Assessment Report
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Appendix A: Potentially Inappropriate Prescriptions (Pips) for Older People (Modified from ‘STOPP/START 2’ O’Mahony Et Al 2014)
Appendix A: Potentially Inappropriate Prescriptions (PIPs) for older people (modified from ‘STOPP/START 2’ O’Mahony et al 2014) Consider holding (or deprescribing - consult with patient): 1. Any drug prescribed without an evidence-based clinical indication 2. Any drug prescribed beyond the recommended duration, where well-defined 3. Any duplicate drug class (optimise monotherapy) Avoid hazardous combinations e.g.: 1. The Triple Whammy: NSAID + ACE/ARB + diuretic in all ≥ 65 year olds (NHS Scotland 2015) 2. Sick Day Rules drugs: Metformin or ACEi/ARB or a diuretic or NSAID in ≥ 65 year olds presenting with dehydration and/or acute kidney injury (AKI) (NHS Scotland 2015) 3. Anticholinergic Burden (ACB): Any additional medicine with anticholinergic properties when already on an Anticholinergic/antimuscarinic (listed overleaf) in > 65 year olds (risk of falls, increased anticholinergic toxicity: confusion, agitation, acute glaucoma, urinary retention, constipation). The following are known to contribute to the ACB: Amantadine Antidepressants, tricyclic: Amitriptyline, Clomipramine, Dosulepin, Doxepin, Imipramine, Nortriptyline, Trimipramine and SSRIs: Fluoxetine, Paroxetine Antihistamines, first generation (sedating): Clemastine, Chlorphenamine, Cyproheptadine, Diphenhydramine/-hydrinate, Hydroxyzine, Promethazine; also Cetirizine, Loratidine Antipsychotics: especially Clozapine, Fluphenazine, Haloperidol, Olanzepine, and phenothiazines e.g. Prochlorperazine, Trifluoperazine Baclofen Carbamazepine Disopyramide Loperamide Oxcarbazepine Pethidine -

Full Portfolio Holdings
Hartford Multifactor International Fund Full Portfolio Holdings* as of August 31, 2021 % of Security Coupon Maturity Shares/Par Market Value Net Assets Merck KGaA 0.000 152 36,115 0.982 Kuehne + Nagel International AG 0.000 96 35,085 0.954 Novo Nordisk A/S 0.000 333 33,337 0.906 Koninklijke Ahold Delhaize N.V. 0.000 938 31,646 0.860 Investor AB 0.000 1,268 30,329 0.824 Roche Holding AG 0.000 74 29,715 0.808 WM Morrison Supermarkets plc 0.000 6,781 26,972 0.733 Wesfarmers Ltd. 0.000 577 25,201 0.685 Bouygues S.A. 0.000 595 24,915 0.677 Swisscom AG 0.000 42 24,651 0.670 Loblaw Cos., Ltd. 0.000 347 24,448 0.665 Mineral Resources Ltd. 0.000 596 23,709 0.644 Royal Bank of Canada 0.000 228 23,421 0.637 Bridgestone Corp. 0.000 500 23,017 0.626 BlueScope Steel Ltd. 0.000 1,255 22,944 0.624 Yangzijiang Shipbuilding Holdings Ltd. 0.000 18,600 22,650 0.616 BCE, Inc. 0.000 427 22,270 0.605 Fortescue Metals Group Ltd. 0.000 1,440 21,953 0.597 NN Group N.V. 0.000 411 21,320 0.579 Electricite de France S.A. 0.000 1,560 21,157 0.575 Royal Mail plc 0.000 3,051 20,780 0.565 Sonic Healthcare Ltd. 0.000 643 20,357 0.553 Rio Tinto plc 0.000 271 20,050 0.545 Coloplast A/S 0.000 113 19,578 0.532 Admiral Group plc 0.000 394 19,576 0.532 Swiss Life Holding AG 0.000 37 19,285 0.524 Dexus 0.000 2,432 18,926 0.514 Kesko Oyj 0.000 457 18,910 0.514 Woolworths Group Ltd. -

