UPPER LIMB 61 Choice of a technique p. 61 Brachial plexus anatomy p. 65 Interscalen block p. 68 Supraclavicular blocks p. 73 Infraclavicular blocks p. 80 Axillary block p. 83 LOWER LIMB 90 Lumbar plexus block p. 90 Iliofascial block p. 100 Obturator block p. 102 Sciatic blocks o Sciatic blocks - parasacral nerve approach p. 109 o Sciatic blocks - posterior popliteal approach p. 115 Ankle blocks p. 119 AXIAL BLOCKS 123 Lumbar epidural p. 123 OBSTETRICS AXIAL BLOCKS 126 Epidural p. 126 PERIPHERAL BLOCKS Pudendal block p. 58 2 Aknowledgement The provenience of the materials included in this handbook is from the Learning Zone on the official site of “European Society of Regional Anesthesia and Pain Therapy”. http://www.esra-learning.com/ 2007 3 HEAD & TABLE OF CONTENTS NECK • Intracranial surgery • Eye blocks • Face anatomy • Face particularity • Ophtalmic nerve blocks • Maxillary nerve blocks • Mandibular nerve blocks • Cervical plexus blocks HEAD & INTRACRANIAL SURGERY NECK Paul J. Zetlaoui, M.D. Kremlin-Bicetre - France In intra-cranial neurosurgery, scalp infiltration aims to prevent systematic and cerebral hemodynamic variations, contemporary of skin incision. The potential morbidity of these hypertension-tachycardia episodes, even in patients profoundly anaesthetized, is secondary in the increase of the cerebral blood flow and in its deleterious consequences on intra-cranial pressure in these compromised patients. -

The Development of an Intramuscular Injection Simulation for Nursing Students
Open Access Technical Report DOI: 10.7759/cureus.12366 The Development of an Intramuscular Injection Simulation for Nursing Students Julia Micallef 1 , Artur Arutiunian 1 , Adam Dubrowski 1 1. Health Sciences, Ontario Tech University, Oshawa, CAN Corresponding author: Adam Dubrowski, [email protected] Abstract Intramuscular (IM) injections are preferred over subcutaneous injections for administering medicine such as epinephrine and vaccines as the muscle tissue contains an increased vascular supply that provides ideal absorption of the drug being administered. However, administering an IM injection requires clinical judgment when choosing the injection site, understanding the relevant anatomy and physiology as well as the principles and techniques for administering an IM injection. Therefore, it is essential to learn and perform IM injections using injection simulators to practice the skill before administering to a real patient. Current IM injection simulators either favor realism at the expense of standardization or are expensive but do not provide a realistic experience. Therefore, it is imperative to develop an inexpensive but realistic intramuscular injection simulator that can be used to train nursing students so that they can be prepared for when they enter the clinical setting. This technical report aims to provide an overview of the development of an inexpensive and realistic deltoid simulator geared to teach nursing students the skill of IM injections. After development, the IM simulators were tested and validated by practicing nurses. An 18-item survey was administered to the nurses, and results indicated positive feedback about the realism of the simulator, in comparison to previous models used, such as the Wallcur® PRACTI-Injecta Pads (Wallcur LLC, San Diego, CA). -

The Digestive System
69 chapter four THE DIGESTIVE SYSTEM THE DIGESTIVE SYSTEM The digestive system is structurally divided into two main parts: a long, winding tube that carries food through its length, and a series of supportive organs outside of the tube. The long tube is called the gastrointestinal (GI) tract. The GI tract extends from the mouth to the anus, and consists of the mouth, or oral cavity, the pharynx, the esophagus, the stomach, the small intestine, and the large intes- tine. It is here that the functions of mechanical digestion, chemical digestion, absorption of nutrients and water, and release of solid waste material take place. The supportive organs that lie outside the GI tract are known as accessory organs, and include the teeth, salivary glands, liver, gallbladder, and pancreas. Because most organs of the digestive system lie within body cavities, you will perform a dissection procedure that exposes the cavities before you begin identifying individual organs. You will also observe the cavities and their associated membranes before proceeding with your study of the digestive system. EXPOSING THE BODY CAVITIES should feel like the wall of a stretched balloon. With your skinned cat on its dorsal side, examine the cutting lines shown in Figure 4.1 and plan 2. Extend the cut laterally in both direc- out your dissection. Note that the numbers tions, roughly 4 inches, still working with indicate the sequence of the cutting procedure. your scissors. Cut in a curved pattern as Palpate the long, bony sternum and the softer, shown in Figure 4.1, which follows the cartilaginous xiphoid process to find the ventral contour of the diaphragm. -

