Physicians As Assistants at Surgery: 2020 Update
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Minimally Invasive Superficial Femoral Artery Endarterectomy: Early Experience with a Modified Technique
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Eur J Vasc Endovasc Surg 16, 254-258 (1998) ENDOVASCULAR AND SURGICAL TECHNIQUES Minimally Invasive Superficial Femoral Artery Endarterectomy: Early Experience with a Modified Technique M. S. Whiteley 1, T. R. Magee 1, E. P. H. Torrie 2 and R. B. Galland* Department of ~Surgery and 2Radiology, Royal Berkshire Hospital, London Road, Reading, U.K. Objectives: To describe our experience of a modified technique for carrying out remote endarterectomy for superficial femoral artery occlusive disease. Methods: A 4-French arterial dilator is inserted using a Smart needle into the popliteal artery below the occlusion. A remote endarterectomy is carried out through an arteriotomy in the proximal superficial femoral artery. The atheroma is cut distal to the lower extent of disease using a Moll ring cutter. The lower flap of atheroma is secured with an intraluminaI stent inserted from the arteriotomy in the superficial femoral artery. The arteriotomy is extended into the common femoral artery and closed with a vein patch. Results: The procedure was completed in 21 of 26 limbs. In 18 cases the superficial femoral artery remained patent at 30 days. Of the 21 cases all but four stayed in hospital for one night. A successful femoropopliteal bypass was carried out in the five patients in whom the procedure was not completed. Conclusion: Insertion of the dilator into the popliteal artery distal to the occlusion before carrying out the remote endarterectomy has two advantages. Firstly, the stent insertion is carried out in the correct plane and prevents dissection of the distal cut atheroma when attempting to pass the guidewire from above. -

Medical Policy
Medical Policy Joint Medical Policies are a source for BCBSM and BCN medical policy information only. These documents are not to be used to determine benefits or reimbursement. Please reference the appropriate certificate or contract for benefit information. This policy may be updated and is therefore subject to change. *Current Policy Effective Date: 5/1/21 (See policy history boxes for previous effective dates) Title: Composite Tissue Allotransplantation Description/Background Composite tissue allotransplantation refers to the transplantation of histologically different tissue that may include skin, connective tissue, blood vessels, muscle, bone, and nerve tissue. The procedure is also known as reconstructive transplantation. To date, primary applications of this type of transplantation have been of the hand and face (partial and full), although there are also reported cases of several other composite tissue allotransplantations, including that of the larynx, knee, and abdominal wall. The first successful partial face transplant was performed in France in 2005, and the first complete facial transplant was performed in Spain in 2010. In the United States, the first facial transplant was done in 2008 at the Cleveland Clinic; this was a near-total face transplant and included the midface, nose, and bone. The first hand transplant with short-term success occurred in 1998 in France. However, the patient failed to follow the immunosuppressive regimen, which led to graft failure and removal of the hand 29 months after transplantation. The -

Rapidly Growing Epstein-Barr Virus-Associated Pulmonary Lymphoma After Heart Transplantation
Eur Respir J., 1994, 7, 612–616 Copyright ERS Journals Ltd 1994 DOI: 10.1183/09031936.94.07030612 European Respiratory Journal Printed in UK - all rights reserved ISSN 0903 - 1936 CASE REPORT Rapidly growing Epstein-Barr virus-associated pulmonary lymphoma after heart transplantation M. Schwend*, M. Tiemann**, H.H. Kreipe**, M.R. Parwaresch**, E.G. Kraatz+, G. Herrmann++, R.P. Spielmann$, J. Barth* Rapidly growing Epstein-Barr virus-associated pulmonary lymphoma after heart trans- Dept of *Internal Medicine, **Hemato- plantation. M. Schwend, M. Tiemann, H.H. Kreipe, M.R. Parwaresch, E.G. Kraatz, G. pathology, +Cardiovascular Surgery, Herrmann, R.P. Spielmann, J. Barth. ERS Journals Ltd 1994. ++Cardiology, and $Radiographic Diagnostics, ABSTRACT: There is strong evidence to show an association of Epstein-Barr virus Christian-Albrechts-University of Kiel, Kiel, Germany. (EBV) infection with the development of post-transplant lymphoproliferative dis- ease. We report the rapid development of a malignant lymphoma in a heart trans- Correspondence: J. Barth plant recipient, which occurred within less than eight weeks. I. Medizinische Universitätsklinik The diagnosis of this malignant high grade B-cell lymphoma was established by Schittenhelmstr. 12 open lung biopsy, and classified as centroblastic lymphoma of polymorphic subtype. D-24105 Kiel Immunohistochemically, the lymphoma showed reactivity with the B-cell markers Germany L-26 (CD20) and Ki-B5 and with the activation marker Ber-H2 (CD30). Furthermore, an expression of the bcl-2 oncoprotein was detected. Monoclonal JH gene rearrange- Keywords: Epstein-Barr virus ment was demonstrated by polymerase chain reaction (PCR), indicating monoclonal heart transplantation pulmonary lymphoma proliferation of B-blasts. -