The Confusions in Nordic ESG Ratings
Rating Objectivity: The Confusions in Nordic ESG Ratings ESG Ratings Subjectivity and its Consequences MASTER THESIS WITHIN: Business Administration NUMBER OF CREDITS: 30 ECTS PROGRAM OF STUDY: Civilekonom AUTHOR: John Rydholm & Samuel Schultzberg Bagge TUTOR: Fredrik Hansen & Toni Duras JÖNKÖPING May 2020 Master Thesis within Business Administration Title: Rating Objectivity: The Confusions in Nordic ESG Ratings - ESG Ratings Subjectivity and its Consequences Authors: John Rydholm, Samuel Schultzberg Bagge Tutors: Fredrik Hansen, Toni Duras Date: May 2020 Key terms: ESG, CSR, Ratings, Rating agencies, MSCI, Thomson Reuters, Sustainalytics, RobecoSAM Abstract: Environmental, Social and Governance measurements have significantly increased in usage due to growing concerns for environmental and sustainability problems in today’s world. However, with no commonly agreed-upon criteria for ESG ratings, the scoring measure creates confusion both at the investor and company level. Besides, ESG agencies have different processes and parameters for measuring ESG compliance, which contributes to the problem. The study examines four ESG rating agencies’ rating models and ESG scores to get a better understanding of deviations in ESG scores among Nordic companies. By also studying the correlation amongst ESG scores and market capitalizations in firms, the paper hopes to shed light on if any relationships exist between them. Our results show that the four major ESG raters in the study showed a weak to a non-significant correlation against each other. The maximum correlation found was 0.419 between Thomson Reuters and MSCI. RobecoSAM and MSCI showed the lowest significant correlation at 0.291. Sustainalytics was detected not to show any significant correlation with the other raters. -

The In¯Uence of Medication on Erectile Function
International Journal of Impotence Research (1997) 9, 17±26 ß 1997 Stockton Press All rights reserved 0955-9930/97 $12.00 The in¯uence of medication on erectile function W Meinhardt1, RF Kropman2, P Vermeij3, AAB Lycklama aÁ Nijeholt4 and J Zwartendijk4 1Department of Urology, Netherlands Cancer Institute/Antoni van Leeuwenhoek Hospital, Plesmanlaan 121, 1066 CX Amsterdam, The Netherlands; 2Department of Urology, Leyenburg Hospital, Leyweg 275, 2545 CH The Hague, The Netherlands; 3Pharmacy; and 4Department of Urology, Leiden University Hospital, P.O. Box 9600, 2300 RC Leiden, The Netherlands Keywords: impotence; side-effect; antipsychotic; antihypertensive; physiology; erectile function Introduction stopped their antihypertensive treatment over a ®ve year period, because of side-effects on sexual function.5 In the drug registration procedures sexual Several physiological mechanisms are involved in function is not a major issue. This means that erectile function. A negative in¯uence of prescrip- knowledge of the problem is mainly dependent on tion-drugs on these mechanisms will not always case reports and the lists from side effect registries.6±8 come to the attention of the clinician, whereas a Another way of looking at the problem is drug causing priapism will rarely escape the atten- combining available data on mechanisms of action tion. of drugs with the knowledge of the physiological When erectile function is in¯uenced in a negative mechanisms involved in erectile function. The way compensation may occur. For example, age- advantage of this approach is that remedies may related penile sensory disorders may be compen- evolve from it. sated for by extra stimulation.1 Diminished in¯ux of In this paper we will discuss the subject in the blood will lead to a slower onset of the erection, but following order: may be accepted. -

Ifu Project Portfolio 2018 2 | Ifu Project Portfolio 2018
IFU PROJECT PORTFOLIO 2018 2 | IFU PROJECT PORTFOLIO 2018 PROJECT Portfolio INDEPENDENT AUDitors’ report ON IFU PROJECT Portfolio 2018 Up to the end of 2018, IFU and IFU managed funds had co-financed To the Executive Board of IFU standards, applicable requirements in Danish law and other regulations. a total of 1,286 projects in 101 countries. Of these, 203 are ongoing As agreed, we have examined the IFU Project Portfolio 2018. The IFU We complied with independence requirements and other ethical stand- investments, while IFU has exited 1,083 projects. The distribution of Project Portfolio 2018 has been listed and presented in accordance with ards under FSR - Danish Auditors’ Code of Ethics for Professional all projects by region is illustrated in the figure below. the methodology and definitions described in “Note to IFU’s project Accountants, which rely on general principles regarding integrity, ob- portfolio – definitions (only in English)”. jectivity, professional competence and due care, confidentiality and professional conduct. TotAL NUMBER OF projects DistriButeD BY REGION We are to conclude on IFU Project Portfolio 2018 to ascertain whether the data and information are correctly reflected in accordance with the As part of our examinations, we performed the below procedures: methodology and definitions reflected in “Note to IFU project portfolio • A sample check of whether IFU’s recordings of the individual share Africa 278 –definitions”. The IFU Project Portfolio 2018 means IFU’s involvement capital investments/project loans, total disbursed and expected total Asia 422 in active and finalised projects, as presented by the Executive Board and investment are reconciled in accordance with the contractual frame- Europe 445 in accordance with the principles and definitions reflected in “Note to work and information provided. -

