Prepatterning and Patterning of the Thalamus Along Embryonic Development of Xenopus Laevis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Downloaded from the National Database for Autism Research (NDAR)
International Journal of Molecular Sciences Article Phenotypic Subtyping and Re-Analysis of Existing Methylation Data from Autistic Probands in Simplex Families Reveal ASD Subtype-Associated Differentially Methylated Genes and Biological Functions Elizabeth C. Lee y and Valerie W. Hu * Department of Biochemistry and Molecular Medicine, The George Washington University, School of Medicine and Health Sciences, Washington, DC 20037, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-202-994-8431 Current address: W. Harry Feinstone Department of Molecular Microbiology and Immunology, y Johns Hopkins Bloomberg School of Public Health, Baltimore, MD 21205, USA. Received: 25 August 2020; Accepted: 17 September 2020; Published: 19 September 2020 Abstract: Autism spectrum disorder (ASD) describes a group of neurodevelopmental disorders with core deficits in social communication and manifestation of restricted, repetitive, and stereotyped behaviors. Despite the core symptomatology, ASD is extremely heterogeneous with respect to the severity of symptoms and behaviors. This heterogeneity presents an inherent challenge to all large-scale genome-wide omics analyses. In the present study, we address this heterogeneity by stratifying ASD probands from simplex families according to the severity of behavioral scores on the Autism Diagnostic Interview-Revised diagnostic instrument, followed by re-analysis of existing DNA methylation data from individuals in three ASD subphenotypes in comparison to that of their respective unaffected siblings. We demonstrate that subphenotyping of cases enables the identification of over 1.6 times the number of statistically significant differentially methylated regions (DMR) and DMR-associated genes (DAGs) between cases and controls, compared to that identified when all cases are combined. Our analyses also reveal ASD-related neurological functions and comorbidities that are enriched among DAGs in each phenotypic subgroup but not in the combined case group. -

Anti-Lunatic Fringe Antibody (ARG40136)
Product datasheet [email protected] ARG40136 Package: 100 μl anti-Lunatic Fringe antibody Store at: -20°C Summary Product Description Rabbit Polyclonal antibody recognizes Lunatic Fringe Tested Reactivity Hu Tested Application FACS, IHC-P, WB Host Rabbit Clonality Polyclonal Isotype IgG Target Name Lunatic Fringe Antigen Species Human Immunogen KLH-conjugated synthetic peptide corresponding to aa. 86-114 of Human Lunatic Fringe. Conjugation Un-conjugated Alternate Names O-fucosylpeptide 3-beta-N-acetylglucosaminyltransferase; EC 2.4.1.222; SCDO3; Beta-1,3-N- acetylglucosaminyltransferase lunatic fringe Application Instructions Application table Application Dilution FACS 1:10 - 1:50 IHC-P 1:50 - 1:100 WB 1:1000 Application Note * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. Positive Control K562 Calculated Mw 42 kDa Properties Form Liquid Purification Purification with Protein A and immunogen peptide. Buffer PBS and 0.09% (W/V) Sodium azide. Preservative 0.09% (W/V) Sodium azide. Storage instruction For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C or below. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. www.arigobio.com 1/3 Note For laboratory research only, not for drug, diagnostic or other use. Bioinformation Gene Symbol LFNG Gene Full Name LFNG O-fucosylpeptide 3-beta-N-acetylglucosaminyltransferase Background This gene is a member of the fringe gene family which also includes radical and manic fringe genes. -

