Edna Captures Microhabitat Partitioning in a Kelp Forest Ecosystem
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genomics and Mapping of Teleostei (Bony fish)
Comparative and Functional Genomics Comp Funct Genom 2003; 4: 182–193. Published online 1 April 2003 in Wiley InterScience (www.interscience.wiley.com). DOI: 10.1002/cfg.259 Featured Organism Genomics and mapping of Teleostei (bony fish) Melody S. Clark* HGMP Resource Centre, Genome Campus, Hinxton, Cambridge CB2 4PP, UK *Correspondence to: Abstract Melody S. Clark, HGMP Resource Centre, Genome Until recently, the Human Genome Project held centre stage in the press releases Campus, Hinxton, Cambridge concerning sequencing programmes. However, in October 2001, it was announced CB2 4PP, UK. that the Japanese puffer fish (Takifugu rubripes, Fugu) was the second vertebrate E-mail: [email protected] organism to be sequenced to draft quality. Briefly, the spotlight was on fish genomes. There are currently two other fish species undergoing intensive sequencing, the green spotted puffer fish (Tetraodon nigroviridis) and the zebrafish (Danio rerio). But this trio are, in many ways, atypical representations of the current state of fish genomic research. The aim of this brief review is to demonstrate the complexity of fish as a Received: 10 November 2002 group of vertebrates and to publicize the ‘lesser-known’ species, all of which have Revised: 5 December 2002 something to offer. Copyright 2003 John Wiley & Sons, Ltd. Accepted: 28 January 2003 Keywords: Teleostei; fish; genomics; BACs; sequencing; aquaculture Background the wild-caught fisheries production figures. The equivalents within the EU are trout, Atlantic Fish have the potential to be immensely useful salmon and sea bass/bream for aquaculture; model organisms in medical research, as evidenced with herring, mackerel and sprat for wild-caught by the genomic sequencing programmes mentioned fisheries. -

Environmental DNA Reveals the Fine-Grained and Hierarchical
www.nature.com/scientificreports OPEN Environmental DNA reveals the fne‑grained and hierarchical spatial structure of kelp forest fsh communities Thomas Lamy 1,2*, Kathleen J. Pitz 3, Francisco P. Chavez3, Christie E. Yorke1 & Robert J. Miller1 Biodiversity is changing at an accelerating rate at both local and regional scales. Beta diversity, which quantifes species turnover between these two scales, is emerging as a key driver of ecosystem function that can inform spatial conservation. Yet measuring biodiversity remains a major challenge, especially in aquatic ecosystems. Decoding environmental DNA (eDNA) left behind by organisms ofers the possibility of detecting species sans direct observation, a Rosetta Stone for biodiversity. While eDNA has proven useful to illuminate diversity in aquatic ecosystems, its utility for measuring beta diversity over spatial scales small enough to be relevant to conservation purposes is poorly known. Here we tested how eDNA performs relative to underwater visual census (UVC) to evaluate beta diversity of marine communities. We paired UVC with 12S eDNA metabarcoding and used a spatially structured hierarchical sampling design to assess key spatial metrics of fsh communities on temperate rocky reefs in southern California. eDNA provided a more‑detailed picture of the main sources of spatial variation in both taxonomic richness and community turnover, which primarily arose due to strong species fltering within and among rocky reefs. As expected, eDNA detected more taxa at the regional scale (69 vs. 38) which accumulated quickly with space and plateaued at only ~ 11 samples. Conversely, the discovery rate of new taxa was slower with no sign of saturation for UVC. -

