Impact of Pharmacogenetic Polymorphisms
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The In¯Uence of Medication on Erectile Function
International Journal of Impotence Research (1997) 9, 17±26 ß 1997 Stockton Press All rights reserved 0955-9930/97 $12.00 The in¯uence of medication on erectile function W Meinhardt1, RF Kropman2, P Vermeij3, AAB Lycklama aÁ Nijeholt4 and J Zwartendijk4 1Department of Urology, Netherlands Cancer Institute/Antoni van Leeuwenhoek Hospital, Plesmanlaan 121, 1066 CX Amsterdam, The Netherlands; 2Department of Urology, Leyenburg Hospital, Leyweg 275, 2545 CH The Hague, The Netherlands; 3Pharmacy; and 4Department of Urology, Leiden University Hospital, P.O. Box 9600, 2300 RC Leiden, The Netherlands Keywords: impotence; side-effect; antipsychotic; antihypertensive; physiology; erectile function Introduction stopped their antihypertensive treatment over a ®ve year period, because of side-effects on sexual function.5 In the drug registration procedures sexual Several physiological mechanisms are involved in function is not a major issue. This means that erectile function. A negative in¯uence of prescrip- knowledge of the problem is mainly dependent on tion-drugs on these mechanisms will not always case reports and the lists from side effect registries.6±8 come to the attention of the clinician, whereas a Another way of looking at the problem is drug causing priapism will rarely escape the atten- combining available data on mechanisms of action tion. of drugs with the knowledge of the physiological When erectile function is in¯uenced in a negative mechanisms involved in erectile function. The way compensation may occur. For example, age- advantage of this approach is that remedies may related penile sensory disorders may be compen- evolve from it. sated for by extra stimulation.1 Diminished in¯ux of In this paper we will discuss the subject in the blood will lead to a slower onset of the erection, but following order: may be accepted. -

)&F1y3x PHARMACEUTICAL APPENDIX to THE
)&f1y3X PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE )&f1y3X PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 3 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABAMECTIN 65195-55-3 ACTODIGIN 36983-69-4 ABANOQUIL 90402-40-7 ADAFENOXATE 82168-26-1 ABCIXIMAB 143653-53-6 ADAMEXINE 54785-02-3 ABECARNIL 111841-85-1 ADAPALENE 106685-40-9 ABITESARTAN 137882-98-5 ADAPROLOL 101479-70-3 ABLUKAST 96566-25-5 ADATANSERIN 127266-56-2 ABUNIDAZOLE 91017-58-2 ADEFOVIR 106941-25-7 ACADESINE 2627-69-2 ADELMIDROL 1675-66-7 ACAMPROSATE 77337-76-9 ADEMETIONINE 17176-17-9 ACAPRAZINE 55485-20-6 ADENOSINE PHOSPHATE 61-19-8 ACARBOSE 56180-94-0 ADIBENDAN 100510-33-6 ACEBROCHOL 514-50-1 ADICILLIN 525-94-0 ACEBURIC ACID 26976-72-7 ADIMOLOL 78459-19-5 ACEBUTOLOL 37517-30-9 ADINAZOLAM 37115-32-5 ACECAINIDE 32795-44-1 ADIPHENINE 64-95-9 ACECARBROMAL 77-66-7 ADIPIODONE 606-17-7 ACECLIDINE 827-61-2 ADITEREN 56066-19-4 ACECLOFENAC 89796-99-6 ADITOPRIM 56066-63-8 ACEDAPSONE 77-46-3 ADOSOPINE 88124-26-9 ACEDIASULFONE SODIUM 127-60-6 ADOZELESIN 110314-48-2 ACEDOBEN 556-08-1 ADRAFINIL 63547-13-7 ACEFLURANOL 80595-73-9 ADRENALONE -

