246 Volodin Et Al 2019 Mamb
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
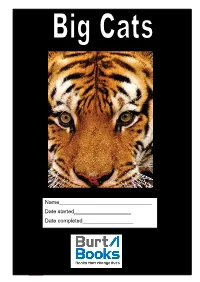
Name___Date Started___Date Completed___
# Name_______________________________ Date started___________________ Date completed_________________ Copyright©2011 Burt Books: All rights reserved worldwide. This 1 worksheet may be reproduced by the purchaser only and not for on- sale in quantities sufficient for pupils. Burt Books - www.burtbooks.com Big Cats Copyright © Burt Books Ltd. 2011 Church Cottage Albemarle Crescent Scarborough North Yorkshire YO11 1XX www.burtbooks.com [email protected] First published in the United Kingdom in 2011 By Burt Books Ltd. All rights reserved worldwide: No part of this publication may be reproduced or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, or stored in any retrieval system of any nature without the written consent of the copyright holder and the publisher, application for which should be made to Burt Books ltd. The right of Coreen Burt to be identified as the author of Big Cats has been asserted by her in accordance with the Copyright, Designs and Patents Act 1988. THE DOWNLOAD OF THIS BIG CATS THEME ALLOWS FOR THE PRINT ING OF COPIES FOR INDIVIDUAL PUPILS ONLY AND NOT FOR DISTRIBUTION OR SALE TO OTHERS Learning Objectives My learning objectives for this theme are to: 1. Revise and remember high frequency spellings 2. Learn complex words that do not conform to regular patterns. 3. Apply spelling rules and recognise exceptions. 4. Appreciate the impact of figurative language in texts. 5. Improve my vocabulary by working out the meaning of unknown words in the text. Copyright©2011 Burt Books: All rights reserved worldwide. This 2 worksheet may be reproduced by the purchaser only and not for on- sale in quantities sufficient for pupils. -

Discrimination of Individual Tigers (Panthera Tigris) from Long Distance Roars an Ji Marquette University, [email protected]
Marquette University e-Publications@Marquette Electrical and Computer Engineering Faculty Electrical and Computer Engineering, Department Research and Publications of 3-1-2013 Discrimination of Individual Tigers (Panthera tigris) from Long Distance Roars An Ji Marquette University, [email protected] Michael T. Johnson Marquette University, [email protected] Edward J. Walsh Boys Town National Research Hospital JoAnn McGee Boys Town National Research Hospital Douglas L. Armstrong Omaha's Henry Doorly Zoo Published version. Journal of the Acoustical Society of America, Vol. 133, No. 3 (March 2013): 1762-1769. DOI. © 2013 Acoustical Society of America. Used with permission. Discrimination of individual tigers (Panthera tigris) from long distance roars An Ji and Michael T. Johnsona) Department of Electrical and Computer Engineering, Marquette University, 1515 West Wisconsin Avenue, Milwaukee, Wisconsin 53233 Edward J. Walsh and JoAnn McGee Developmental Auditory Physiology Laboratory, Boys Town National Research Hospital, 555 North 30th Street, Omaha, Nebraska 68132 Douglas L. Armstrong Omaha’s Henry Doorly Zoo, 3701 South 10th Street, Omaha, Nebraska 68107 (Received 8 December 2011; revised 11 January 2013; accepted 16 January 2013) This paper investigates the extent of tiger (Panthera tigris) vocal individuality through both qualita- tive and quantitative approaches using long distance roars from six individual tigers at Omaha’s Henry Doorly Zoo in Omaha, NE. The framework for comparison across individuals includes sta- tistical and discriminant function analysis across whole vocalization measures and statistical pattern classification using a hidden Markov model (HMM) with frame-based spectral features comprised of Greenwood frequency cepstral coefficients. Individual discrimination accuracy is evaluated as a function of spectral model complexity, represented by the number of mixtures in the underlying Gaussian mixture model (GMM), and temporal model complexity, represented by the number of se- quential states in the HMM. -