Molecular Genetic Delineation of 2Q37.3 Deletion in Autism and Osteodystrophy: Report of a Case and of New Markers for Deletion Screening by PCR
UC Irvine UC Irvine Previously Published Works Title Molecular genetic delineation of 2q37.3 deletion in autism and osteodystrophy: report of a case and of new markers for deletion screening by PCR. Permalink https://escholarship.org/uc/item/83f0x61r Journal Cytogenetics and cell genetics, 94(1-2) ISSN 0301-0171 Authors Smith, M Escamilla, JR Filipek, P et al. Publication Date 2001 DOI 10.1159/000048775 License https://creativecommons.org/licenses/by/4.0/ 4.0 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Original Article Cytogenet Cell Genet 94:15–22 (2001) Molecular genetic delineation of 2q37.3 deletion in autism and osteodystrophy: report of a case and of new markers for deletion screening by PCR M. Smith, J.R. Escamilla, P. Filipek, M.E. Bocian, C. Modahl, P. Flodman, and M.A. Spence Department of Pediatrics, University of California, Irvine CA (USA) Abstract. We recently studied a patient who meets criteria us to determine the parental origin of the deletion in our for autistic disorder and has a 2q37 deletion. Molecular cyto- patient. DNA from 8–13 unrelated individuals was used to genetic studies were carried out using DNA isolated from 22 determine heterozygosity estimates for these markers. We re- different 2q37 mapped BACs to more precisely define the view four genes deleted in our patient – genes whose known extent of the chromosome deletion. We also analyzed 2q37 functions and sites of expression in the brain and/or bone make mapped polymorphic markers. In addition DNA sequences of them candidates for involvement in autism and/or the osteo- BACs in the deletion region were scanned to identify microsa- dystrophy observed in patients with 2q37.3 deletions. -

Segmentation in Vertebrates: Clock and Gradient Finally Joined
Downloaded from genesdev.cshlp.org on September 24, 2021 - Published by Cold Spring Harbor Laboratory Press REVIEW Segmentation in vertebrates: clock and gradient finally joined Alexander Aulehla1 and Bernhard G. Herrmann2 Max-Planck-Institute for Molecular Genetics, Department of Developmental Genetics, 14195 Berlin, Germany The vertebral column is derived from somites formed by terior (A–P) axis. Somite formation takes place periodi- segmentation of presomitic mesoderm, a fundamental cally in a fixed anterior-to-posterior sequence. process of vertebrate embryogenesis. Models on the In the chick embryo, a new somite is formed approxi- mechanism controlling this process date back some mately every 90 min, whereas in the mouse embryo, the three to four decades. Access to understanding the mo- periodicity varies, dependent on the axial position (Tam lecular control of somitogenesis has been gained only 1981). Classical embryology experiments revealed that recently by the discovery of molecular oscillators (seg- periodicity and directionality of somite formation are mentation clock) and gradients of signaling molecules, controlled by an intrinsic program set off in the cells as as predicted by early models. The Notch signaling path- they are recruited into the psm. For instance, when the way is linked to the oscillator and plays a decisive role in psm is inverted rostro–caudally, somite formation in the inter- and intrasomitic boundary formation. An Fgf8 sig- inverted region proceeds from caudal to rostral, main- naling gradient is involved in somite size control. And taining the original direction (Christ et al. 1974). More- the (canonical) Wnt signaling pathway, driven by Wnt3a, over, neither the transversal bisection nor the isolation appears to integrate clock and gradient in a global of the psm from all surrounding tissues stops the seg- mechanism controlling the segmentation process. -

