Conserving Panamanian Harlequin Frogs by Integrating Captive-Breeding and T Research Programs ⁎ Carrie H.R
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Gibbs Thesis Final Revised Final
Species Declines: Examining patterns of species distribution, abundance, variability and conservation status in relation to anthropogenic activities Mary Katherine Elizabeth Gibbs Thesis submitted to the Faculty of Graduate and Postdoctoral Studies in partial fulfillment of the requirements for the PhD degree in the Ottawa-Carleton Institute of Biology Thèse soumise à la Faculté des étude supérieures et postdoctorales Université d’Ottawa en vue de l’obtention du doctorat de L’institut de biologie d’Ottawa-Carleton Department of Biology Faculty of Science University of Ottawa © Mary Katherine Elizabeth Gibbs, Ottawa, Canada, 2012 Acknowledgements First and foremost, I would like to thank my supervisor, Dr. David Currie. Most importantly, you gave me the freedom to work on questions that I’m passionate about. You never stopped challenging me and always asked that I push a little bit farther. I really think your commitment to scientific rigor goes above and beyond the average. And thank you for the many, many cups of tea over the years. My committee members, Dr. Scott Findlay and Dr. Mark Forbes, your feedback made this a better thesis. Scott, thank you for being scary enough to motivate me to always bring my A-game, and for providing endless new words to look up in the dictionary. By the end of this, I may actually know what ‘epistemological’ means. I would also like to thank Dr. Jeremy Kerr for his insight and not being afraid to be a scientist and an advocate. I need to thank all the lab mates that have come and gone and added so much to the last six years. -

Threat Abatement Plan
gus resulting in ch fun ytridio trid myc chy osis ith w s n ia ib h p m a f o n o i t THREAT ABATEMENTc PLAN e f n I THREAT ABATEMENT PLAN INFECTION OF AMPHIBIANS WITH CHYTRID FUNGUS RESULTING IN CHYTRIDIOMYCOSIS Department of the Environment and Heritage © Commonwealth of Australia 2006 ISBN 0 642 55029 8 Published 2006 This work is copyright. Apart from any use as permitted under the Copyright Act 1968, no part may be reproduced by any process without prior written permission from the Commonwealth, available from the Department of the Environment and Heritage. Requests and inquiries concerning reproduction and rights should be addressed to: Assistant Secretary Natural Resource Management Policy Branch Department of the Environment and Heritage PO Box 787 CANBERRA ACT 2601 This publication is available on the Internet at: www.deh.gov.au/biodiversity/threatened/publications/tap/chytrid/ For additional hard copies, please contact the Department of the Environment and Heritage, Community Information Unit on 1800 803 772. Front cover photo: Litoria genimaculata (Green-eyed tree frog) Sequential page photo: Taudactylus eungellensis (Eungella day frog) Banner photo on chapter pages: Close up of the skin of Litoria genimaculata (Green-eyed tree frog) ii Foreword ‘Infection of amphibians with chytrid fungus resulting Under the EPBC Act the Australian Government in chytridiomycosis’ was listed in July 2002 as a key implements the plan in Commonwealth areas and seeks threatening process under the Environment Protection the cooperation of the states and territories where the and Biodiversity Conservation Act 1999 (EPBC Act). disease impacts within their jurisdictions. -

Effects of Emerging Infectious Diseases on Amphibians: a Review of Experimental Studies
diversity Review Effects of Emerging Infectious Diseases on Amphibians: A Review of Experimental Studies Andrew R. Blaustein 1,*, Jenny Urbina 2 ID , Paul W. Snyder 1, Emily Reynolds 2 ID , Trang Dang 1 ID , Jason T. Hoverman 3 ID , Barbara Han 4 ID , Deanna H. Olson 5 ID , Catherine Searle 6 ID and Natalie M. Hambalek 1 1 Department of Integrative Biology, Oregon State University, Corvallis, OR 97331, USA; [email protected] (P.W.S.); [email protected] (T.D.); [email protected] (N.M.H.) 2 Environmental Sciences Graduate Program, Oregon State University, Corvallis, OR 97331, USA; [email protected] (J.U.); [email protected] (E.R.) 3 Department of Forestry and Natural Resources, Purdue University, West Lafayette, IN 47907, USA; [email protected] 4 Cary Institute of Ecosystem Studies, Millbrook, New York, NY 12545, USA; [email protected] 5 US Forest Service, Pacific Northwest Research Station, Corvallis, OR 97331, USA; [email protected] 6 Department of Biological Sciences, Purdue University, West Lafayette, IN 47907, USA; [email protected] * Correspondence [email protected]; Tel.: +1-541-737-5356 Received: 25 May 2018; Accepted: 27 July 2018; Published: 4 August 2018 Abstract: Numerous factors are contributing to the loss of biodiversity. These include complex effects of multiple abiotic and biotic stressors that may drive population losses. These losses are especially illustrated by amphibians, whose populations are declining worldwide. The causes of amphibian population declines are multifaceted and context-dependent. One major factor affecting amphibian populations is emerging infectious disease. Several pathogens and their associated diseases are especially significant contributors to amphibian population declines. -