(12) United States Patent (10) Patent No.: US 8,080,578 B2 Liggett Et Al
USO08080578B2 (12) United States Patent (10) Patent No.: US 8,080,578 B2 Liggett et al. (45) Date of Patent: *Dec. 20, 2011 (54) METHODS FOR TREATMENT WITH 5,998.458. A 12/1999 Bristow ........................ 514,392 BUCNDOLOL BASED ON GENETIC 6,004,744. A 12/1999 Goelet et al. ...... 435/5 6,013,431 A 1/2000 Söderlund et al. 435/5 TARGETING 6,156,503 A 12/2000 Drazen et al. ..... ... 435/6 6,221,851 B1 4/2001 Feldman ... 51446 (75) Inventors: Stephen B. Liggett, Clarksville, MD 6,316,188 B1 1 1/2001 Yan et al. .......................... 435/6 6,365,618 B1 4/2002 Swartz ... 514,411 (US); Michael Bristow, Englewood, CO 6,498,009 B1 12/2002 Liggett ............................. 435/6 (US) 6,566,101 B1 5/2003 Shuber et al. 435,912 6,586,183 B2 7/2003 Drysdale et al. .................. 435/6 (73) Assignee: The Regents of the University of 6,784, 177 B2 8/2004 Cohn et al. 514,248 Colorado, a body corporate, Denver, 6,797.472 B1 9/2004 Liggett ......... ... 435/6 6,821,724 B1 1 1/2004 Mittman et al. ... 435/6 CO (US) 6,861.217 B1 3/2005 Liggett ......... ... 435/6 7,041,810 B2 5/2006 Small et al. ... ... 435/6 (*) Notice: Subject to any disclaimer, the term of this 7, 195,873 B2 3/2007 Fligheddu et al. ... 435/6 patent is extended or adjusted under 35 7,211,386 B2 5/2007 Small et al. ....... ... 435/6 7,229,756 B1 6/2007 Small et al. -

Long Acting, Reversible Veterinary Sedative and Analgesic And
University of Kentucky UKnowledge Veterinary Science Faculty Patents Veterinary Science 7-11-2006 Long Acting, Reversible Veterinary Sedative and Analgesic and Method of Use Thomas Tobin University of Kentucky, [email protected] Right click to open a feedback form in a new tab to let us know how this document benefits oy u. Follow this and additional works at: https://uknowledge.uky.edu/gluck_patents Part of the Veterinary Medicine Commons Recommended Citation Tobin, Thomas, "Long Acting, Reversible Veterinary Sedative and Analgesic and Method of Use" (2006). Veterinary Science Faculty Patents. 14. https://uknowledge.uky.edu/gluck_patents/14 This Patent is brought to you for free and open access by the Veterinary Science at UKnowledge. It has been accepted for inclusion in Veterinary Science Faculty Patents by an authorized administrator of UKnowledge. For more information, please contact [email protected]. US007074834B2 (12) United States Patent (10) Patent N0.: US 7,074,834 B2 Tobin (45) Date of Patent: Jul. 11, 2006 (54) LONG ACTING, REVERSIBLE VETERINARY 4,742,054 A 5/1988 Naftchi SEDATIVE AND ANALGESIC AND METHOD 4,950,648 A 8/1990 Raddatz et a1. OF USE 5,635,204 A * 6/1997 GevirtZ et a1. ............ .. 424/449 5,942,241 A 8/1999 Chasin et a1. (75) Inventor: Thomas Tobin, Lexington, KY (US) 5,958,933 A 9/1999 Naftchi (73) Assignee: University of Kentucky Foundation, OTHER PUBLICATIONS Lexington, KY (US) MEDLINE AN 20000025586, Veveris et al, Brit. J. Pharmacol, 128 (5), 1089-97, Nov. 1999, abstract.* ( * ) Notice: Subject to any disclaimer, the term of this Veterinary Pharmacology and Therapeutics, Adams, pp. -

Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DIX to the HTSUS—Continued
20558 Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DEPARMENT OF THE TREASURY Services, U.S. Customs Service, 1301 TABLE 1.ÐPHARMACEUTICAL APPEN- Constitution Avenue NW, Washington, DIX TO THE HTSUSÐContinued Customs Service D.C. 20229 at (202) 927±1060. CAS No. Pharmaceutical [T.D. 95±33] Dated: April 14, 1995. 52±78±8 ..................... NORETHANDROLONE. A. W. Tennant, 52±86±8 ..................... HALOPERIDOL. Pharmaceutical Tables 1 and 3 of the Director, Office of Laboratories and Scientific 52±88±0 ..................... ATROPINE METHONITRATE. HTSUS 52±90±4 ..................... CYSTEINE. Services. 53±03±2 ..................... PREDNISONE. 53±06±5 ..................... CORTISONE. AGENCY: Customs Service, Department TABLE 1.ÐPHARMACEUTICAL 53±10±1 ..................... HYDROXYDIONE SODIUM SUCCI- of the Treasury. NATE. APPENDIX TO THE HTSUS 53±16±7 ..................... ESTRONE. ACTION: Listing of the products found in 53±18±9 ..................... BIETASERPINE. Table 1 and Table 3 of the CAS No. Pharmaceutical 53±19±0 ..................... MITOTANE. 53±31±6 ..................... MEDIBAZINE. Pharmaceutical Appendix to the N/A ............................. ACTAGARDIN. 53±33±8 ..................... PARAMETHASONE. Harmonized Tariff Schedule of the N/A ............................. ARDACIN. 53±34±9 ..................... FLUPREDNISOLONE. N/A ............................. BICIROMAB. 53±39±4 ..................... OXANDROLONE. United States of America in Chemical N/A ............................. CELUCLORAL. 53±43±0 -