Synovial Fluidfluid 11
LWBK461-c11_p253-262.qxd 11/18/09 6:04 PM Page 253 Aptara Inc CHAPTER SynovialSynovial FluidFluid 11 Key Terms ANTINUCLEAR ANTIBODY ARTHROCENTESIS BULGE TEST CRYSTAL-INDUCED ARTHRITIS GROUND PEPPER HYALURONATE MUCIN OCHRONOTIC SHARDS RHEUMATOID ARTHRITIS (RA) RHEUMATOID FACTOR (RF) RICE BODIES ROPE’S TEST SEPTIC ARTHRITIS Learning Objectives SYNOVIAL SYSTEMIC LUPUS ERYTHEMATOSUS 1. Define synovial. VISCOSITY 2. Describe the formation and function of synovial fluid. 3. Explain the collection and handling of synovial fluid. 4. Describe the appearance of normal and abnormal synovial fluids. 5. Correlate the appearance of synovial fluid with possible cause. 6. Interpret laboratory tests on synovial fluid. 7. Suggest further testing for synovial fluid, based on preliminary results. 8. List the four classes or categories of joint disease. 9. Correlate synovial fluid analyses with their representative disease classification. 253 LWBK461-c11_p253-262.qxd 11/18/09 6:04 PM Page 254 Aptara Inc 254 Graff’s Textbook of Routine Urinalysis and Body Fluids oint fluid is called synovial fluid because of its resem- blance to egg white. It is a viscous, mucinous substance Jthat lubricates most joints. Analysis of synovial fluid is important in the diagnosis of joint disease. Aspiration of joint fluid is indicated for any patient with a joint effusion or inflamed joints. Aspiration of asymptomatic joints is beneficial for patients with gout and pseudogout as these fluids may still contain crystals.1 Evaluation of physical, chemical, and microscopic characteristics of synovial fluid comprise routine analysis. This chapter includes an overview of the composition and function of synovial fluid, and laboratory procedures and their interpretations. -

Validation of a Clinical Prediction Rule for the Differentiation Between Septic Arthritis and Transient Synovitis of the Hip in Children by MININDER S
COPYRIGHT © 2004 BY THE JOURNAL OF BONE AND JOINT SURGERY, INCORPORATED Validation of a Clinical Prediction Rule for the Differentiation Between Septic Arthritis and Transient Synovitis of the Hip in Children BY MININDER S. KOCHER, MD, MPH, RAHUL MANDIGA, BS, DAVID ZURAKOWSKI, PHD, CAROL BARNEWOLT, MD, AND JAMES R. KASSER, MD Investigation performed at the Departments of Orthopaedic Surgery, Biostatistics, and Radiology, Children’s Hospital, Boston, Massachusetts Background: The differentiation between septic arthritis and transient synovitis of the hip in children can be difficult. The purpose of the present study was to validate a previously published clinical prediction rule for this differentiation in a new patient population. Methods: We prospectively studied children who presented to a major children’s hospital between 1997 and 2002 with an acutely irritable hip. As in the previous study, diagnoses of septic arthritis (fifty-one patients) and transient synovitis (103 patients) were operationally defined on the basis of the white blood-cell count in the joint fluid, the re- sults of cultures of joint fluid and blood, and the clinical course. Univariate analysis and multiple logistic regression were used to compare the two groups. The predicted probability of septic arthritis of the hip from the prediction rule was compared with actual distributions in the current patient population. The area under the receiver operating char- acteristic curve was determined. Results: The same four independent predictors of septic arthritis of the hip (a history of fever, non-weight-bearing, an erythrocyte sedimentation rate of 40 mm/hr, and a serum white blood-cell count of >12,000 cells/mm3 (>12.0 × 109/L)) were identified in the current patient population. -