Aggressive Body Language of Bears and Wildlife Viewing: a Response to Geist (2011) STEPHEN F
Human–Wildlife Interactions 5(2):177–191, Fall 2011 Aggressive body language of bears and wildlife viewing: a response to Geist (2011) STEPHEN F. S TRINGHAM, WildWatch LLC, 39200 Alma Avenue, Soldotna, AK 99669, USA [email protected] Key words: attack, bear, behavior, broadside display, communication, curiosity, frontal display, human–wildlife confl icts, threat, Ursus, viewing Geist’s (2011) commentary has 3 main (Stringham 2008, Smith et al., in press). points: (1) habituation increases risk that Defensiveness is the major cause of serious or large-bodied wildlife will injure people; (2) an fatal injuries infl icted by brown bears (Ursus animal’s body language oft en provides reliable arctos) and a cause of lesser injury by black clues of impending assault; and (3) although bears (Ursus americanus) Herrero 1985, Herrero some of those clues are obvious, others are and Higgins 1995, 2003). easily overlooked by untrained people. Geist (1978) and Walther (1984) provide Whereas Geist’s emphasis is on ungulates, he much of the information on contexts for also suggests that similar behaviors by bears agonistic signals by ungulates; but litt le of it have the same signifi cance; this is an issue that has been published for bears, and that litt le requires clarifi cation. is widely scatt ered through the literature. In The importance of recognizing signals that this paper, I summarize that literature plus sometimes preface att ack is beyond questioning my own fi ndings. These are based on 22 fi eld for those of us who frequently encounter seasons observing bears and bear viewers—15 potentially dangerous wildlife, whether as seasons with brown bears in Alaska and 7 with professional or recreational observers. -

Animal Communication Safari with Anna Breytenbach Tswalu, South
In association with Animal Communication Safari with Anna Breytenbach Tswalu, South Africa | 15-21 January 2018 Guided by James Kydd ITINERARY OVERVIEW Tswalu Motse for 6 nights on a fully inclusive basis In: 15 January 2018 Out: 21 January 2018 Day 01 Arrive at Johannesburg O R Tambo International Airport, Johannesburg or Cape Town International (depending on your travel plans) You will be met on arrival and transferred to Tswalu Meet Anna and James on arrival followed by a welcome lunch Afternoon opening meditation, introductions and briefing Days 02 to 06 Exciting programme of learning and practical application in the field, including but not limited to: Morning activities in the wild Workshop sessions in camp Game drives Sensory expansion Tracking excursions and bush walks Evening interpretive adventures Guided processes and mentoring Day 07 Closing session in camp Depart from Tswalu Transfer to Johannesburg or Cape Town airports (depending on your travel plans) ANIMAL COMMUNICATION SAFARI DETAILS Learn about the natural world by seeing into the lives of wild animals through the tracks they leave behind and through direct communications with them. You’ll be shown how to connect telepathically and be given ample opportunity to play with your intuition in the field during walking adventures and open-vehicle drives. Getting to know the wildlife personally takes the concept of game-viewing to a new and profound level of empathy, intimacy and insight. Anna introduces you to the principles and techniques of interspecies communication, using fun exercises to expand your senses. Each day comprises of two hours of workshop sessions designed to empower you in using your own telepathic abilities. -

