Package Leaflet: Information for the Patient HALDOL® Decanoate 50
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

HALDOL Brand of Haloperidol Injection (For Immediate Release) WARNING Increased Mortality in Elderly Patients with Dementia
HALDOL® brand of haloperidol injection (For Immediate Release) WARNING Increased Mortality in Elderly Patients with Dementia-Related Psychosis Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. HALDOL Injection is not approved for the treatment of patients with dementia-related psychosis (see WARNINGS). DESCRIPTION Haloperidol is the first of the butyrophenone series of major antipsychotics. The chemical designation is 4-[4-(p-chlorophenyl)-4-hydroxypiperidino] 4’-fluorobutyrophenone and it has the following structural formula: HALDOL (haloperidol) is available as a sterile parenteral form for intramuscular injection. The injection provides 5 mg haloperidol (as the lactate) and lactic acid for pH adjustment between 3.0 – 3.6. -

Prochlorperazine 5Mg Tablets
Package leaflet: Information for the patient Prochlorperazine 5mg tablets Read all of this leaflet carefully before you start taking this • the person is a child. This is because children may develop unusual face and body medicine, because it contains important information for you. movements (dystonic reactions) • Keep this leaflet. You may need to read it again. • you are diabetic or have high levels of sugar in your blood (hyperglycaemia). Your doctor • If you have any further questions, ask your doctor or pharmacist. may want to monitor you more closely. • This medicine has been prescribed for you only. Do not pass it on to If you are not sure if any of the above apply to you, talk to your doctor or pharmacist before others. It may harm them, even if their signs of illness are the same taking Prochlorperazine Tablets. as yours. Other medicines and Prochlorperazine tablets • If you get any side effects, talk to your doctor or pharmacist. Tell your doctor or pharmacist if you are taking, have recently taken or might take any This includes any possible side effects not listed in this leaflet. other medicines. This includes medicines you buy without a prescription, including herbal See section 4. medicines. This is because Prochlorperazine Tablets can affect the way some other medicines work. What is in this leaflet: Also some medicines can affect the way Prochlorperazine Tablets work. 1 What Prochlorperazine tablets are and what they In particular, tell your doctor if you are taking any of the following: • medicines to help you sleep -

AMANTADIP\Dle: on Nemolebtic-II[WDUCED CATALEPSY UV the RAT
J. Fac. Plmnz. Istanbul lstanbul Ecz. Fak. Mec. 29, 1 (1993) 29, 1 (1993) THE EFFECT OF CYPROHEPT~~AWEb AMANTADIP\dlE: ON NEmOLEBTIC-II[WDUCED CATALEPSY UV THE RAT SUMMARY Catalepsy is not a unitary phenomenon. With respect to biochemical mechanisms and neurotransmitter sytems, the origin of this behavioural response varies according to different substances which cause catalepsy. For example, the catdeptogenic effect of ne- uroleptics has been related to the blockage of striatal doparnine receptors. Neurophysio- logical and biochemical data have shown that a mechanism caused by gama-aminobuty- ric acid and serotonin have been effective in the proper functioning of the dopaminergic nigrosbriatal tract. We have studied the effects of cyproheptadine, a serotonin antagonist, and of amantadin which is used in the treatment of Parkinson syndrome on the catalepsy indu- ced by trifluoperazine, a phenothiazin compound, and pimozid, a drug used as a neuro- leptic. It was observed that when rats were pretreated with cyproheptadine as well as amantadiie, the catalepsy induced by the above neuroleptic drugs was attenuated. The results of our study show that substances which have an effect on the seroto- nergic and dopaminergic systems also play a role on cases of catalepsy induced by the neuroleptic drugs mentioned above. (*) Faculty of Pharmacy, Depament of Pharmacology, University of Istanbul, 31152, Istanbul, Turkey Katalepsi bir tek nedene bagh OWortaya qllian bit durum degildir. Biyokimya- sal ve norotransmitter sistemler agsmdan bu davran~glnsebebi, katalepsi olughYan qe- gitli maddeler iqin fad&&. Omegin noroleptiklerin kataleptojenik etkisi striatal dopa- min reseptorlerinin blokaj~nabajjlanmqtx. Niirofizyolojik ve biyokimyasal veriler do- pamine jik nigro-striatal sistemin duzenli $&$masmda gma-aminobutirik asid ve sero- tonin'e bagh olan bir mekanizmmn etkili oldugunu gostermigtir. -