Table of Antihypertensive Drugs
Chapter 14 Table of Antihypertensive Drugs F. GROSS and J. B. LUTH The following tables give information about the most important drugs used today in the treatment of high blood pressure. These lists do not in any way aim for completeness, in particular with respect to brand names, which are much more numerous for several of the mentioned products. The usual forms of adminis tration and average dosages are included, but various other schemes are also recommended for some drugs in some countries. What is said for the single entities with respect to completeness holds true even more for the fixed combinations, of which only a fraction of those marketed in a number or'countries has been included. Some examples of the combinations that make sense to the authors have been mentioned; many have been omitted; however, deletion does not mean in all cases - but it does in some - that the combi nation is considered useless. If a barbiturate is added to reserpine and a diuretic, it is difficult to ascribe to the new combination an improved effect on the high blood pressure. Quite a few more examples could be given, where the addition of a further ingredient to the basic combination of reserpine and a diuretic is nothing but the mixing in of a special flavor, and not of an antihypertensive component. Some of the readers may also feel that too many drugs and combinations have been listed, and they may never have heard the names of quite a few of them. Despite the incompleteness of the list, it gives an impression of the huge variety of pre parations, which will further increase with the growing number of /J-adrenergic blockers and their combinations with other drugs. -

Stembook 2018.Pdf
The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 WHO/EMP/RHT/TSN/2018.1 © World Health Organization 2018 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances. Geneva: World Health Organization; 2018 (WHO/EMP/RHT/TSN/2018.1). Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. -
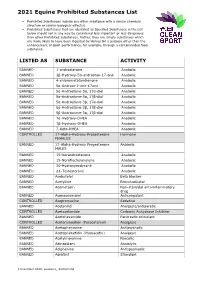
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). -

Rubidomycin in Acute Leukaemia Foods
3 June 1967 Leading Articles BRnITH 587 sympathetic efferent function should be given. These com- pathetic blocking agents will give better results than a single prise the ganglion-blocking agents, of which only mecamyl- one.'4 Good results depend to a great extent on the care with amine and pempidine are now used in any quantity, methyl- which the regimen is managed: this is usually more impor- Br Med J: first published as 10.1136/bmj.2.5552.587 on 3 June 1967. Downloaded from dopa, and the drugs which block sympathetic fibres at the tant than the initial choice of drug. neuro-effector junction-bretylium, guanethidine, bethan- idine, guanoxan, guanoclor, and debrisoquine. Pargyline has an effect similar to these drugs on arterial pressure,' but should not usually be prescribed for the maintenance treat- ment of hypertensive patients because it is a monoamine- oxidase inhibitor, and patients receiving drugs of this type may suffer dangerous reactions after some other drugs or Rubidomycin in Acute Leukaemia foods. The ganglion-blocking drugs affect parasympathetic as well as sympathetic functions and thus may cause addi- Rubidomycin is the first antibiotic to show therapeutic effect tional side-effects. Few people would now consider them the in management of acute leukaemia in man. It is produced first choice for the treatment of severe hypertension, though from a strain of Streptomyces coeruleorubidus, and it is active they may be very useful if other drugs fail. Bretylium has in all forms of acute leukaemia. J. Bernard and his now fallen into disfavour because many patients become colleagues,' in an extensive clinical trial, have found it useful tolerant of its hypotensive effects. -