Silent Roar | UNDP and GEF in the Snow Leopard Landscape
25years Empowered lives. Resilient nations. Silent Roar UNDP AND GEF IN THE SNOW LEOPARD LANDSCAPE ACKNOWLEDGEMENTS CONTENTS Managing Editors: Midori Paxton, Tim Scott, Yoko Watanabe Compilation and Editing: Erin Charles INTRODUCTION 2 CO-MANAGEMENT ON THE ROOF 21 CONCLUSION 44 OF THE WORLD—CHINA Core Writing Team: Erin Charles, Midori Paxton, Tim Scott, Doley Tshering, Inela Weeks SNOW LEOPARD RANGE MAP 4 PARTNER SPOTLIGHT: 23 We wish to acknowledge the UNDP and GEF staff, consultants, and partners who contributed to this publication: GOVERNMENTS AND GOVERNANCE Tehmina Akhtar, Ana Maria Currea, Adriana Dinu, Lisa Farroway, Gustavo Fonseca, Uyanga Gankhuyag, Christian LESSONS LEARNED: RUSSIA, 23 ART AND CULTURE Hofer, Daniar Ibragimov, Kyle Kaufman, Khurshed Kholov, Fan Longqing, Cathy Maize, Ruchi Pant, Pakamon MONGOLIA, AND KAZAKHSTAN Pinprayoon, Evgeniia Postnova, Ajiniyaz Reimov, Olga Romanova, Nadisha Sidhu, Nargizakhon Usmanova, SNOW LEOPARDS IN LITERATURE 19 SECTION 1: WHY SNOW LEOPARDS? 6 STRENGTHENING AND EXPANDING 24 Maxim Vergeichik, Katerina Yushenko, Yuqiong Zhou, with special thanks to Marc Foggin and John MacKinnon WHY PROTECT THE HIGH 8 PROTECTED AREAS—KAZAKHSTAN SNOW LEOPARDS AND 21 for generously permitting the extensive use of their photographs. TIBETAN BUDDHISM MOUNTAIN LANDSCAPES? PARTNER SPOTLIGHT: TRAINING 25 We wish to acknowledge the central role of the GSLEP Secretariat: Hamid Zahid (Chair), Abdykalyk Rustamov CULTURAL-POLITICAL SYMBOLS 37 UNIQUE BIODIVERSITY 8 OF TRAINERS (Co-chair), Kyial Alygulova, Yash Veer Bhatnagar, Chyngyz Kochorov, Andrey Kushlin, Keshav Varma, with special LAND USE PLANNING & BIOLOGICAL 26 SACRED BELIEFS, MYTHS 40 ASIA'S WATER TOWER 9 thanks to Koustubh Sharma and Matthias Fiechter for their technical review of this document, contributions CORRIDORS— KYRGYZSTAN AND LEGENDS of text, photographs, maps and invaluable feedback on all aspects of the publication; and the Permanent CULTURAL HERITAGE 9 LESSONS LEARNED: BHUTAN 27 Mission of the Kyrgyz Republic to the UN in New York: Madina Karabaeva. -

Evaluation of Reproductive Behavior in White-Tailed Deer Through Genetic Parentage Analysis by Stephanie Kimie Irvin a Thesis Su
Evaluation of Reproductive Behavior in White-tailed Deer through Genetic Parentage Analysis by Stephanie Kimie Irvin A thesis submitted to the Graduate Faculty of Auburn University in partial fulfillment of the requirements for the Degree of Master of Science Auburn, Alabama August 6, 2011 Keywords: White-tailed deer, inbreeding avoidance, male reproductive success, female mate choice Approved by Stephen S. Ditchkoff, Chair, Professor of Forestry and Wildlife Sciences Mark D. Smith, Assistant Professor of Forestry and Wildlife Sciences Todd D. Steury, Assistant Professor of Forestry and Wildlife Sciences Abstract Few studies have utilized genetic technology to examine reproductive behavior in white-tailed deer through parentage analysis, resulting in gaps in knowledge of reproductive behavior in white-tailed deer. This study examined relationships between reproductive success in male white-tailed deer and body size, age, and antler characteristics, age-related mate choice, and inbreeding avoidance using genetic parentage assignment. Between December 2007 and August 2010, 115 deer from inside a 174-ha high-fence facility in Alabama were sampled and genotyped. Fifty-four paternities and 48 maternities were assigned using the program CERVUS. No relationship was detected between male reproductive success, body size, and antler characteristics. However, prime-aged males were more likely to breed and sired more offspring on average than younger males. Three cases of multiple paternity were observed, including the first reported triplet multiple paternity with 3 different sires. Evidence of age-related mate choice was identified, but no evidence of inbreeding avoidance behaviors was found. ii Acknowledgments I thank Dr. Steve Ditchkoff for taking me on as his student and tempering me into a better scientist. -

SAMPLE – Animals – Simple Sentences – ESL Library
Simple Sentences Animals VocabularySample What kind of animal is this? What do you know about this animal? Use the words below to fill in the blanks. Sentence 1 Sentence 2 • bear • lion • cub • cute • monkey • dog • ooh-ooh ah-ah • stripes • pandas • deer • calves • kitten • zebra • kangaroos • doe • trunk • elephant • hippos • joey • roar • rabbit • cat • puppies • bunny # Picture Sentences This is a bear . 1 Her baby is a cub . This is a . 2 Her baby is a . This is my , Sandy. 3 Sandy’s babies are . Copyright 2018, Red River Press Inc. For use by ESL Library members only. (BEG – LOW INT / VERSION 3.1) 1 Animals Simple Sentences Vocabulary cont. # Picture Sentences SampleThis is a . 4 A female is called a . are dangerous. 5 Their babies are called . live in Australia. 6 A baby is a . This is a . 7 These animals . This is a . 8 He says “ .” I have a pet . 9 I love my . This is a . 10 It has black and white . I love . 11 They are . This is an . 12 It has a long . Copyright 2018, Red River Press Inc. For use by ESL Library members only. (BEG – LOW INT / VERSION 3.1) 2 Animals Simple Sentences Expressions A. Defining Animals SampleAnimal Classes Here are some ways to define animals. • mammals • rodents animal + be + animal class + with + feature • birds • A moose is a mammal with antlers. • reptiles • A crow is a bird with black feathers. • amphibians • A mouse is a rodent with a long tail and sharp teeth. • fish • A frog is an amphibian with strong legs. -