Papers and Originals
BRITISH MEDICAL JOURNAL 19 FEBRUARY 1972 463 PAPERS AND ORIGINALS Drug Interaction: Inhibitory Effect of Neuroleptics on Metabolism of Tricyclic Antidepressants in Man LARS F. GRAM, KERSTIN FREDRICSON OVERO British Medical Journal, 1972, 1, 463-465 excretion (Anders, 1971). There are relatively few reports on interaction concerning neuroleptics or tricyclic antidepressants. Summary Chlorpromazine has both stimulatory and inhibitory effects on the metabolism of hexobarbitone in Total urinary excretion of radioactivity after oral or experimental animals of a test of (Riimke and Bout, 1960-1). Furthermore, chlorpromazine intravenous administration dose '4C-imi- accelerates the metabolism pramine was measured in were tested ofmeprobamate (Kato and Vassanelli, eight patients. They 1962). Tricyclic antidepressants have been shown to inhibit the before, during, and after treatment with neuroleptics. metabolism Excretion diminished while the were of tremorine and oxotremorine in rats in vitro and patients being in vivo (Hammer and Sjoqvist, 1967). It has also been found treated with perphenazine, haloperidol, or chlorproma- that zine, not treatment. desipramine hydrochloride inhibits the metabolism of though during flupenthixol amphetamine in isolated, perfused rat liver (Dingell and Bass, Total urinary excretion of radioactivity and plasma 1969). Pretreatment levels of metabolites and were measured with phenobarbitone decreased steady-state unchanged drug plasma levels of desipramine and nortriptyline in man (Sjoqvist in five patients after a test dose of 14C-nortriptyline. Each et was before and al. 1968). These findings were later confirmed in a twin study patient tested again during perphenazine (Alexanderson et al., 1969). Forrest et treatment. In all patients perphenazine treatment caused: al. (1970) found increased (1) decrease of total urinary excretion, (2) decreased urinary excretion of chlorpromazine metabolites when patients plasma level of metabolites, and (3) increased plasma were given additional treatment with phenobarbitone. -

203050Orig1s000
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 203050Orig1s000 LABELING HIGHLIGHTS OF PRESCRIBING INFORMATION ------------------------------CONTRAINDICATIONS-------------------------------- These highlights do not include all the information needed to use Hypersensitivity to the drug or any of its components (4) PALONOSETRON HYDROCHLORIDE INJECTION safely and effectively. See full prescribing information for PALONOSETRON ------------------------WARNINGS AND PRECAUTIONS------------------------- HYDROCHLORIDE INJECTION. • Hypersensitivity reactions, including anaphylaxis, have been reported with or without known hypersensitivity to other selective 5-HT3 receptor antagonists PALONOSETRON HYDROCHLORIDE injection, for intravenous (5.1) use • Serotonin syndrome has been reported with 5-HT3 receptor antagonists alone Initial U.S. Approval: 2003 but particularly with concomitant use of serotonergic drugs (5.2, 7.1) -------------------------INDICATIONS AND USAGE------------------------ --------------------------------ADVERSE REACTIONS------------------------------- Palonosetron Hydrochloride (HCl) Injection is a serotonin-3 (5-HT3) The most common adverse reactions in chemotherapy-induced nausea and receptor antagonist indicated in adults for: vomiting (≥5%) are headache and constipation (6.1) • Moderately emetogenic cancer chemotherapy -- prevention of acute and delayed nausea and vomiting associated with initial and repeat The most common adverse reactions in postoperative nausea and vomiting (≥ courses (1.1) 2%) are QT prolongation, -