Agonists Regulate the Expression of Stemness and Differentiation Genes in Brain Tumour Stem Cells
British Journal of Cancer (2012) 106, 1702–1712 & 2012 Cancer Research UK All rights reserved 0007 – 0920/12 www.bjcancer.com PPARg agonists regulate the expression of stemness and differentiation genes in brain tumour stem cells E Pestereva1, S Kanakasabai1 and JJ Bright*,1,2 1 Neuroscience Research Laboratory, Methodist Research Institute, Indiana University Health, 1800 North Capitol Avenue, Noyes Building E504C, 2 Indianapolis, IN 46202, USA; Department of Medicine, Indiana University School of Medicine, Indianapolis, IN, USA BACKGROUND: Brain tumour stem cells (BTSCs) are a small population of cancer cells that exhibit self-renewal, multi-drug resistance, and recurrence properties. We have shown earlier that peroxisome proliferator-activated receptor gamma (PPARg) agonists inhibit the expansion of BTSCs in T98G and U87MG glioma. In this study, we analysed the influence of PPARg agonists on the expression of stemness and differentiation genes in BTSCs. METHODS: The BTSCs were isolated from T98G and DB29 glioma cells, and cultured in neurobasal medium with epidermal growth factor þ basic fibroblast growth factor. Proliferation was measured by WST-1 (4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2 H-5- tetrazolio]-1,3-benzene disulphonate) and 3H thymidine uptake assays, and gene expression was analysed by quantitative reverse– transcription PCR and Taqman array. The expression of CD133, SRY box 2, and nanog homeobox (Nanog) was also evaluated by western blotting, immunostaining, and flow cytometry. 12,14 RESULTS: We found that PPARg agonists, ciglitazone and 15-deoxy-D -ProstaglandinJ2, inhibited cell viability and proliferation of þ T98G- and DB29-BTSCs. The PPARg agonists reduced the expansion of CD133 BTSCs and altered the expression of stemness and differentiation genes. -

The Regulation of Lunatic Fringe During Somitogenesis
THE REGULATION OF LUNATIC FRINGE DURING SOMITOGENESIS DISSERTATION Presented in Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy in the Graduate School of The Ohio State University By Emily T. Shifley ***** The Ohio State University 2009 Dissertation Committee: Approved by Professor Susan Cole, Advisor Professor Christine Beattie _________________________________ Professor Mark Seeger Advisor Graduate Program in Molecular Genetics Professor Michael Weinstein ABSTRACT Somitogenesis is the morphological hallmark of vertebrate segmentation. Somites bud from the presomitic mesoderm (PSM) in a sequential, periodic fashion and give rise to the rib cage, vertebrae, and dermis and muscles of the back. The regulation of somitogenesis is complex. In the posterior region of the PSM, a segmentation clock operates to organize cohorts of cells into presomites, while in the anterior region of the PSM the presomites are patterned into rostral and caudal compartments (R/C patterning). Both of these stages of somitogenesis are controlled, at least in part, by the Notch pathway and Lunatic fringe (Lfng), a glycosyltransferase that modifies the Notch receptor. To dissect the roles played by Lfng during somitogenesis, we created a novel allele that lacks cyclic Lfng expression within the segmentation clock, but that maintains expression during R/C somite patterning (Lfng∆FCE1). Lfng∆FCE1/∆FCE1 mice have severe defects in their anterior vertebrae and rib cages, but relatively normal sacral and tail vertebrae, unlike Lfng knockouts. Segmentation clock function is differentially affected by the ∆FCE1 deletion; during anterior somitogenesis the expression patterns of many clock genes are disrupted, while during posterior somitogenesis, certain clock components have recovered. R/C patterning occurs relatively normally in Lfng∆FCE1/∆FCE1 embryos, likely contributing to the partial phenotype rescue, and confirming that Lfng ii plays separate roles in the two regions of the PSM. -