Targeting Endothelial Kruppel-Like Factor 2 (KLF2) in Arteriovenous
Targeting Endothelial Krüppel-like Factor 2 (KLF2) in Arteriovenous Fistula Maturation Failure A dissertation submitted to the Graduate School of the University of Cincinnati in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY (Ph.D.) in the Biomedical Engineering Program Department of Biomedical Engineering College of Engineering and Applied Science 2018 by Keith Louis Saum B.S., Wright State University, 2012 Dissertation Committee: Albert Phillip Owens III, Ph.D. (Committee Chair) Begona Campos-Naciff, Ph.D Christy Holland, Ph.D. Daria Narmoneva, Ph.D. Prabir Roy-Chaudhury, M.D., Ph.D Charuhas Thakar, M.D. Abstract The arteriovenous fistula (AVF) is the preferred form of vascular access for hemodialysis. However, 25-60% of AVFs fail to mature to a state suitable for clinical use, resulting in significant morbidity, mortality, and cost for end-stage renal disease (ESRD) patients. AVF maturation failure is recognized to result from changes in local hemodynamics following fistula creation which lead to venous stenosis and thrombosis. In particular, abnormal wall shear stress (WSS) is thought to be a key stimulus which alters endothelial function and promotes AVF failure. In recent years, the transcription factor Krüppel-like factor-2 (KLF2) has emerged as a key regulator of endothelial function, and reduced KLF2 expression has been shown to correlate with disturbed WSS and AVF failure. Given KLF2’s importance in regulating endothelial function, the objective of this dissertation was to investigate how KLF2 expression is regulated by the hemodynamic and uremic stimuli within AVFs and determine if loss of endothelial KLF2 is responsible for impaired endothelial function. -

114.3 Cmr: Division of Health Care Finance and Policy Ambulatory Care
114.3 CMR: DIVISION OF HEALTH CARE FINANCE AND POLICY AMBULATORY CARE 114.3 CMR 40.00: RATES FOR SERVICES UNDER M.G.L. c. 152, WORKERS’ COMPENSATION ACT Section 40.01: General Provisions 40.02: General Definitions 40.03: Service and Rate Coverage Provisions 40.04: Provisions Affecting Eligible Providers 40.05: Policies for Individual Service Types 40.06: Fees 40.07: Appendices 40.08: Severability 40.01: General Provisions (1) Scope, Purpose and Effective Date. 114.3 CMR 40.00 governs the payment rates effective April 1, 2009 for purchasers of health care services under M.G.L. c. 152, the Workers’ Compensation Act. Payment rates for services provided by hospitals are set forth in 114.1 CMR 41.00. Program policies relating to medical necessity and clinical appropriateness are determined pursuant to M.G.L. c. 152 and 452 CMR 6.00. (2) Coverage. The payment rates set forth in 114.3 CMR 40.06 are full payment for services provided under M.G.L. c. 152, § 13, including any related administrative or overhead costs. The insurer, employer and health care service provider may agree upon a different payment rate for any service set forth in the fee schedule in 114.3 CMR 40.00. No employee may be held liable for the payment for health care services determined compensable under M.G.L. c. 152, § 13. (3) Administrative Bulletins. The Division may issue administrative bulletins to clarify substantive provisions of 114.3 CMR 40.00, or to publish procedure code updates and corrections. For coding updates and correction, the bulletin will list: (a) new code numbers for existing codes, with the corresponding cross references between existing and new codes numbers; (b) deleted codes for which there are no corresponding new codes; and (c) codes for entirely new services that require pricing. -