Catalogue of the Amphibians of Venezuela: Illustrated and Annotated Species List, Distribution, and Conservation 1,2César L
Mannophryne vulcano, Male carrying tadpoles. El Ávila (Parque Nacional Guairarepano), Distrito Federal. Photo: Jose Vieira. We want to dedicate this work to some outstanding individuals who encouraged us, directly or indirectly, and are no longer with us. They were colleagues and close friends, and their friendship will remain for years to come. César Molina Rodríguez (1960–2015) Erik Arrieta Márquez (1978–2008) Jose Ayarzagüena Sanz (1952–2011) Saúl Gutiérrez Eljuri (1960–2012) Juan Rivero (1923–2014) Luis Scott (1948–2011) Marco Natera Mumaw (1972–2010) Official journal website: Amphibian & Reptile Conservation amphibian-reptile-conservation.org 13(1) [Special Section]: 1–198 (e180). Catalogue of the amphibians of Venezuela: Illustrated and annotated species list, distribution, and conservation 1,2César L. Barrio-Amorós, 3,4Fernando J. M. Rojas-Runjaic, and 5J. Celsa Señaris 1Fundación AndígenA, Apartado Postal 210, Mérida, VENEZUELA 2Current address: Doc Frog Expeditions, Uvita de Osa, COSTA RICA 3Fundación La Salle de Ciencias Naturales, Museo de Historia Natural La Salle, Apartado Postal 1930, Caracas 1010-A, VENEZUELA 4Current address: Pontifícia Universidade Católica do Río Grande do Sul (PUCRS), Laboratório de Sistemática de Vertebrados, Av. Ipiranga 6681, Porto Alegre, RS 90619–900, BRAZIL 5Instituto Venezolano de Investigaciones Científicas, Altos de Pipe, apartado 20632, Caracas 1020, VENEZUELA Abstract.—Presented is an annotated checklist of the amphibians of Venezuela, current as of December 2018. The last comprehensive list (Barrio-Amorós 2009c) included a total of 333 species, while the current catalogue lists 387 species (370 anurans, 10 caecilians, and seven salamanders), including 28 species not yet described or properly identified. Fifty species and four genera are added to the previous list, 25 species are deleted, and 47 experienced nomenclatural changes. -

Amphibian Alliance for Zero Extinction Sites in Chiapas and Oaxaca
Amphibian Alliance for Zero Extinction Sites in Chiapas and Oaxaca John F. Lamoreux, Meghan W. McKnight, and Rodolfo Cabrera Hernandez Occasional Paper of the IUCN Species Survival Commission No. 53 Amphibian Alliance for Zero Extinction Sites in Chiapas and Oaxaca John F. Lamoreux, Meghan W. McKnight, and Rodolfo Cabrera Hernandez Occasional Paper of the IUCN Species Survival Commission No. 53 The designation of geographical entities in this book, and the presentation of the material, do not imply the expression of any opinion whatsoever on the part of IUCN concerning the legal status of any country, territory, or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. The views expressed in this publication do not necessarily reflect those of IUCN or other participating organizations. Published by: IUCN, Gland, Switzerland Copyright: © 2015 International Union for Conservation of Nature and Natural Resources Reproduction of this publication for educational or other non-commercial purposes is authorized without prior written permission from the copyright holder provided the source is fully acknowledged. Reproduction of this publication for resale or other commercial purposes is prohibited without prior written permission of the copyright holder. Citation: Lamoreux, J. F., McKnight, M. W., and R. Cabrera Hernandez (2015). Amphibian Alliance for Zero Extinction Sites in Chiapas and Oaxaca. Gland, Switzerland: IUCN. xxiv + 320pp. ISBN: 978-2-8317-1717-3 DOI: 10.2305/IUCN.CH.2015.SSC-OP.53.en Cover photographs: Totontepec landscape; new Plectrohyla species, Ixalotriton niger, Concepción Pápalo, Thorius minutissimus, Craugastor pozo (panels, left to right) Back cover photograph: Collecting in Chamula, Chiapas Photo credits: The cover photographs were taken by the authors under grant agreements with the two main project funders: NGS and CEPF. -