Pressure Ulcer Staging Guide
Pressure Ulcer Staging Guide Pressure Ulcer Staging Guide STAGE I STAGE IV Intact skin with non-blanchable Full thickness tissue loss with exposed redness of a localized area usually Reddened area bone, tendon, or muscle. Slough or eschar may be present on some parts Epidermis over a bony prominence. Darkly Epidermis pigmented skin may not have of the wound bed. Often includes undermining and tunneling. The depth visible blanching; its color may Dermis of a stage IV pressure ulcer varies by Dermis differ from the surrounding area. anatomical location. The bridge of the This area may be painful, firm, soft, nose, ear, occiput, and malleolus do not warmer, or cooler as compared to have subcutaneous tissue and these adjacent tissue. Stage I may be Adipose tissue ulcers can be shallow. Stage IV ulcers Adipose tissue difficult to detect in individuals with can extend into muscle and/or Muscle dark skin tones. May indicate "at supporting structures (e.g., fascia, Muscle risk" persons (a heralding sign of Bone tendon, or joint capsule) making risk). osteomyelitis possible. Exposed bone/ Bone tendon is visible or directly palpable. STAGE II DEEP TISSUE INJURY Partial thickness loss of dermis Blister Purple or maroon localized area of Reddened area presenting as a shallow open ulcer discolored intact skin or blood-filled Epidermis with a red pink wound bed, without Epidermis blister due to damage of underlying soft slough. May also present as an tissue from pressure and/or shear. The intact or open/ruptured serum-filled Dermis area may be preceded by tissue that is Dermis blister. -

Avoid Empiric Treatment for Vulvar Skin Disorders
December 1, 2008 • www.familypracticenews.com Women’s Health 27 Avoid Empiric Treatment for Vulvar Skin Disorders BY NANCY WALSH vulva and anus. Hypopigmentation also is characteristic, ment once daily after soaking. “I believe the Temovate New York Bureau with scarring and architectural changes including phimo- brand is much better than the generic, probably because sis of the clitoris, resorption of the labia minora, and nar- of the vehicle,” he said. The corticosteroid should be con- L AKE B UENA V ISTA, FLA. — Empiric treatment rowing of the introitus causing recurrent tearing. It prob- tinued until active disease has resolved, not just for the 2 with corticosteroids should be avoided in patients who pre- ably is autoimmune, because patients have a high incidence weeks specified in the package insert. sent with vulvar symptoms such as burning, itching, pain, of other autoimmune diseases, especially thyroid disease. A second vulvar condition, lichen simplex chronicus, and dyspareunia, according to Dr. Andrew T. Goldstein. Lichen sclerosus can develop at any age, including is characterized by thick, lichenified skin of the labia ma- These patients should have a careful examination of the childhood, and is more common than generally appreci- jora and interlabial sulcus, accompanied by erosions, fis- vulva using a colposcope, and if a lesion is present, a 4- ated, with a prevalence of 1 in 70 women. But “you have suring, and tears in the skin that result from the patient’s mm punch biopsy is warranted. to look for it. The vulva is not just something to separate scratching in her sleep, said Dr. -