A Preliminary Global Assessment of the Status of Exploited Marine Fish and Invertebrate Populations
A PRELIMINARY GLOBAL ASSESSMENT OF THE STATUS OF EXPLOITED MARINE FISH AND INVERTEBRATE POPULATIONS June 30 2018 A PRELIMINARY GLOBAL ASSESSMENT OF THE STATUS OF EXPLOITED MARINE FISH AND INVERTEBRATE POPULATIONS Maria. L.D. Palomares, Rainer Froese, Brittany Derrick, Simon-Luc Nöel, Gordon Tsui Jessika Woroniak Daniel Pauly A report prepared by the Sea Around Us for OCEANA June 30, 2018 A PRELIMINARY GLOBAL ASSESSMENT OF THE STATUS OF EXPLOITED MARINE FISH AND INVERTEBRATE POPULATIONS Maria L.D. Palomares1, Rainer Froese2, Brittany Derrick1, Simon-Luc Nöel1, Gordon Tsui1, Jessika Woroniak1 and Daniel Pauly1 CITE AS: Palomares MLD, Froese R, Derrick B, Nöel S-L, Tsui G, Woroniak J, Pauly D (2018) A preliminary global assessment of the status of exploited marine fish and invertebrate populations. A report prepared by the Sea Around Us for OCEANA. The University of British Columbia, Vancouver, p. 64. 1 Sea Around Us, Institute for the Oceans and Fisheries, University of British Columbia, 2202 Main Mall, Vancouver BC V6T1Z4 Canada 2 Helmholtz Centre for Ocean Research GEOMAR, Düsternbrooker Weg 20, 24105 Kiel, Germany TABLE OF CONTENTS Executive Summary 1 Introduction 2 Material and Methods 3 − Reconstructed catches vs official catches 3 − Marine Ecoregions vs EEZs 3 − The CMSY method 5 Results and Discussion 7 − Stock summaries reports 9 − Problematic stocks and sources of bias 14 − Stocks in the countries where OCEANA operates 22 − Stock assessments on the Sea Around Us website 31 − The next steps 32 Acknowledgements 33 References 34 Appendices I. List of marine ecoregions by EEZ 37 II. Summaries of number of stock by region and 49 by continent III. -

2019 Annual Marine Environmental Analysis and Interpretation Report
2019 ANNUAL MARINE ENVIRONMENTAL ANALYSIS AND INTERPRETATION San Onofre Nuclear Generating Station ANNUAL MARINE ENVIRONMENTAL ANALYSIS AND INTERPRETATION San Onofre Nuclear Generating Station July 2020 Page intentionally blank Report Preparation/Data Collection – Oceanography and Marine Biology MBC Aquatic Sciences 3000 Red Hill Avenue Costa Mesa, CA 92626 Page intentionally blank TABLE OF CONTENTS LIST OF FIGURES ........................................................................................................................... iii LIST OF TABLES ...............................................................................................................................v LIST OF APPENDICES ................................................................................................................... vi EXECUTIVE SUMMARY .............................................................................................................. vii CHAPTER 1 STUDY INTRODUCTION AND GENERATING STATION DESCRIPTION 1-1 INTRODUCTION ................................................................................................................... 1-1 PURPOSE OF SAMPLING ......................................................................................... 1-1 REPORT APPROACH AND ORGANIZATION ........................................................ 1-1 DESCRIPTION OF THE STUDY AREA ................................................................... 1-1 HISTORICAL BACKGROUND ........................................................................................... -

A List of Common and Scientific Names of Fishes from the United States And
t a AMERICAN FISHERIES SOCIETY QL 614 .A43 V.2 .A 4-3 AMERICAN FISHERIES SOCIETY Special Publication No. 2 A List of Common and Scientific Names of Fishes -^ ru from the United States m CD and Canada (SECOND EDITION) A/^Ssrf>* '-^\ —---^ Report of the Committee on Names of Fishes, Presented at the Ei^ty-ninth Annual Meeting, Clearwater, Florida, September 16-18, 1959 Reeve M. Bailey, Chairman Ernest A. Lachner, C. C. Lindsey, C. Richard Robins Phil M. Roedel, W. B. Scott, Loren P. Woods Ann Arbor, Michigan • 1960 Copies of this publication may be purchased for $1.00 each (paper cover) or $2.00 (cloth cover). Orders, accompanied by remittance payable to the American Fisheries Society, should be addressed to E. A. Seaman, Secretary-Treasurer, American Fisheries Society, Box 483, McLean, Virginia. Copyright 1960 American Fisheries Society Printed by Waverly Press, Inc. Baltimore, Maryland lutroduction This second list of the names of fishes of The shore fishes from Greenland, eastern the United States and Canada is not sim- Canada and the United States, and the ply a reprinting with corrections, but con- northern Gulf of Mexico to the mouth of stitutes a major revision and enlargement. the Rio Grande are included, but those The earlier list, published in 1948 as Special from Iceland, Bermuda, the Bahamas, Cuba Publication No. 1 of the American Fisheries and the other West Indian islands, and Society, has been widely used and has Mexico are excluded unless they occur also contributed substantially toward its goal of in the region covered. In the Pacific, the achieving uniformity and avoiding confusion area treated includes that part of the conti- in nomenclature. -