Maritime Southeast Asia and Oceania Regional Focus
November 2011 Vol. 99 www.amphibians.orgFrogLogNews from the herpetological community Regional Focus Maritime Southeast Asia and Oceania INSIDE News from the ASG Regional Updates Global Focus Recent Publications General Announcements And More..... Spotted Treefrog Nyctixalus pictus. Photo: Leong Tzi Ming New The 2012 Sabin Members’ Award for Amphibian Conservation is now Bulletin open for nomination Board FrogLog Vol. 99 | November 2011 | 1 Follow the ASG on facebook www.facebook.com/amphibiansdotor2 | FrogLog Vol. 99| November 2011 g $PSKLELDQ$UN FDOHQGDUVDUHQRZDYDLODEOH 7KHWZHOYHVSHFWDFXODUZLQQLQJSKRWRVIURP $PSKLELDQ$UN¶VLQWHUQDWLRQDODPSKLELDQ SKRWRJUDSK\FRPSHWLWLRQKDYHEHHQLQFOXGHGLQ $PSKLELDQ$UN¶VEHDXWLIXOZDOOFDOHQGDU7KH FDOHQGDUVDUHQRZDYDLODEOHIRUVDOHDQGSURFHHGV DPSKLELDQDUN IURPVDOHVZLOOJRWRZDUGVVDYLQJWKUHDWHQHG :DOOFDOHQGDU DPSKLELDQVSHFLHV 3ULFLQJIRUFDOHQGDUVYDULHVGHSHQGLQJRQ WKHQXPEHURIFDOHQGDUVRUGHUHG±WKHPRUH \RXRUGHUWKHPRUH\RXVDYH2UGHUVRI FDOHQGDUVDUHSULFHGDW86HDFKRUGHUV RIEHWZHHQFDOHQGDUVGURSWKHSULFHWR 86HDFKDQGRUGHUVRIDUHSULFHGDW MXVW86HDFK 7KHVHSULFHVGRQRWLQFOXGH VKLSSLQJ $VZHOODVRUGHULQJFDOHQGDUVIRU\RXUVHOIIULHQGV DQGIDPLO\ZK\QRWSXUFKDVHVRPHFDOHQGDUV IRUUHVDOHWKURXJK\RXU UHWDLORXWOHWVRUIRUJLIWV IRUVWDIIVSRQVRUVRUIRU IXQGUDLVLQJHYHQWV" 2UGHU\RXUFDOHQGDUVIURPRXUZHEVLWH ZZZDPSKLELDQDUNRUJFDOHQGDURUGHUIRUP 5HPHPEHU±DVZHOODVKDYLQJDVSHFWDFXODUFDOHQGDU WRNHHSWUDFNRIDOO\RXULPSRUWDQWGDWHV\RX¶OODOVREH GLUHFWO\KHOSLQJWRVDYHDPSKLELDQVDVDOOSUR¿WVZLOOEH XVHGWRVXSSRUWDPSKLELDQFRQVHUYDWLRQSURMHFWV ZZZDPSKLELDQDUNRUJ FrogLog Vol. 99 | November -

Real-Time DNA Barcoding in a Remote Rainforest Using Nanopore Sequencing
bioRxiv preprint doi: https://doi.org/10.1101/189159; this version posted September 15, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. 1 Real-time DNA barcoding in a remote rainforest using nanopore sequencing 2 3 Aaron Pomerantz1,*, Nicolás Peñafiel2, Alejandro Arteaga3, Lucas Bustamante3, Frank Pichardo3, 4 Luis A. Coloma4, César L. Barrio-Amorós5, David Salazar-Valenzuela2, Stefan Prost 1,6,* 5 6 1 Department of Integrative Biology, University of California, Berkeley, CA, USA 7 2 Centro de Investigación de la Biodiversidad y Cambio Climático (BioCamb) e Ingeniería en 8 Biodiversidad y Recursos Genéticos, Facultad de Ciencias de Medio Ambiente, Universidad 9 Tecnológica Indoamérica, Machala y Sabanilla, Quito, Ecuador 10 3 Tropical Herping, Quito, Ecuador 11 4 Centro Jambatu de Investigación y Conservación de Anfibios, Fundación Otonga, Quito, 12 Ecuador 13 5 Doc Frog Expeditions, Uvita, Costa Rica 14 6 Program for Conservation Genomics, Department of Biology, Stanford University, Stanford, CA, 15 USA 16 17 * Corresponding authors: [email protected] (A. Pomerantz) and 18 [email protected] (S. Prost) 19 20 Abstract 21 Advancements in portable scientific instruments provide promising avenues to expedite 22 field work in order to understand the diverse array of organisms that inhabit our planet. 23 Here we tested the feasibility for in situ molecular analyses of endemic fauna using a 24 portable laboratory fitting within a single backpack, in one of the world’s most imperiled 25 biodiversity hotspots: the Ecuadorian Chocó rainforest. -