Neural Development
31 October 2007 NEURAL DEVELOPMENT www.neuraldevelopment.com Analysis of Lrrn1 expression and its relationship to neuromeric boundaries during chick neural development Laura C Andreae et al. Neural Development 2007, 2:22 http://www.neuraldevelopment.com/content/2/1/22 Neural Development BioMed Central Research article Open Access Analysis of Lrrn1 expression and its relationship to neuromeric boundaries during chick neural development LauraCAndreae1,2, Daniela Peukert1, Andrew Lumsden1 and Jonathan D Gilthorpe*1 Address: 1MRC Centre for Developmental Neurobiology, King's College London, New Hunt's House, Guy's Campus, London, UK, SE1 1UL and 2Department of Neurophysiology, National Institute for Medical Research, The Ridgeway, Mill Hill, London, UK, NW7 1AA Email: Laura C Andreae - [email protected]; Daniela Peukert - [email protected]; Andrew Lumsden - [email protected]; Jonathan D Gilthorpe* - [email protected] * Corresponding author Published: 31 October 2007 Received: 26 March 2007 Accepted: 31 October 2007 Neural Development 2007, 2:22 doi:10.1186/1749-8104-2-22 This article is available from: http://www.neuraldevelopment.com/content/2/1/22 © 2007 Andreae et al.; licensee BioMed Central Ltd. This is an open access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Abstract Background: The Drosophila leucine-rich repeat proteins Tartan (TRN) and Capricious (CAPS) mediate cell affinity differences during compartition of the wing imaginal disc. This study aims to identify and characterize the expression of a chick orthologue of TRN/CAPS and examine its potential function in relation to compartment boundaries in the vertebrate central nervous system. -

PDF Download
Review Xatzipsalti Maria et al. Congenital Hypopituitarism: Various Genes, … Horm Metab Res 2018; 00: 00–00 Congenital Hypopituitarism: Various Genes, Various Phenotypes Authors Maria Xatzipsalti1, 2, Antonis Voutetakis1, Lela Stamoyannou2, George P. Chrousos1, Christina Kanaka-Gantenbein1 Affiliations ABSTRacT 1 Division of Endocrinology, Diabetes and Metabolism, The ontogenesis and development of the pituitary gland is a First Department of Pediatrics, Medical School, National highly complex process that depends on a cascade of transcrip- and Kapodistrian University of Athens, “Aghia Sofia” tion factors and signaling molecules. Spontaneous mutations Children's Hospital, Athens, Greece and transgenic murine models have demonstrated a role for 2 First Department of Pediatrics, “Aglaia Kyriakou” many of these factors, including HESX1, PROP1, PIT1, LHX3, Children's Hospital, Athens, Greece LHX4, SOX2, SOX3, OTX2, PAX6, FGFR1, SHH, GLI2, and FGF8 in the etiology of congenital hypopituitarism. Genetic muta- Key words tions in any of these factors can lead to congenital hypopitui- pituitary, combined pituitary hormone deficiency, congenital tarism, which is characterized by the deficiency in one or more hypopituitarism, transcription factors, syndromic hypopitui- pituitary hormones. The phenotype can be highly variable, tarism, non-syndromic hypopituitarism consisting of isolated hypopituitarism or more complex disor- ders. The same phenotype can be attributed to different gene received 27.03.2018 mutations; while a given gene mutation can -