Isolation of an Endogenous Clonidine-Displacing Substance from Rat Brain
Volume 170, number 2 FEBS 1474 May 1984 Isolation of an endogenous clonidine-displacing substance from rat brain Daphne Atlas and Yigal Burstein* Department of Biological Chemistry, Institute of Life Sciences, The Hebrew University of Jerusalem, 91904 Jerusalem and *Department of Organic Chemistry, The Weizmann Institute of Science, 76100 Rehovot, Israel Received 9 April 1984 An endogenous substance which specifically displaces clonidine, yohimbine and rauwolscine from rat brain cyz-adrenergic receptors, has been isolated. The new compound, designed clonidine-displacing- substance (CDS), has been partially purified by ion exchange chromatography, zone electrophoresis and high performance liquid chromatography (HPLC). CDS binds specifically to cYz-adrenergic receptors by competing with either cuz-adrenergic agonists or cYz-antagonists, but has no effect on the specific binding of [3H]prazosin to cYr-adrenergic receptors in rat brain membranes. In the course of isolation, CDS was shown to be neither the endogenous neurotransmitter (- )norepinephrine (NE) nor the guanyl nucleotide GTP which lowers the specific binding of cYz-agonists to the cus-adrenergic receptors. Clonidine cYrAdrenergic receptor Yohimbine Hypertension 1. INTRODUCTION oblongata [lo], prompted us to look for an en- dogenous clonidine-like substance in the brain. The wide-spread interest in m-adrenergic agonists is mostly due to their clinical application as antihypertensive drugs. Clonidine, guanabenz, 2. EXPERIMENTAL guanadrel, labaz, guanoxan and tramazoline are but a few representatives of numerous imidazoline 2.1. Chemicals and guanido-containing az-agonists (review [3H]Clonidine (25.5 Ci/mmol; Ci = 3.7 x 10” [l-4]). Their potent antihypertensive activity becquerels), [3H]prazosin (40 Ci/mmol), [3H]yo- results from stimulation of cuz-adrenergic receptors himbine (80 Ci/mmol), [3H]NE (24.6 Ci/mmol) in the central nervous system (CNS). -

Caco-2: Predicting Drug Permeability and Relationship with COVID-19 Infected Caco-2 Cells Clifford Fong
Caco-2: Predicting drug permeability and relationship with COVID-19 infected Caco-2 cells Clifford Fong To cite this version: Clifford Fong. Caco-2: Predicting drug permeability and relationship with COVID-19 infected Caco-2 cells. [Research Report] Eigenenergy Adelaide South Australia. 2021. hal-03147823 HAL Id: hal-03147823 https://hal.archives-ouvertes.fr/hal-03147823 Submitted on 21 Feb 2021 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Caco-2: Predicting drug permeability and relationship with COVID-19 infected Caco-2 cells Clifford W. Fong Eigenenergy, Adelaide, South Australia, Australia. Email: [email protected] Keywords: Caco-2, drug cellular permeability, passive transcellular and paracellular diffusion, active transport, COVID-2019 or SARS-CoV-2; ACE2 receptor binding, spike serine proteases, pro S-RBD, TMPRSS2, M , IC50, host cell membrane fusion or endocytosis, translatome and proteome proteomics, linear free energy relationships, HOMO-LUMO; quantum mechanics; Abbreviations: Structure activity relationships SAR, ΔGdesolv,CDS -

Serotonin Transporter Gene and Treatment Of
(19) TZZ __T (11) EP 2 255 184 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: G01N 33/48 (2006.01) C12Q 1/68 (2006.01) 24.04.2013 Bulletin 2013/17 G01N 33/68 (2006.01) G01N 33/94 (2006.01) A61K 45/06 (2006.01) (21) Application number: 09714591.6 (86) International application number: (22) Date of filing: 27.02.2009 PCT/US2009/035420 (87) International publication number: WO 2009/108837 (03.09.2009 Gazette 2009/36) (54) SEROTONIN TRANSPORTER GENE AND TREATMENT OF ALCOHOLISM SEROTONINTRANSPORTERGEN UND BEHANDLUNG VON ALKOHOLSUCHT GÈNE DU TRANSPORTEUR DE LA SÉROTONINE ET TRAITEMENT DE L’ALCOOLISME (84) Designated Contracting States: • MANNELLI PAOLO ET AL: "Polymorphism in the AT BE BG CH CY CZ DE DK EE ES FI FR GB GR serotonin transporter gene and response to HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL treatment in African American cocaine and PT RO SE SI SK TR alcohol-abusing individuals", ADDICTION BIOLOGY, ABINGDON CARFAX PUBLISHING, (30) Priority: 28.02.2008 US 32263 P ABINGDON, GB, vol. 10, no. 3, 1 September 2005 06.06.2008 US 59301 P (2005-09-01), pages261-268, XP002595912, ISSN: 22.01.2009 US 146440 P 1355-6215, DOI: DOI: 10.1080/13556210500235540 [retrieved on (43) Date of publication of application: 2006-06-09] 01.12.2010 Bulletin 2010/48 • MARY-ANNE ENOCH ET AL: "Genetics of Alcoholism Using Intermediate Phenotypes", (73) Proprietor: UNIVERSITY OF VIRGINIA PATENT ALCOHOLISM: CLINICAL & EXPERIMENTAL FOUNDATION RESEARCH, vol.