Aark: Keeping Threatened Amphibian Species Afloat
APRIL 2017 AMPHIBIAN SURVIVAL ALLIANCE NEWTSLETTER Got a story you want to share? Drop Candace an email today! [email protected] Stories from our partners around the world AArk: Keeping threatened amphibian species afloat which will hopefully mean less work for the expert as- sessors, and will increase the number of assessments that can be completed. We have enlisted 10 very eager and capable volunteers, who are currently drafting as- sessments, based on the data in previously-published species accounts. Once the draft assessments have been made, using all available data, we will then ask the appropriate species experts to review the drafts, update any additional data, and then approve them. This process of pre-filling assessments has been used for amphibian assessments in the IUCN Red List, and has proven to be very beneficial. © Gilbert Alvarado Barboza © Alvarado Gilbert Our Training Officer is planning for a couple of capacity- During the past two months Amphibian Ark has been building courses in the coming months - the Biology, involved in a number of projects. Management and Conservation of North American Salamanders course will be held at Zoo Atlanta, Geor- We are continuing to facilitate national Conservation gia, USA, September 18th–22nd, and the Guatemalan Needs Assessments, and at the moment we are concen- Amphibian Biology, Management and Conservation trating on species from the Western Ghats in India, and Training Course will be held at the Universidad del Valle North American salamanders. With several groups keen de Guatemala in November 2017. Information about to establish ex situ conservation programs for species both courses can be found on the AArk web site, www. -
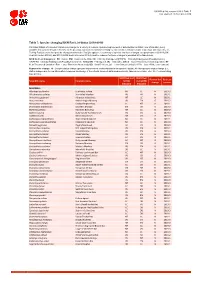
Table 7: Species Changing IUCN Red List Status (2018-2019)
IUCN Red List version 2019-3: Table 7 Last Updated: 10 December 2019 Table 7: Species changing IUCN Red List Status (2018-2019) Published listings of a species' status may change for a variety of reasons (genuine improvement or deterioration in status; new information being available that was not known at the time of the previous assessment; taxonomic changes; corrections to mistakes made in previous assessments, etc. To help Red List users interpret the changes between the Red List updates, a summary of species that have changed category between 2018 (IUCN Red List version 2018-2) and 2019 (IUCN Red List version 2019-3) and the reasons for these changes is provided in the table below. IUCN Red List Categories: EX - Extinct, EW - Extinct in the Wild, CR - Critically Endangered [CR(PE) - Critically Endangered (Possibly Extinct), CR(PEW) - Critically Endangered (Possibly Extinct in the Wild)], EN - Endangered, VU - Vulnerable, LR/cd - Lower Risk/conservation dependent, NT - Near Threatened (includes LR/nt - Lower Risk/near threatened), DD - Data Deficient, LC - Least Concern (includes LR/lc - Lower Risk, least concern). Reasons for change: G - Genuine status change (genuine improvement or deterioration in the species' status); N - Non-genuine status change (i.e., status changes due to new information, improved knowledge of the criteria, incorrect data used previously, taxonomic revision, etc.); E - Previous listing was an Error. IUCN Red List IUCN Red Reason for Red List Scientific name Common name (2018) List (2019) change version Category -

Chytridiomycosis Causes Amphibian Mortality Associated with Population Declines in the Rain Forests of Australia and Central America
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 9031–9036, July 1998 Population Biology Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America LEE BERGERa,b,c,RICK SPEAREa,PETER DASZAKd,D.EARL GREENe,ANDREW A. CUNNINGHAMf,C.LOUISE GOGGINg, RON SLOCOMBEh,MARK A. RAGANi,ALEX D. HYATTb,KEITH R. MCDONALDj,HARRY B. HINESk,KAREN R. LIPSl, GERRY MARANTELLIm, AND HELEN PARKESb aSchool of Public Health and Tropical Medicine, James Cook University, Townsville, Queensland 4811, Australia; bAustralian Animal Health Laboratory, Commonwealth Scientific and Industrial Research Organization, Ryrie Street, Geelong, Victoria 3220, Australia; dSchool of Life Sciences, Kingston University, Kingston-upon-Thames, Surrey KT1 2EE, United Kingdom; eMaryland Animal Health Laboratory, College Park, MD 20740; fInstitute of Zoology, Zoological Society of London, Regent’s Park, London NW1 4RY, United Kingdom; gCommonwealth Scientific and Industrial Research Organization, Marine Research, Hobart, Tasmania 7001, Australia; hVeterinary Clinical Centre, University of Melbourne, Werribee, Victoria 3030, Australia; iCanadian Institute for Advanced Research, Program in Evolutionary Biology, National Research Council of Canada, Halifax, NS Canada B3H 3Z1; jConservation Strategy Branch, Queensland Department of Environment, Atherton, Queensland 4883, Australia; kConservation Resource Unit, Queensland Department of Environment, Moggill, Queensland 4070, Australia; lDepartment of Zoology, Southern Illinois University, Carbondale, IL 62901-6501; and mAmphibian Research Centre, 15 Suvla Grove, Nth Coburg, Victoria 3058, Australia Edited by Robert May, University of Oxford, Oxford, United Kingdom, and approved May 18, 1998 (received for review March 9, 1998) ABSTRACT Epidermal changes caused by a chytridiomy- primary degraders or saprobes, using substrates such as chitin, cete fungus (Chytridiomycota; Chytridiales) were found in plant detritus, and keratin. -