FOS Rescues Neuronal Differentiation of Sox2-Deleted Neural Stem Cells by Genome-Wide Regulation of Common SOX2 and AP1(FOS-JUN) Target Genes
cells Article FOS Rescues Neuronal Differentiation of Sox2-Deleted Neural Stem Cells by Genome-Wide Regulation of Common SOX2 and AP1(FOS-JUN) Target Genes Miriam Pagin 1,† , Mattias Pernebrink 2,3,†, Mattia Pitasi 1, Federica Malighetti 1, Chew-Yee Ngan 4, Sergio Ottolenghi 1, Giulio Pavesi 5, Claudio Cantù 2,3,* and Silvia K. Nicolis 1,* 1 Department of Biotechnology and Biosciences, University of Milano-Bicocca, Piazza della Scienza 2, 20126 Milano, Italy; [email protected] (M.P.); [email protected] (M.P.); [email protected] (F.M.); [email protected] (S.O.) 2 Wallenberg Centre for Molecular Medicine, Linköping University, SE-581 83 Linköping, Sweden; [email protected] 3 Department of Biomedical and Clinical Sciences, Division of Molecular Medicine and Virology, Faculty of Medicine and Health Sciences, Linköping University, SE-581 83 Linköping, Sweden 4 The Jackson Laboratory for Genomic Medicine, Farmington, CT 06032, USA; [email protected] 5 Department of Biosciences, University of Milano, Via Celoria 26, 20134 Milano, Italy; [email protected] * Correspondence: [email protected] (C.C.); [email protected] (S.K.N.) † Joint first authors. Abstract: The transcription factor SOX2 is important for brain development and for neural stem Citation: Pagin, M.; Pernebrink, M.; cells (NSC) maintenance. Sox2-deleted (Sox2-del) NSC from neonatal mouse brain are lost after Pitasi, M.; Malighetti, F.; Ngan, C.-Y.; few passages in culture. Two highly expressed genes, Fos and Socs3, are strongly downregulated in Ottolenghi, S.; Pavesi, G.; Cantù, C.; Sox2-del NSC; we previously showed that Fos or Socs3 overexpression by lentiviral transduction fully Nicolis, S.K. -
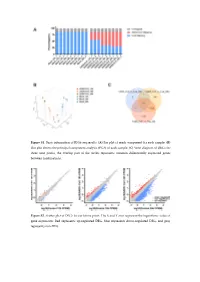
Figure S1. Basic Information of RNA-Seq Results. (A) Bar Plot of Reads Component for Each Sample
Figure S1. Basic information of RNA-seq results. (A) Bar plot of reads component for each sample. (B) Dot plot shows the principal component analysis (PCA) of each sample. (C) Venn diagram of DEGs for three time points, the overlap part of the circles represents common differentially expressed genes between combinations. Figure S2. Scatter plot of DEGs for each time point. The X and Y axes represent the logarithmic value of gene expression. Red represents up-regulated DEG, blue represents down-regulated DEG, and gray represents non-DEG. Table S1. Primers used for quantitative real-time PCR analysis of DEGs. Gene Primer Sequence Forward 5’-CTACGAGTGGATGGTCAAGAGC-3’ FOXO1 Reverse 5’-CCAGTTCCTTCATTCTGCACACG-3’ Forward 5’-GACGTCCGGCATCAGAGAAA-3’ IRS2 Reverse 5’-TCCACGGCTAATCGTCACAG-3’ Forward 5’-CACAACCAGGACCTCACACC-3’ IRS1 Reverse 5’-CTTGGCACGATAGAGAGCGT-3’ Forward 5’-AGGATACCACTCCCAACAGACCT-3’ IL6 Reverse 5’-CAAGTGCATCATCGTTGTTCATAC-3’ Forward 5’-TCACGTTGTACGCAGCTACC-3’ CCL5 Reverse 5’-CAGTCCTCTTACAGCCTTTGG-3’ Forward 5’-CTGTGCAGCCGCAGTGCCTACC-3’ BMP7 Reverse 5’-ATCCCTCCCCACCCCACCATCT-3’ Forward 5’-CTCTCCCCCTCGACTTCTGA-3’ BCL2 Reverse 5’-AGTCACGCGGAACACTTGAT-3’ Forward 5’-CTGTCGAACACAGTGGTACCTG-3’ FGF7 Reverse 5’-CCAACTGCCACTGTCCTGATTTC-3’ Forward 5’-GGGAGCCAAAAGGGTCATCA-3’ GAPDH Reverse 5’-CGTGGACTGTGGTCATGAGT-3’ Supplementary material: Differentially expressed genes log2(SADS-CoV_12h/ Qvalue (SADS-CoV _12h/ Gene Symbol Control_12h) Control_12h) PTGER4 -1.03693 6.79E-04 TMEM72 -3.08132 3.66E-04 IFIT2 -1.02918 2.11E-07 FRAT2 -1.09282 4.66E-05 -

Mouse Thalamic Differentiation: Gli-Dependent Pattern and Gli-Independent Prepattern