Unit 4: Medication Administration Fundamental of Nursing
Unit 4: Medication Administration Fundamental of Nursing Unit 4: Medication Administration: Medication: Is a substance administered for the diagnosis, cure, treatment, relief, or prevention of disease. Six Rights of Medication Administration After paramedics have received the medication or fluid order, they should then administer the drug in question. In performing drug administration, pre-hospital care providers adhere to the six rights of medication administration: 1. Right patient 2. Right medication 3. Right dose 4. Right route 5. Right time 6. Right documentation Basic principle of nurse on drugs administration 1. The nurse must know the drug's prescribed dose, method of administration, actions, expected therapeutic effect, possible interactions with other drugs, and adverse effects. 2. The nurse must know the institution's administration procedures for the client's welfare and the nurse's legal protection. 3. The nurse must Review physician's order for completeness the client's name, date of the order, name of the drug, dose, rout, time of administration, and the physician's signature. 1 Unit 4: Medication Administration Fundamental of Nursing 4. The nurse discusses the medication and its actions with the client; recheck the medication order if the client disagrees with the dose or the physician's order. 5. The nurse must check the physician's order against the client's medication administration record for accuracy. 6. The nurse gives the patient the right to know about the medication he is receiving and the right to refuse it. Routes of Administration A: Enteral Tract Routes The common enteral routes of administration used in general medical practice are as follows: 1. -

Cryoneurolysis and Percutaneous Peripheral Nerve Stimulation To
REVIEW ARTICLE Deborah J. Culley, M.D., Editor ABSTRACT Two regional analgesic modalities currently cleared by the U.S. Food and Drug Administration hold promise to provide postoperative analgesia free of many of the limitations of both opioids and local anesthetic-based techniques. Cryoneurolysis and Cryoneurolysis uses exceptionally low temperature to reversibly ablate a peripheral nerve, resulting in temporary analgesia. Where applicable, it offers Percutaneous Peripheral a unique option given its extended duration of action measured in weeks to months after a single application. Percutaneous peripheral nerve stimula- Nerve Stimulation to Treat tion involves inserting an insulated lead through a needle to lie adjacent to a peripheral nerve. Analgesia is produced by introducing electrical current with Downloaded from http://pubs.asahq.org/anesthesiology/article-pdf/133/5/1127/513611/20201100.0-00028.pdf by guest on 27 September 2021 Acute Pain an external pulse generator. It is a unique regional analgesic in that it does not induce sensory, motor, or proprioception deficits and is cleared for up to A Narrative Review 60 days of use. However, both modalities have limited validation when applied to acute pain, and randomized, controlled trials are required to define both Brian M. Ilfeld, M.D., M.S., John J. Finneran IV, M.D. benefits and risks. (ANESTHESIOLOGY 2020; 133:1127–49) ANESTHESIOLOGY 2020; 133:1127–49 OTENT site-specific analgesia may be provided with at the probe tip results in a corresponding volume expansion Pregional anesthetics -

DERMIS PLUS PADS and STRIPS SKIN INTEGRITY PRODUCTS Ensuring Comfort and Care
DERMIS PLUS PADS AND STRIPS SKIN INTEGRITY PRODUCTS Ensuring Comfort and Care 1 2 Based on the policy that prevention is better MacMed Dermis Plus Gel Pads and Dermis Plus Gel Strips are soft, conformable than cure, MacMed Healthcare have and elastic, and may be used for many different applications such as: introduced the MacMed Dermis Plus Gel Pad • and Dermis Plus Gel Strip to assist with Podiatry applications such as protection between toes, plantar fasciitis, pressure offloading, in difficult to reach and and hallux valgus (bunions) • treat areas. Protection under breasts and over nipples • Ear protection and bony cartilage >1 DERMIS PLUS GEL PADS • Protection with orthopedic appliances such as splints and cervical collars >2 DERMIS PLUS GEL STRIPS • Protection from collars, and splints, oxygen cannulas, nasal-gastric tubing and catheter tubing Dermis Plus Gel Pads and Dermis Plus • General operating theatre use to preserve and protect skin integrity Gel Strips are recommended for pressure • Protection between skin folds and other vulnerable areas off-loading in patients that are medium to • Protection of bony prominences such as elbows, shins and heels high risk of developing compromised skin • integrity issues such as pressure ulcers, Use with prosthesis and other amputee issues, including contractions skin tears or calluses due to unrelieved • Protection of the sacral area pressure, shear and friction. • Protection of the occipital area Can be secured in place Non-toxic with low tack tape Hypo-allergenic and latex free Can be cut -