Visual Signaling in Anuran Amphibians
.. Hödl, W. and Amezquita, A. (2001). Visual signaling in anuran amphibians. In: Anuran communication, (M.J. Ryan, ed.). .. Smithsonian lust. Press, Washington. Pp. 121-141. 10 WALTER HÖDL AND ADOLFO AMEZQUITA Visual Signaling in Anuran Amphibians lntroduction cation. social behavior, or natural history. visual signaling was either not considered or was treated as a minor subject Acoustic communication plays a fundamental role in an- (Wells 1977a, 1977b; Arak 1983; Duellman and Trueb 1986; uran reproduction and thus is involved in evolutionary Rand 1988; Halliday and Tejedo 1995; Stebbins and Cohen processes such as mate recognition. reproductive isolation. 1995; Sullivan et al. 1995). The most detailed review ofthe speciation. and character displacement (Wells 1977a. 1977b. subject is now more than 20 years old (Wells 1977b). Never- 1988;Rand 1988;Gerhardt and Schwartz 1995;Halliday and theless some authors have discussed the possible evolution- Tejedo 1995;Sullivan et al. 1995).Visual cues. however. have ary link between visual signaling and the reproductive ecol- been thought to function only during dose-range inter- ogy of species, such as reproduction associated with streams actions (Wells 1977c; Duellman and Trueb 1986). Visual sig- (Heyer et aI. 1990; Lindquist and Hetherington 1996. 1998; naling is predicted to be predominantly employed by diur- Hödl et al. 1997;Haddad and Giaretta 1999) or reproduction nal species at sites with an unobstructed view (Endler 1992). within feeding territories (Wells 1977c). Diurnality. however. is not common for the majority offrog Our aim in this review is (1) to propose a dassmcation of species. Thus vocalizations. which are highly efficient for reported behavioral patterns of visual signaling in frags; (2) communicating at night or in dense vegetation, are by far to describe the diversity of visual signals among living an- the best studied anuran signals (Duellman and Trueb 1986; uran taxa; and (3) to apply a comparative approach to explor- Fritzsch et aI. -

Amphibian Conservation INTRODUCTION
2014 | HIGHLIGHTS AND ACCOMPLISHMENTS amphibian conservation INTRODUCTION Zoos and aquariums accredited by the Association of Zoos and Aquariums (AZA) have made long-term commitments, both individually and as a community organized under the Amphibian Taxon Advisory Group (ATAG), to the conservation of amphibians throughout the Americas and around the world. With the support and hard work of directors, curators, keepers and partners, 85 AZA-accredited zoos and aquariums reported spending more than $4.2 million to maintain, adapt and expand amphibian conservation programs in 2014. The stories in this report are drawn primarily from annual submissions to AZA’s field conservation database (available when logged into AZA’s website under “Conservation”), as well as from articles submitted directly to AZA. They share the successes and advances in the areas of reintroduction and research, conservation breeding and husbandry and citizen science and community engagement. These efforts are the result of extensive collaborations and multi-year (even multi-decadal!) commitments. AZA congratulates each of the members included in this report for their dedication, and encourages other facilities to become involved. The ATAG has many resources to help people get started or to expand their engagement in amphibian conservation, and people are also welcome to contact the facilities included in this report or the ATAG Chair, Diane Barber ([email protected]). Cover: Spring peeper (Pseudacris crucifer). Widespread throughout the eastern United States and with a familiar call to many, the spring peeper was the most frequently reported frog by FrogWatch USA volunteers in 2014. Although reports of spring peepers began in February, they peaked in April.