ORIGINAL RESEARCH ARTICLE published: 22 February 2012 doi: 10.3389/fnins.2012.00027 Mouse thalamic differentiation: Gli-dependent pattern and Gli-independent prepattern Roberta Haddad-Tóvolli 1, Michael Heide 1, Xunlei Zhou 1, Sandra Blaess 2 and Gonzalo Alvarez-Bolado1* 1 Department of Neuroanatomy, Institute of Anatomy and Cell Biology, University of Heidelberg, Heidelberg, Germany 2 Institute of Reconstructive Neurobiology, Life and Brain Center, University of Bonn, Bonn, Germany Edited by: Sonic hedgehog (Shh) signaling is essential for thalamic development.The Gli transcription Steffen Scholpp, Karlsruhe Institute of factors act downstream of Shh – while Gli2 is the major activator (GliA), Gli3 acts primarily Technology, Germany as a repressor (GliR). The thalamus is remarkable among dorsal structures because of its Reviewed by: Tomomi Shimogori, RIKEN Brain proximity to the mid-diencephalic organizer, a unique dorsal Shh source. This lends com- Science Institute, Japan plexity to the interactions between Shh, Gli2, and Gli3, suggesting the presence of a dorsal David J. Price, University of Gli activator which elsewhere is found only ventrally, and making the dissection of thala- Edinburgh, UK mic Gli functions particularly interesting. A current model based on mutant phenotypes in *Correspondence: telencephalon and midbrain postulates a degree of reciprocal antagonism of Shh and Gli3 Gonzalo Alvarez-Bolado, Department of Neuroanatomy, Institute of in dorsal brain regions. To approach the role of Gli factors in thalamic specification we first Anatomy and Cell Biology, University analyzed mice deficient in Gli2 or Gli3.InGli2 mutants, the thalamus is small and poorly of Heidelberg, Im Neuenheimer Feld differentiated with the exception of the medial and intralaminar nuclei which, in contrast, 307, 69120 Heidelberg, Germany. -

Zeb2 Regulates Cell Fate at the Exit from Epiblast State In
View metadata, citation and similar papers at core.ac.uk EMBRYONIC STEM CELLS/INDUCEDbrought to you by CORE PLURIPOTENT STEMprovidedC byELLS Ghent University Academic Bibliography Zeb2 Regulates Cell Fate at the Exit from Epiblast State in Mouse Embryonic Stem Cells aDepartment of Development and Regeneration, KU a a,b c,d,e,f a b Leuven, Leuven 3000, AGATA STRYJEWSKA, RUBEN DRIES, TIM PIETERS, GRIET VERSTAPPEN, ANDREA CONIDI, a a a b,g Belgium; bDepartment of Cell KATHLEEN CODDENS, ANNICK FRANCIS, LIEVE UMANS, WILFRED F. J. VAN IJCKEN, g d,e h b c,d,e,i Biology, Center for Biomics, GEERT BERX, LEO A. VAN GRUNSVEN, FRANK G. GROSVELD, STEVEN GOOSSENS, c,d,i a,b Erasmus University Medical JODY J. HAIGH, DANNY HUYLEBROECK Center, Rotterdam 3015 CN, c The Netherlands; VIB Key Words. Cell differentiation • DNA-methylation • Embryonic stem cells • Pluripotent stem cells Inflammation Research • Repressors • RNA-sequencing • Transcription factors • Transcriptom Center (IRC), Unit Vascular Cell Biology, dDepartment of Biomedical Molecular Biology, eVIB-IRC, Unit ABSTRACT Molecular and Cellular In human embryonic stem cells (ESCs) the transcription factor Zeb2 regulates neuroectoderm ver- Oncology, Ghent University, sus mesendoderm formation, but it is unclear how Zeb2 affects the global transcriptional regulato- f Ghent 9052, Belgium; Center ry network in these cell-fate decisions. We generated Zeb2 knockout (KO) mouse ESCs, subjected for Medical Genetics, Ghent them as embryoid bodies (EBs) to neural and general differentiation and carried out temporal University Hospital, Ghent h RNA-sequencing (RNA-seq) and reduced representation bisulfite sequencing (RRBS) analysis in neu- 9000, Belgium; Department ral differentiation.