Medications and Alcohol Craving
Medications and Alcohol Craving Robert M. Swift, M.D., Ph.D. The use of medications as an adjunct to alcoholism treatment is based on the premise that craving and other manifestations of alcoholism are mediated by neurobiological mechanisms. Three of the four medications approved in the United States or Europe for treating alcoholism are reported to reduce craving; these include naltrexone (ReVia™), acamprosate, and tiapride. The remaining medication, disulfiram (Antabuse®), may also possess some anticraving activity. Additional medications that have been investigated include ritanserin, which has not been shown to decrease craving or drinking levels in humans, and ondansetron, which shows promise for treating early onset alcoholics, who generally respond poorly to psychosocial treatment alone. Use of anticraving medications in combination (e.g., naltrexone plus acamprosate) may enhance their effectiveness. Future studies should address such issues as optimal dosing regimens and the development of strategies to enhance patient compliance. KEY WORDS: AOD (alcohol and other drug) craving; anti alcohol craving agents; alcohol withdrawal agents; drug therapy; neurobiological theory; alcohol cue; disulfiram; naltrexone; calcium acetylhomotaurinate; dopamine; serotonin uptake inhibitors; buspirone; treatment outcome; reinforcement; neurotransmitters; patient assessment; literature review riteria for defining alcoholism Results of craving research are often tions (i.e., pharmacotherapy) to improve vary widely. Most definitions difficult to interpret, -
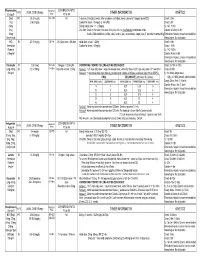
Medication Conversion Chart
Fluphenazine FREQUENCY CONVERSION RATIO ROUTE USUAL DOSE (Range) (Range) OTHER INFORMATION KINETICS Prolixin® PO to IM Oral PO 2.5-20 mg/dy QD - QID NA ↑ dose by 2.5mg/dy Q week. After symptoms controlled, slowly ↓ dose to 1-5mg/dy (dosed QD) Onset: ≤ 1hr 1mg (2-60 mg/dy) Caution for doses > 20mg/dy (↑ risk EPS) Cmax: 0.5hr 2.5mg Elderly: Initial dose = 1 - 2.5mg/dy t½: 14.7-15.3hr 5mg Oral Soln: Dilute in 2oz water, tomato or fruit juice, milk, or uncaffeinated carbonated drinks Duration of Action: 6-8hr 10mg Avoid caffeinated drinks (coffee, cola), tannics (tea), or pectinates (apple juice) 2° possible incompatibilityElimination: Hepatic to inactive metabolites 5mg/ml soln Hemodialysis: Not dialyzable HCl IM 2.5-10 mg/dy Q6-8 hr 1/3-1/2 po dose = IM dose Initial dose (usual): 1.25mg Onset: ≤ 1hr Immediate Caution for doses > 10mg/dy Cmax: 1.5-2hr Release t½: 14.7-15.3hr 2.5mg/ml Duration Action: 6-8hr Elimination: Hepatic to inactive metabolites Hemodialysis: Not dialyzable Decanoate IM 12.5-50mg Q2-3 wks 10mg po = 12.5mg IM CONVERTING FROM PO TO LONG-ACTING DECANOATE: Onset: 24-72hr (4-72hr) Long-Acting SC (12.5-100mg) (1-4 wks) Round to nearest 12.5mg Method 1: 1.25 X po daily dose = equiv decanoate dose; admin Q2-3wks. Cont ½ po daily dose X 1st few mths Cmax: 48-96hr 25mg/ml Method 2: ↑ decanoate dose over 4wks & ↓ po dose over 4-8wks as follows (accelerate taper for sx of EPS): t½: 6.8-9.6dy (single dose) ORAL DECANOATE (Administer Q 2 weeks) 15dy (14-100dy chronic administration) ORAL DOSE (mg/dy) ↓ DOSE OVER (wks) INITIAL DOSE (mg) TARGET DOSE (mg) DOSE OVER (wks) Steady State: 2mth (1.5-3mth) 5 4 6.25 6.25 0 Duration Action: 2wk (1-6wk) Elimination: Hepatic to inactive metabolites 10 4 6.25 12.5 4 Hemodialysis: Not dialyzable 20 8 6.25 12.5 4 30 8 6.25 25 4 40 8 6.25 25 4 Method 3: Admin equivalent decanoate dose Q2-3wks. -

Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients
© Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 | Fax 503-947-1119 Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients Research Questions: 1. How many schizophrenia patients are prescribed recommended first-line second-generation treatments for schizophrenia? 2. How many schizophrenia patients switch to an injectable antipsychotic after stabilization on an oral antipsychotic? 3. How many schizophrenia patients are prescribed 2 or more concomitant antipsychotics? 4. Are claims for long-acting injectable antipsychotics primarily billed as pharmacy or physician administered claims? 5. Does adherence to antipsychotic therapy differ between patients with claims for different routes of administration (oral vs. long-acting injectable)? Conclusions: In total, 4663 schizophrenia patients met inclusion criteria, and approximately 14% of patients (n=685) were identified as treatment naïve without claims for antipsychotics in the year before their first antipsychotic prescription. Approximately 45% of patients identified as treatment naïve had a history of remote antipsychotic use, but it is unclear if antipsychotics were historically prescribed for schizophrenia. Oral second-generation antipsychotics which are recommended as first-line treatment in the MHCAG schizophrenia algorithm were prescribed as initial treatment in 37% of treatment naive patients and 28% of all schizophrenia patients. Recommended agents include risperidone, paliperidone, and aripiprazole. Utilization of parenteral antipsychotics was limited in patients with schizophrenia. Overall only 8% of patients switched from an oral to an injectable therapy within 6 months of their first claim. Approximately, 60% of all schizophrenia patients (n=2512) had claims for a single antipsychotic for at least 12 continuous weeks and may be eligible to transition to a long-acting injectable antipsychotic. -

Schizophrenia Care Guide
August 2015 CCHCS/DHCS Care Guide: Schizophrenia SUMMARY DECISION SUPPORT PATIENT EDUCATION/SELF MANAGEMENT GOALS ALERTS Minimize frequency and severity of psychotic episodes Suicidal ideation or gestures Encourage medication adherence Abnormal movements Manage medication side effects Delusions Monitor as clinically appropriate Neuroleptic Malignant Syndrome Danger to self or others DIAGNOSTIC CRITERIA/EVALUATION (PER DSM V) 1. Rule out delirium or other medical illnesses mimicking schizophrenia (see page 5), medications or drugs of abuse causing psychosis (see page 6), other mental illness causes of psychosis, e.g., Bipolar Mania or Depression, Major Depression, PTSD, borderline personality disorder (see page 4). Ideas in patients (even odd ideas) that we disagree with can be learned and are therefore not necessarily signs of schizophrenia. Schizophrenia is a world-wide phenomenon that can occur in cultures with widely differing ideas. 2. Diagnosis is made based on the following: (Criteria A and B must be met) A. Two of the following symptoms/signs must be present over much of at least one month (unless treated), with a significant impact on social or occupational functioning, over at least a 6-month period of time: Delusions, Hallucinations, Disorganized Speech, Negative symptoms (social withdrawal, poverty of thought, etc.), severely disorganized or catatonic behavior. B. At least one of the symptoms/signs should be Delusions, Hallucinations, or Disorganized Speech. TREATMENT OPTIONS MEDICATIONS Informed consent for psychotropic -