Humboldt Bay Fishes
Humboldt Bay Fishes ><((((º>`·._ .·´¯`·. _ .·´¯`·. ><((((º> ·´¯`·._.·´¯`·.. ><((((º>`·._ .·´¯`·. _ .·´¯`·. ><((((º> Acknowledgements The Humboldt Bay Harbor District would like to offer our sincere thanks and appreciation to the authors and photographers who have allowed us to use their work in this report. Photography and Illustrations We would like to thank the photographers and illustrators who have so graciously donated the use of their images for this publication. Andrey Dolgor Dan Gotshall Polar Research Institute of Marine Sea Challengers, Inc. Fisheries And Oceanography [email protected] [email protected] Michael Lanboeuf Milton Love [email protected] Marine Science Institute [email protected] Stephen Metherell Jacques Moreau [email protected] [email protected] Bernd Ueberschaer Clinton Bauder [email protected] [email protected] Fish descriptions contained in this report are from: Froese, R. and Pauly, D. Editors. 2003 FishBase. Worldwide Web electronic publication. http://www.fishbase.org/ 13 August 2003 Photographer Fish Photographer Bauder, Clinton wolf-eel Gotshall, Daniel W scalyhead sculpin Bauder, Clinton blackeye goby Gotshall, Daniel W speckled sanddab Bauder, Clinton spotted cusk-eel Gotshall, Daniel W. bocaccio Bauder, Clinton tube-snout Gotshall, Daniel W. brown rockfish Gotshall, Daniel W. yellowtail rockfish Flescher, Don american shad Gotshall, Daniel W. dover sole Flescher, Don stripped bass Gotshall, Daniel W. pacific sanddab Gotshall, Daniel W. kelp greenling Garcia-Franco, Mauricio louvar -

CALIFORNIA FISH and GAME “Journal for Conservation and Management of California’S Species and Ecosystems”
CALIFORNIA FISH AND GAME “Journal for Conservation and Management of California’s Species and Ecosystems” Volume 105 Fall 2019 Number 4 Lorraine Elrod © California Academy of Sciences Published Quarterly by the California Department of Fish and Wildlife STATE OF CALIFORNIA Gavin Newsom, Governor CALIFORNIA NATURAL RESOURCES AGENCY Wade Crowfoot, Secretary for Natural Resources FISH AND GAME COMMISSION Eric Sklar, President Jacque Hostler-Carmesin, Vice President Russell Burns, Member Peter S. Silva, Member Samantha Murray, Member Melissa Miller-Henson, Acting Executive Director DEPARTMENT OF FISH AND WILDLIFE Charlton “Chuck” Bonham, Director CALIFORNIA FISH AND GAME EDITORIAL STAFF Ange Darnell Baker ...........................................................................Editor-in-Chief Lorna Bernard ...........................Office of Communication, Education and Outreach Neil Clipperton, Scott Osborn, Laura Patterson, Joel Trumbo, Dan Skalos, and Karen Converse .................................................... Wildlife Branch Felipe La Luz ...................................................................................... Water Branch Jeff Rodzen, Jeff Weaver, and Ken Kundargi ................................. Fisheries Branch Cherilyn Burton ........................................... Habitat Conservation Planning Branch Kevin Fleming ...............................................Watershed Restoration Grants Branch Jeff Villepique, Steve Parmenter ............................................ Inland Deserts Region Paul Reilly, -