ICD~10~PCS Complete Code Set Procedural Coding System Sample
ICD~10~PCS Complete Code Set Procedural Coding System Sample Table.of.Contents Preface....................................................................................00 Mouth and Throat ............................................................................. 00 Introducton...........................................................................00 Gastrointestinal System .................................................................. 00 Hepatobiliary System and Pancreas ........................................... 00 What is ICD-10-PCS? ........................................................................ 00 Endocrine System ............................................................................. 00 ICD-10-PCS Code Structure ........................................................... 00 Skin and Breast .................................................................................. 00 ICD-10-PCS Design ........................................................................... 00 Subcutaneous Tissue and Fascia ................................................. 00 ICD-10-PCS Additional Characteristics ...................................... 00 Muscles ................................................................................................. 00 ICD-10-PCS Applications ................................................................ 00 Tendons ................................................................................................ 00 Understandng.Root.Operatons..........................................00 -

Canadian Surgery Forum Canadien De Chirurgie
Vol. 44, Suppl., August / août 2001 ISSN 0008-428X ABSTRACTS RÉSUMÉS of presentations to the des communications présentées Annual Meetings of the aux congrès annuels de la Canadian Society of Colon Société canadienne and Rectal Surgeons des chirurgiens du côlon et du rectum Canadian Association of General Surgeons Association canadienne des chirurgiens généraux Canadian Association of Thoracic Surgeons Association canadienne des chirurgiens thoraciques CANADIAN SURGERY FORUM CANADIEN DE CHIRURGIE Québec, QC September 6 to 9, 2001 Québec (QC) du 6 au 9 septembre 2001 Abstracts Résumés Canadian Surgery Forum canadien de chirurgie 2001 Canadian Society of Colon and Rectal Surgeons Société canadienne des chirurgiens du côlon et du rectum 1 2 ARTIFICIAL BOWEL SPHINCTER IMPLANTATION COMPARISON OF DELORME AND ALTEMEIER IN THE MANAGEMENT OF SEVERE FECAL IN- PROCEDURES FOR RECTAL PROLAPSE. E.C. McKe- CONTINENCE — EXPERIENCE FROM A SINGLE vitt, B.J. Sullivan, P.T. Phang. Department of Surgery, St. INSTITUTION. A.R. MacLean, G. Stewart, K. Sabr, M. Paul’s Hospital, University of British Columbia, Vancou- Burnstein. Department of Surgery, St Michael’s Hospital, ver, BC University of Toronto, Toronto, Ont. We wish to compare the outcomes of 2 perineal operations for The purpose of this study was to evaluate the safety and effi- rectal prolapse: rectal mucosectomy (Delorme’s operation) cacy of artificial bowel sphincter (ABS) implantation in the and perineal rectosigmoidectomy (Altemeier’s operation). management of severe fecal incontinence (FI). We reviewed all 34 patients who had a perineal repair of Ten patients (6 males), with a mean age of 40.6 years, un- rectal prolapse at our hospital from July 1997 to June 2000. -