Cell Culture in Three-Dimensional Lattices of Hydrated Collagen Fibrils − Its Progress and Perspectives
N. Saito et al., Int. J. of Design & Nature and Ecodynamics. Vol. 10, No. 1 (2015) 1–9 CELL CULTURE IN THREE-DIMENSIONAL LATTICES OF HYDRATED COLLAGEN FIBRILS − ITS PROGRESS AND PERSPECTIVES N. SAITO1, H. ADACHI2, H. TANAKA2, S. NAKATA2, N. KAWADA1 & K. YOSHIZATO1,3 1Department of Hepatology, Osaka City University, Japan. 2Nippon Menard Cosmetic Co. Ltd., Japan. 3PhoenixBio Co. Ltd., Japan ABSTRACT Cell culture methodology has been utilized as a powerful tool to characterize cells with a presupposition that the cells in culture faithfully reproduce their in vivo phenotypes. However, it is generally recognized that the cells cultured currently by the most commonly utilized two-dimensional (2D) monolayer technology exhibit different phenotypes from those in living tissues. Cultivation of cells in three-dimensional (3D) lattices of collagen fibrils (collagen gel culture) has been thought to overcome the shortages of 2D cell culture such that the cells behave as in vivo by interacting with not only nearby other cells but also by surrounding extracellular matrices. A remarkable outcome of studies on collagen gel culture was a demonstration of contractile nature of the cells. One of the not-fully appreciated issues about the 3D cell culture is the effect of fluid flow through the collagen gels on cells’ phenotypes. In this review, we make a short overview of historical and current studies of the collagen gel culture from a viewpoint of ‘how we can more correctly extract the cell-related phenomena in living tissues/organs by in vitro culture technology, placing an emphasis on the importance of stimuli caused by flow of cell culture medium.’ Keywords: Collagen gel culture, connective tissue modeling, ECM compositions, gel contraction, interstitial fluid flow, tensile strength. -

Biopharmaceutical Study of Triamcinolone Acetonide Semisolid Formulations for Sublingual and Buccal Administration †
pharmaceutics Article Biopharmaceutical Study of Triamcinolone Acetonide Semisolid Formulations for Sublingual and Buccal Administration † Marta Márquez Valls 1, Alejandra Martínez Labrador 1, Lyda Halbaut Bellowa 1 , Doménica Bravo Torres 1, Paulo C. Granda 1 , Montserrat Miñarro Carmona 1, David Limón 2 and Ana C. Calpena Campmany 1,* 1 Department of Pharmacy and Pharmaceutical Technology and Physical Chemistry, Faculty of Pharmacy and Food Science, University of Barcelona, Av. Joan XXIII 29-31, 08028 Barcelona, Spain; [email protected] (M.M.V.); [email protected] (A.M.L.); [email protected] (L.H.B.); [email protected] (D.B.T.); [email protected] (P.C.G.); [email protected] (M.M.C.) 2 Department of Pharmacology, Toxicology and Therapeutic Chemistry, Faculty of Pharmacy and Food Science, University of Barcelona, Av. Joan XXIII 29-31, 08028 Barcelona, Spain; [email protected] * Correspondence: [email protected] † This paper is an extended versión of paper published in the 1st International Electronic Conference on Pharmaceutics, 1–15 December 2020. Abstract: The mouth can be affected by important inflammatory processes resulting from localized or systemic diseases such as diabetes, AIDS and leukemia, among others, and are manifested in Citation: Márquez Valls, M.; various types of buccal sores typically presenting pain. This work focuses on the design, formulation, Martínez Labrador, A.; Halbaut and characterization of four semisolid formulations for oral mucosa in order to symptomatically treat Bellowa, L.; Bravo Torres, D.; Granda, these painful processes. The formulations have two active pharmaceutical ingredients, triamcinolone P.C.; Miñarro Carmona, M.; Limón, acetonide (TA) and lidocaine hydrochloride (LIDO). The formula also contains, as an excipient, D.; Calpena Campmany, A.C.