Haloperidol As Prophylactic Treatment for Postoperative
Document downloaded from http://http://www.revcolanest.com.co, day 15/02/2013. This copy is for personal use. Any transmission of this document by any media or format is strictly prohibited. r e v c o l o m b a n e s t e s i o l . 2 0 1 3;4 1(1):34–43 Revista Colombiana de Anestesiología Colombian Journal of Anesthesiology www.revcolanest.com.co Review Haloperidol as prophylactic treatment for postoperative ଝ nausea and vomiting: Systematic literature review ∗ Catalina Chaparro , Diego Moreno, Verónica Ramírez, Angélica Fajardo, David González, Alejandra Sanín, Reinaldo Grueso Anesthesiology Service, Hospital Universitario San Ignacio, Bogotá, Colombia a r t i c l e i n f o a b s t r a c t Article history: Introduction: The effectiveness of haloperidol for the prophylaxis of postoperative nausea Received 3 July 2012 and vomiting (PONV) has been proven in prior trials summarized by Buttner in 2004. New Accepted 24 July 2012 evidence has surfaced since then. Our objective is thus to update the current knowledge on the topic. A systematic review and a meta-analysis were performed, in order to determine the effectiveness and safety of the use of haloperidol as prophylaxis for PONV. Keywords: Methodology: The systematic search, the selection of relevant articles, the extraction of data, Haloperidol the critical analysis of the primary studies, the comparisons and analyses were all based on Vomiting the recommendations of the Cochrane Collaboration and using RevMan5 software. Nausea Results: Ten controlled clinical trials published between 1962 and 2010, that included 2,711 Clinical trials patients, met the selection criteria. -

Delusional Parasitosis Mimicking Cutaneous Infestation in Elderly Patients
LESSONS FROM PRACTICE LESSONS FROM PRACTICE Delusional parasitosis mimicking cutaneous infestation in elderly patients Clinical records Patient 1 Patient 3 An 88-year-old man gave a 12-month history of seeing insects attacking An 81-year-old woman was referred with a persistent belief that his legs and crawling along the floor of his house. He described these she had scabies and lice infestation of her eyes, nose, arms and insects as 4 cm long, black and white bugs with beaks, which pecked anus. This resulted in her persistently washing her clothes and at hisThe legs, Medical causing Journal wounds. of He Australia often felt ISSN:a sharp 0025-729X stinging sensation 18 herself and reporting the retirement village where she lived to the heraldingAugust their 2003 presence. 179 4 209-210 He also had burning pain in both legs below Health Department. She had received anti-scabies treatment the knees. He had had his house fumigated twice in the previous year empirically. ©The Medical Journal of Australia 2003 www.mja.com.au andLessons put various from chemicals practice across his doorways and bed to ward off the She had a long history of severe depression after the death of her bugs. He described no other hallucinations or delusions. husband, for which she took doxepin. She had paranoid ideation He was not using any regular medications and had never been a about her neighbours and saw things crawling down the walls, consumer of alcohol. He lived alone and managed all activities of and had moved residences several times to avoid these problems. -
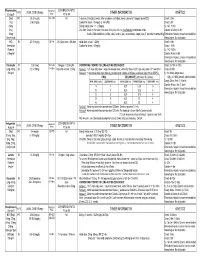
Medication Conversion Chart
Fluphenazine FREQUENCY CONVERSION RATIO ROUTE USUAL DOSE (Range) (Range) OTHER INFORMATION KINETICS Prolixin® PO to IM Oral PO 2.5-20 mg/dy QD - QID NA ↑ dose by 2.5mg/dy Q week. After symptoms controlled, slowly ↓ dose to 1-5mg/dy (dosed QD) Onset: ≤ 1hr 1mg (2-60 mg/dy) Caution for doses > 20mg/dy (↑ risk EPS) Cmax: 0.5hr 2.5mg Elderly: Initial dose = 1 - 2.5mg/dy t½: 14.7-15.3hr 5mg Oral Soln: Dilute in 2oz water, tomato or fruit juice, milk, or uncaffeinated carbonated drinks Duration of Action: 6-8hr 10mg Avoid caffeinated drinks (coffee, cola), tannics (tea), or pectinates (apple juice) 2° possible incompatibilityElimination: Hepatic to inactive metabolites 5mg/ml soln Hemodialysis: Not dialyzable HCl IM 2.5-10 mg/dy Q6-8 hr 1/3-1/2 po dose = IM dose Initial dose (usual): 1.25mg Onset: ≤ 1hr Immediate Caution for doses > 10mg/dy Cmax: 1.5-2hr Release t½: 14.7-15.3hr 2.5mg/ml Duration Action: 6-8hr Elimination: Hepatic to inactive metabolites Hemodialysis: Not dialyzable Decanoate IM 12.5-50mg Q2-3 wks 10mg po = 12.5mg IM CONVERTING FROM PO TO LONG-ACTING DECANOATE: Onset: 24-72hr (4-72hr) Long-Acting SC (12.5-100mg) (1-4 wks) Round to nearest 12.5mg Method 1: 1.25 X po daily dose = equiv decanoate dose; admin Q2-3wks. Cont ½ po daily dose X 1st few mths Cmax: 48-96hr 25mg/ml Method 2: ↑ decanoate dose over 4wks & ↓ po dose over 4-8wks as follows (accelerate taper for sx of EPS): t½: 6.8-9.6dy (single dose) ORAL DECANOATE (Administer Q 2 weeks) 15dy (14-100dy chronic administration) ORAL DOSE (mg/dy) ↓ DOSE OVER (wks) INITIAL DOSE (mg) TARGET DOSE (mg) DOSE OVER (wks) Steady State: 2mth (1.5-3mth) 5 4 6.25 6.25 0 Duration Action: 2wk (1-6wk) Elimination: Hepatic to inactive metabolites 10 4 6.25 12.5 4 Hemodialysis: Not dialyzable 20 8 6.25 12.5 4 30 8 6.25 25 4 40 8 6.25 25 4 Method 3: Admin equivalent decanoate dose Q2-3wks. -

Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients
© Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 | Fax 503-947-1119 Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients Research Questions: 1. How many schizophrenia patients are prescribed recommended first-line second-generation treatments for schizophrenia? 2. How many schizophrenia patients switch to an injectable antipsychotic after stabilization on an oral antipsychotic? 3. How many schizophrenia patients are prescribed 2 or more concomitant antipsychotics? 4. Are claims for long-acting injectable antipsychotics primarily billed as pharmacy or physician administered claims? 5. Does adherence to antipsychotic therapy differ between patients with claims for different routes of administration (oral vs. long-acting injectable)? Conclusions: In total, 4663 schizophrenia patients met inclusion criteria, and approximately 14% of patients (n=685) were identified as treatment naïve without claims for antipsychotics in the year before their first antipsychotic prescription. Approximately 45% of patients identified as treatment naïve had a history of remote antipsychotic use, but it is unclear if antipsychotics were historically prescribed for schizophrenia. Oral second-generation antipsychotics which are recommended as first-line treatment in the MHCAG schizophrenia algorithm were prescribed as initial treatment in 37% of treatment naive patients and 28% of all schizophrenia patients. Recommended agents include risperidone, paliperidone, and aripiprazole. Utilization of parenteral antipsychotics was limited in patients with schizophrenia. Overall only 8% of patients switched from an oral to an injectable therapy within 6 months of their first claim. Approximately, 60% of all schizophrenia patients (n=2512) had claims for a single antipsychotic for at least 12 continuous weeks and may be eligible to transition to a long-acting injectable antipsychotic. -