2019 Annual Report Panthera’S Mission Is to Ensure a Future for Wild Cats and the Vast Landscapes on Which They Depend
Panthera 2019 Annual Report Panthera’s mission is to ensure a future for wild cats and the vast landscapes on which they depend. Panthera Our vision is a world where wild cats thrive in healthy, natural and developed landscapes that sustain people and biodiversity. Contents 04 08 12 14 Nature Bats Last Cores and Conservation Program by Thomas S. Kaplan, Ph.D. Corridors in a Global Highlights Community 34 36 38 40 CLOUDIE ON CAMERA The Arabian A Corridor Searching for Conservation “I am particularly fond of this photograph of a clouded leopard Leopard to the World New Frontiers Science and because of the high likelihood that I wouldn’t capture it. After a leech and mosquito-filled five-day jungle trek, the biologists Initiatives Technology and I arrived at a ranger station at the top of the mountain in Highlights Malaysian Borneo, close to where this camera trap was located. I checked it but saw the battery was on its last leg. I decided to take the grueling full day’s hike back and forth to pick up a fresh battery. When I checked it the following afternoon, this young adult had come through just hours before. The physical 43 44 46 49 exhaustion was totally worth getting this amazing photograph.” 2019 Financial Board, Staff and Conservation After the Fires - Sebastian Kennerknecht, Panthera Partner Photographer Summary Science Council Council by Esteban Payán, Ph.D. 2 — 2019 ANNUAL REPORT A leopard in the Okavango Delta, Botswana Nature Bats Last The power of nature is an awesome thing to contemplate. the Jaguar Corridor. -