Sustainable Exploitation of Small Pelagic Fish Stocks Challenged by Environmental and Ecosystem Changes: a Review
BULLETIN OF MARINE SCIENCE, 76(2): 385–462, 2005 SUSTAINABLE EXPLOITATION OF SMALL PELAGIC FISH STOCKS CHALLENGED BY ENVIRONMENTAL AND ECOSYSTEM CHANGES: A REVIEW Pierre Fréon, Philippe Cury, Lynne Shannon, and Claude Roy ABSTRACT Small pelagic fish contribute up to 50% of the total landing of marine species. They are most abundant in upwelling areas and contribute to food security. Exploit- ed stocks of these species are prone to large interannual and interdecadal variation of abundance as well as to collapse. We discuss why small pelagic fish and fisheries are so “special” with regard to their biology, ecology, and behavior. Two adjectives can sum up the characteristics of pelagic species: variability and instability. Analy- ses of the relationships between small pelagic fish and their physical environment at different time-scales illustrate the complexity of the interplay between exploita- tion and environmental impacts. How small pelagic fish species are positioned and related within the trophic web suggests that these species play a central role in the functioning and dynamics of upwelling ecosystems. Finally, we discuss the sustain- able exploitation of small pelagic fisheries through appropriate management, focus- ing on the resilience to exploitation, a comparison of different management options and regulatory mechanisms. We recommend that statistical, socio-economical, and political merits of a proposed two-level (short- and long-term) management strategy be undertaken. Despite constant progress in understanding the complex processes involved in the variability of pelagic stock abundance, especially at short and medium time scales, our ability to predict abundance and catches is limited, which in turn limits our ca- pacity to properly manage the fisheries and ensure sustainable exploitation. -

Kevin A. GASTON 1, Stephen J. JACQUEMIN 2, and Thomas E
ACTA ICHTHYOLOGICA ET PISCATORIA (2013) 43 (3): 219–227 DOI: 10.3750/AIP2013.43.3.06 THE INFLUENCE OF PRESERVATION ON FISH MORPHOLOGY IN MUSEUM COLLECTIONS BASED ON TWO SPECIES OF THE GENUS LEPOMIS (ACTINOPTERYGII: PERCIFORMES: CENTRARCHIDAE) Kevin A. GASTON 1, Stephen J. JACQUEMIN 2, and Thomas E. LAUER 1* 1 Department of Biology, Aquatic Biology and Fisheries Center, Ball State University, Muncie, IN 47306, USA 2 Department of Biological Sciences, Dwyer Hall, Wright State University, Lake Campus, Celina, OH 45822, USA Gaston K.A., Jacquemin S.J., Lauer T.E. 2013. The influence of preservation on fish morphology in muse - um collections based on two species of the genus Lepomis (Actinopterygii: Perciformes: Centrarchidae). Acta Ichthyol. Piscat. 43 (3): 219–227 . Background. The wide availability of specimen-based museum collections allows scientists to address a variety of ecological and systematic hypotheses from a broad timeframe and extensive spatial scale. However, tech - niques employed to preserve and maintain these collections affect morphology, potentially altering or skewing any morphological comparisons inherent in research study questions. The purpose of this study was to better understand and describe the effects that preservation techniques may have on fish specimens . Materials and methods. Bluegill, Lepomis macrochirus Rafinesque, 1819, and green sunfish, Lepomis cyanel - lus Rafinesque, 1819, were initially preserved for six weeks in a 10% formalin solution and then transferred to 70% ethyl alcohol for 46 additional weeks to determine how the commonly used preservation methods influence morphology. Fish were photographed at 0, 3, 6, 12, 24, 39, and 52 weeks of preservation to assess changes in shape. -

Blennioidei: Clinidae)
BULLETIN OF MARINE SCIENCE, 41(1): 45-58,1987 COLOR PATTERN AND HABITAT DIFFERENCES BETWEEN MALE, FEMALE AND JUVENILE GIANT KELPFISH (BLENNIOIDEI: CLINIDAE) Carol A. Stepien ABSTRACT The giant kelpfish, Heterostjehus ROSTRATUS occurs in three color morphs; red, brown, and green, which vary in shade according to number ofmelanophores. Color morphs were usually collected from plant habitats matching their colors. Frequencies of the color morphs were linked to sexual dimorphism; adult males are brown (infrequently olive green) and adult females exhibit all three morphs. Juveniles are either brown or green and not sexually di- morphic. In addition to color differences between the sexes, adult males and females display different melanin patterns. These patterns are apparently used for intraspecific communication and cryptic coloration. Brown males are distinguishable from brown females by their sexually dimorphic melanin patterns. Melanin patterns, unlike coloration, change (often rapidly) dur- ing courtship and territorial displays. Adult males and females occupy plant habitats that differ in depth, predominant color, and species composition. The brown males closely ap- proximate color of the plants where the nests they guard are found. Females occupy other habitats, including red algae, green surfgrass, and other species of brown algae. Females venture away from matching habitats during the spawning season to reach male territories. The giant kelpfish Heterostichus ROSTRATUS Girard is one of the largest members of the family Clinidae, reaching a total length of 41.2 cm (J. E. Fitch in Feder et aI., 1974). Ranging from British Columbia (Canada) to Cape San Lucas, Baja California (Mexico), it is most commonly encountered from Point Conception to central Baja California (Roedel, 1953). -