Gender Confirmation Surgery Reference Number: PA.CP.MP.95 Effective Date: 01/18 Coding Implications Last Review Date: 09/17 Revision Log
Clinical Policy: Gender Confirmation Surgery Reference Number: PA.CP.MP.95 Effective Date: 01/18 Coding Implications Last Review Date: 09/17 Revision Log Description Services for gender confirmation most often include hormone treatment, counseling, psychotherapy, complete hysterectomy, bilateral mastectomy, chest reconstruction or augmentation as appropriate, genital reconstruction, facial hair removal, and certain facial plastic reconstruction. Not every individual will require each intervention so necessity needs to be considered on an individualized basis. This criteria outlines medical necessity criteria for gender confirmation surgery when such services are included under the members’ benefit plan contract provisions. Policy/Criteria It is the policy of Pennsylvania Health and Wellness® (PHW) that the gender confirmation surgeries listed in section III are considered medically necessary for members when diagnosed with gender dysphoria per criteria in section I and when meeting eligibility criteria in section II. I. Gender Dysphoria Criteria, meets A and B A. Marked incongruence between the member’s experienced/expressed gender and assigned gender, of at least 6 month’s duration, as indicated by two or more of the following: 1. Marked incongruence between the member’s experienced/expressed gender and primary and/or secondary sex characteristics; 2. A strong desire to be rid of one’s primary and/or secondary sex characteristics because of a marked incongruence with one’s experienced/expressed gender; 3. A strong desire for the primary and/or secondary sex characteristics of the other gender; 4. A strong desire to be of the other gender (or some alternative gender different from one’s assigned gender); 5. A strong desire to be treated as the other gender (or some alternative gender different from one’s assigned gender); 6. -

Knee Joint Surgery: Open Synovectomy
Musculoskeletal Surgical Services: Open Surgical Procedures; Knee Joint Surgery: Open Synovectomy POLICY INITIATED: 06/30/2019 MOST RECENT REVIEW: 06/30/2019 POLICY # HH-5588 Overview Statement The purpose of these clinical guidelines is to assist healthcare professionals in selecting the medical service that may be appropriate and supported by evidence to improve patient outcomes. These clinical guidelines neither preempt clinical judgment of trained professionals nor advise anyone on how to practice medicine. The healthcare professionals are responsible for all clinical decisions based on their assessment. These clinical guidelines do not provide authorization, certification, explanation of benefits, or guarantee of payment, nor do they substitute for, or constitute, medical advice. Federal and State law, as well as member benefit contract language, including definitions and specific contract provisions/exclusions, take precedence over clinical guidelines and must be considered first when determining eligibility for coverage. All final determinations on coverage and payment are the responsibility of the health plan. Nothing contained within this document can be interpreted to mean otherwise. Medical information is constantly evolving, and HealthHelp reserves the right to review and update these clinical guidelines periodically. No part of this publication may be reproduced, stored in a retrieval system or transmitted, in any form or by any means, electronic, mechanical, photocopying, or otherwise, without permission from HealthHelp. -
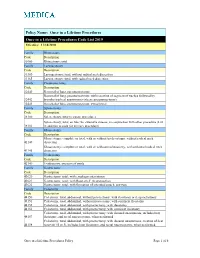
Once in a Lifetime Procedures Code List 2019 Effective: 11/14/2010
Policy Name: Once in a Lifetime Procedures Once in a Lifetime Procedures Code List 2019 Effective: 11/14/2010 Family Rhinectomy Code Description 30160 Rhinectomy; total Family Laryngectomy Code Description 31360 Laryngectomy; total, without radical neck dissection 31365 Laryngectomy; total, with radical neck dissection Family Pneumonectomy Code Description 32440 Removal of lung, pneumonectomy; Removal of lung, pneumonectomy; with resection of segment of trachea followed by 32442 broncho-tracheal anastomosis (sleeve pneumonectomy) 32445 Removal of lung, pneumonectomy; extrapleural Family Splenectomy Code Description 38100 Splenectomy; total (separate procedure) Splenectomy; total, en bloc for extensive disease, in conjunction with other procedure (List 38102 in addition to code for primary procedure) Family Glossectomy Code Description Glossectomy; complete or total, with or without tracheostomy, without radical neck 41140 dissection Glossectomy; complete or total, with or without tracheostomy, with unilateral radical neck 41145 dissection Family Uvulectomy Code Description 42140 Uvulectomy, excision of uvula Family Gastrectomy Code Description 43620 Gastrectomy, total; with esophagoenterostomy 43621 Gastrectomy, total; with Roux-en-Y reconstruction 43622 Gastrectomy, total; with formation of intestinal pouch, any type Family Colectomy Code Description 44150 Colectomy, total, abdominal, without proctectomy; with ileostomy or ileoproctostomy 44151 Colectomy, total, abdominal, without proctectomy; with continent ileostomy 44155 Colectomy,