Sonorensis 2008
sonorensis Introduction Arizona-Sonora Desert Museum contents Newsletter Volume 28, Number 1 Winter 2008 Introduction I 1 D The Arizona-Sonora Desert Museum By Craig Ivanyi E The Big Cat of the Americas Co-founded in 1952 by & Shawnee Arthur N. Pack and William H. Carr Riplog Peterson 3 Jaguars: The New World Monarchs By Craig Ivanyi, Associate Executive Director for Living Collections and Exhibits, and Robert J. Edison By Peter Warshall, Ph.D. P J Shawnee Riplog Peterson, Curator of Mammalogy and Ornithology Executive Director N Arizona-Sonora Desert Museum Christine Conte, Ph.D. Director, Center for Sonoran The Future of Jaguar Conservation P 9 J Desert Studies N in Mexico and our Conscience By Rodrigo A. Medellín, Ph.D. Richard C. Brusca, Ph.D. Senior Director, Science and Conservation 12 Binational Collaboration to Save This issue of sonorensis focuses on one of the Sonoran Desert Region’s the Planet’s Northernmost Jaguar I Linda M. Brewer D E most beautiful and powerful inhabitants—the jaguar, Panthera onca. Chiefly Editing By Oscar Moctezuma & Diana Hadley t h solitary, this wide-ranging predator is the third-largest cat in the world and the g i n Martina Clary K d l a Design and Production n only true big, or “roaring,” cat from the Americas! With an exceptionally o D 18 Jaguar Conservation through Education and Outreach powerful bite, it can crack the armor of turtles and tortoises, and will often kill Board of Trustees – Officers By Juan Carlos G. Bravo mammals by biting directly through the skull, between the ears, puncturing its William H. -
DIY Storytime #2
DIY HOME STORYTIME: Lions and Tigers and Bears, Oh My! Use your Amesbury Public Library card & Boston Public Library eCard to find these suggested titles and more on Libby/Overdrive & Hoopla! Amesbury Public Library is now offering curbside pickup! Visit our website at www.amesburylibrary.org or call us at 978-388-8148 for more information about placing holds and the pickup process! BOOKS: Going on a Bear Hunt Act out each verse as you sing (feel free to go on a tiger or a lion Little Red and the Very Hungry Lion (Overdrive-BPL) hunt instead!) Need to see it in action? Check out this version by Alex T. Smith by The Kiboomers: https://www.youtube.com/watch?v=5_ShP3fiEhU Lion Lessons (Overdrive-BPL) by Jon Agee We’re going on a bear hunt! (We’re going on a bear hunt!) It's a Tiger! (Overdrive-BPL) It’s a beautiful day! (It’s a beautiful day!) by David LaRochelle We’re not scared! (We’re not scared!) I Am a Cat (Overdrive-BPL) We’re coming to some grass. (We’re coming to some grass). by Galia Bernstein Can’t go over it. (Can’t go over it.) Big Bear Hug (Overdrive-BPL) Can’t go under it. (Can’t go under it.) by Nicholas Oldland Have to go through it. (Have to go through it.) Breathe Like a Bear (Overdrive-BPL) Swish! Swish! Swish! Swish! by Kira Willey Repeat with the following verses: Coming to some mud. (Squilch! Squelch! Squilch! Squelch!) MUSIC & MOVEMENT: Coming to a lake. -
An Acoustic Analysis of Lion Roars. II: Vocal Tract Characteristics
TMH - QPSR Vol. 51 An acoustic analysis of lion roars. II: Vocal tract characteristics G. Ananthakrishnan 1, Robert Eklund 2,3,4, Gustav Peters 5 & Evans Mabiza 6 1 Centre for Speech Technology, KTH, Stockholm, Sweden 2 Voice Provider, Stockholm, Sweden 3 Department of Cognitive Neuroscience, Karolinska Institute, Stockholm, Sweden 4 Department of Computer Science, Linkoping¨ University, Linkoping,¨ Sweden 5 Forschungsinstitut Alexander Koenig, Bonn, Germany 6 Antelope Park, Gweru, Zimbabwe Abstract This paper makes the first attempt to perform an acoustic-to-articulatory inversion of a lion (Panthera leo) roar. The main problems that one encounters in attempting this, is the fact that little is known about the dimensions of the vocal tract, other than a general range of vocal tract lengths. Precious little is also known about the articulation strategies that are adopted by the lion while roaring. The approach used here is to iterate between possible values of vocal tract lengths and vocal tract configurations. Since there seems to be a distinct articulatory changes during the process of a roar, we find a smooth path that minimizes the error function between a recorded roar and the simulated roar using a variable length articulatory model. Introduction of three different phases (Peters, 1978). The first phase is a series of low-intensity calls The roar is a distinct mammalian vocalization similar to ‘mews’. The second phase, builds up made by only five species of Felidae. Researchers to the climax with calls of increasing duration suggest that the ability to roar is made possible (shortening again towards the climax). Finally the due to the specialized hyoid apparatus present sequence ends with a series of ‘grunt’ like sounds. -

Snow Leopard Factfile
SNOW LEOPARD FACTFILE NAME Snow Leopard SCIENTIFIC NAME Panthera Uncia GEOGRAPHIC RANGE 12 countries across central Asia. HABITAT Mountains LIFESPAN 10-12 years in the wild. Up to 20 years in captivity. WEIGHT 20– 55kg DIET Mainly ibex (wild goat), blue sheep and argali (wild sheep). WILD POPULATION Between 4,000 and 6,500 individuals. IUCN RED LIST STATUS A high risk of becoming extinct in the wild. GENERAL DESCRIPTION Snow leopards are a very elusive cat, perfectly adapted for living in the high altitudes of the Himalayas. They have short, strong front limbs and longer back limbs, allowing them to jump distances of up to 30 feet! They have large paws, acting like snow shoes, preventing them from sinking in the snow. Snow leopards have small ears relative to their body size, reducing heat loss and short, wide nasal cavities, warming the air before it hits the lungs. The snow leopards have a very tail! It helps when balancing and wraps around the body to the face, providing extra warmth. Snow leopards have soft, dense fur, keeping them warm in cold climate. Snow leopards are seasonal breeders and will come into season between January and mid-March. Typically snow leopards are solitary, but during mating season may be seen together for several days. Where prey is sparse a snow leopards home range may be as large as 1,000km², making it difficult to track down a mate. To communicate snow leopards will leave markings on the landscape. Despite being a big cat, snow leopards are unable to roar due to the physiology of their throat and instead mew, growl, yowl and chuff.