Assessment of Coastal Water Resources and Watershed Conditions at Channel Islands National Park, California
National Park Service U.S. Department of the Interior Technical Report NPS/NRWRD/NRTR-2006/354 Water Resources Division Natural Resource Program Center Natural Resource Program Centerent of the Interior ASSESSMENT OF COASTAL WATER RESOURCES AND WATERSHED CONDITIONS AT CHANNEL ISLANDS NATIONAL PARK, CALIFORNIA Dr. Diana L. Engle The National Park Service Water Resources Division is responsible for providing water resources management policy and guidelines, planning, technical assistance, training, and operational support to units of the National Park System. Program areas include water rights, water resources planning, marine resource management, regulatory guidance and review, hydrology, water quality, watershed management, watershed studies, and aquatic ecology. Technical Reports The National Park Service disseminates the results of biological, physical, and social research through the Natural Resources Technical Report Series. Natural resources inventories and monitoring activities, scientific literature reviews, bibliographies, and proceedings of technical workshops and conferences are also disseminated through this series. Mention of trade names or commercial products does not constitute endorsement or recommendation for use by the National Park Service. Copies of this report are available from the following: National Park Service (970) 225-3500 Water Resources Division 1201 Oak Ridge Drive, Suite 250 Fort Collins, CO 80525 National Park Service (303) 969-2130 Technical Information Center Denver Service Center P.O. Box 25287 Denver, CO 80225-0287 Cover photos: Top Left: Santa Cruz, Kristen Keteles Top Right: Brown Pelican, NPS photo Bottom Left: Red Abalone, NPS photo Bottom Left: Santa Rosa, Kristen Keteles Bottom Middle: Anacapa, Kristen Keteles Assessment of Coastal Water Resources and Watershed Conditions at Channel Islands National Park, California Dr. -
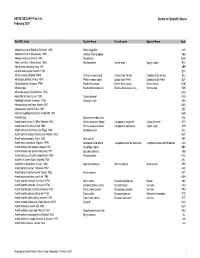
ASFIS ISSCAAP Fish List February 2007 Sorted on Scientific Name
ASFIS ISSCAAP Fish List Sorted on Scientific Name February 2007 Scientific name English Name French name Spanish Name Code Abalistes stellaris (Bloch & Schneider 1801) Starry triggerfish AJS Abbottina rivularis (Basilewsky 1855) Chinese false gudgeon ABB Ablabys binotatus (Peters 1855) Redskinfish ABW Ablennes hians (Valenciennes 1846) Flat needlefish Orphie plate Agujón sable BAF Aborichthys elongatus Hora 1921 ABE Abralia andamanika Goodrich 1898 BLK Abralia veranyi (Rüppell 1844) Verany's enope squid Encornet de Verany Enoploluria de Verany BLJ Abraliopsis pfefferi (Verany 1837) Pfeffer's enope squid Encornet de Pfeffer Enoploluria de Pfeffer BJF Abramis brama (Linnaeus 1758) Freshwater bream Brème d'eau douce Brema común FBM Abramis spp Freshwater breams nei Brèmes d'eau douce nca Bremas nep FBR Abramites eques (Steindachner 1878) ABQ Abudefduf luridus (Cuvier 1830) Canary damsel AUU Abudefduf saxatilis (Linnaeus 1758) Sergeant-major ABU Abyssobrotula galatheae Nielsen 1977 OAG Abyssocottus elochini Taliev 1955 AEZ Abythites lepidogenys (Smith & Radcliffe 1913) AHD Acanella spp Branched bamboo coral KQL Acanthacaris caeca (A. Milne Edwards 1881) Atlantic deep-sea lobster Langoustine arganelle Cigala de fondo NTK Acanthacaris tenuimana Bate 1888 Prickly deep-sea lobster Langoustine spinuleuse Cigala raspa NHI Acanthalburnus microlepis (De Filippi 1861) Blackbrow bleak AHL Acanthaphritis barbata (Okamura & Kishida 1963) NHT Acantharchus pomotis (Baird 1855) Mud sunfish AKP Acanthaxius caespitosa (Squires 1979) Deepwater mud lobster Langouste