Prozac and Prozac Weekly (Fluoxetine Hcl)
CENTER FOR DRUG EVALUATION AND RESEARCH Approval Package for: APPLICATION NUMBER: NDA 18-936/S-093 NDA 21-235/S-016 Trade Name: Prozac and Prozac Weekly Generic Name: fluoxetine HCl Sponsor: Lilly Approval Date: April 4, 2011 ,QGLFDWLRQ Revisions to the following section 8.1 of the label. CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: NDA 18-936/S-093 NDA 21-235/S-016 CONTENTS Reviews / Information Included in this NDA Review. Approval Letter X Other Action Letters X Labeling Summary Review X Officer/Employee List Office Director Memo Cross Discipline Team Leader Review Medical Review(s) X Chemistry Review(s) Pharmacology Review(s) Statistical Review(s) Clinical Pharmacology/Biopharmaceutics Review(s) Other Reviews X Proprietary Name Review(s) Administrative/Correspondence Document(s) X CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: NDA 18-936/S-093 NDA 21-235/S-016 APPROVAL LETTER DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration Silver Spring MD 20993 NDA 018936/S-091/S-093/S-095 NDA 021235/S-015/S-016/S-017 SUPPLEMENT APPROVAL Eli Lilly & Company Attention: Kevin C. Sheehan, MS, Pharm.D. Manager, Global Regulatory Affairs - US Lilly Corporate Center Indianapolis, IN 46285 Dear Dr. Sheehan: Please refer to your Supplemental New Drug Applications (sNDA) dated and received May 21, 2009 (018936/S-091 and 021235/S-015), November 6, 2009 (018936/S-093 and 021235/S-016), and April 14, 2010 (018936/S-095 and 021235/S-017), submitted under section 505(b) of the Federal Food, Drug, and Cosmetic Act (FDCA) for Prozac (fluoxetine hydrochloride) 10 mg, 20 mg, and 40 mg capsules and Prozac Weekly (fluoxetine hydrochloride) 90 mg delayed-release capsules. -

Management of Major Depressive Disorder Clinical Practice Guidelines May 2014
Federal Bureau of Prisons Management of Major Depressive Disorder Clinical Practice Guidelines May 2014 Table of Contents 1. Purpose ............................................................................................................................................. 1 2. Introduction ...................................................................................................................................... 1 Natural History ................................................................................................................................. 2 Special Considerations ...................................................................................................................... 2 3. Screening ........................................................................................................................................... 3 Screening Questions .......................................................................................................................... 3 Further Screening Methods................................................................................................................ 4 4. Diagnosis ........................................................................................................................................... 4 Depression: Three Levels of Severity ............................................................................................... 4 Clinical Interview and Documentation of Risk Assessment............................................................... -

Appendix 18: References to Studies from the Previous Guideline
Appendix 18: References to studies from the previous guideline This appendix contains references to the studies from the previous guideline, as they appeared in that guideline. References to studies added to the guideline update are in Appendix 17a-d. Contents Service references…………………………………………………………………..1 Psychology references……………………………………………………………..15 Pharmacology references………………………………………………………….28 Service references Araya2003 {published data only} Araya R, Rojas G, Fritsch R, Gaete J, Rojas M, Simon G, Peters TJ. Treating depression in primary care in low-income women in Santiago, Chile: A randomised controlled trial. Lancet 2003;361(9362):995-1000. Arthur2002 {published data only} Arthur AJ, Jagger C, Lindesay J, Matthews RJ. Evaluating a mental health assessment for older people with depressive symptoms in general practice: a randomised controlled trial. British Journal of General Practice 2002;52(476):202-207. Austin-Los Angeles Austin NK, Liberman RP, King LW, DeRisi WJ. A comparative evaluation of two day hospitals. Goal attainment scaling of behaviour therapy vs. milieu therapy. Journal of Nervous and Mental Disease 1976;163:253-62. Azim-Alberta Azim HF, Weiden TD, Ratcliffe WD, Nutter RW, Dyck RJ, Howarth BG. Current utilization of day hospitalization. Canadian Psychiatric Association Journal 1978;23:557-66 Baker2001 {published data only} Baker R, Reddish S, Robertson N, Hearnshaw H, Jones B. Randomised controlled trial of tailored strategies to implement guidelines for the management of patients with depression in general practice. British Journal of General Practice 2001;51:737-741. Barkley-Ontario Barkley AL, Fagen K, Lawson JS. Day care: can it prevent readmission to a psychiatric hospital? Psychiatric Journal of the University of Ottawa 1989;14:536-41.