Schizophrenia Care Guide
August 2015 CCHCS/DHCS Care Guide: Schizophrenia SUMMARY DECISION SUPPORT PATIENT EDUCATION/SELF MANAGEMENT GOALS ALERTS Minimize frequency and severity of psychotic episodes Suicidal ideation or gestures Encourage medication adherence Abnormal movements Manage medication side effects Delusions Monitor as clinically appropriate Neuroleptic Malignant Syndrome Danger to self or others DIAGNOSTIC CRITERIA/EVALUATION (PER DSM V) 1. Rule out delirium or other medical illnesses mimicking schizophrenia (see page 5), medications or drugs of abuse causing psychosis (see page 6), other mental illness causes of psychosis, e.g., Bipolar Mania or Depression, Major Depression, PTSD, borderline personality disorder (see page 4). Ideas in patients (even odd ideas) that we disagree with can be learned and are therefore not necessarily signs of schizophrenia. Schizophrenia is a world-wide phenomenon that can occur in cultures with widely differing ideas. 2. Diagnosis is made based on the following: (Criteria A and B must be met) A. Two of the following symptoms/signs must be present over much of at least one month (unless treated), with a significant impact on social or occupational functioning, over at least a 6-month period of time: Delusions, Hallucinations, Disorganized Speech, Negative symptoms (social withdrawal, poverty of thought, etc.), severely disorganized or catatonic behavior. B. At least one of the symptoms/signs should be Delusions, Hallucinations, or Disorganized Speech. TREATMENT OPTIONS MEDICATIONS Informed consent for psychotropic -

(Orion) 5 Mg Tablets Buspirone (Orion) 10 Mg Tablets
NEW ZEALAND DATA SHEET 1. PRODUCT NAME Buspirone (Orion) 5 mg tablets Buspirone (Orion) 10 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 5 mg buspirone hydrochloride. Each tablet contains 10 mg buspirone hydrochloride. Excipient with known effect: 5 mg tablet: Each tablet contains 59.5 mg lactose (as monohydrate) 10 mg tablet: Each tablet contains 118.9 mg lactose (as monohydrate). For the full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM Tablet. 5 mg tablet: White or almost white, oval tablets debossed with ‘ORN 30’ on one side and a score on the other side. The tablet can be divided into equal doses. 10 mg tablet: White or almost white, oval tablets debossed with ‘ORN 31’ on one side and a score on the other side. The tablet can be divided into equal doses. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Buspirone hydrochloride is indicated for the management of anxiety with or without accompanying depression in adults. Buspirone hydrochloride is indicated for the management of anxiety disorders or the short- term relief of symptoms of anxiety with or without accompanying depression. 4.2 Posology and method of administration The usual starting dose is 5 mg given three times daily. This may be titrated according to the needs of the patient and the daily dose increased by 5 mg increments every two or three days depending upon the therapeutic response to a maximum daily dose of 60 mg. After dosage titration the usual daily dose will be 20 to 30 mg per day in divided doses. -

MODERN INDICATIONS for the USE of OPIPRAMOL Krzysztof Krysta1, Sławomir Murawiec2, Anna Warchala1, Karolina Zawada3, Wiesław J
Psychiatria Danubina, 2015; Vol. 27, Suppl. 1, pp 435–437 Conference paper © Medicinska naklada - Zagreb, Croatia MODERN INDICATIONS FOR THE USE OF OPIPRAMOL Krzysztof Krysta1, Sławomir Murawiec2, Anna Warchala1, Karolina Zawada3, Wiesław J. Cubała4, Mariusz S. Wiglusz4, Katarzyna Jakuszkowiak-Wojten4, Marek Krzystanek5 & Irena Krupka-Matuszczyk1 1Department of Psychiatry and Psychotherapy, Medical University of Silesia, Katowice, Poland 2“Dialogue” Therapy Centre, Warsaw, Poland 3Department of Pneumonology, Medical University of Silesia, Katowice, Poland 4Department of Psychiatry, Medical University of Gdańsk, Gdańsk, Poland 5Department of Rehabilitation Psychiatry, Medical University of Silesia, Katowice, Poland SUMMARY Opipramol is considered as a pharmacological agent that does not fit the classification taking into account the division of antidepressants, antipsychotics and anxiolytics. It has a structure related to tricyclic antidepressants but it has a different mechanism of action, i.e. binding to sigma1 and to sigma2 sites. It has been regarded as an effective drug in general anxiety disorders together with other agents like SSRI`s, SNRI`s, buspirone and pregabalin for many years. It can however also be indicated in other conditions, e.g. it may be used as a premedication in the evening prior to surgery, positive results are also observed in psychopharmacological treatment with opipramol in somatoform disorders, symptoms of depression can be significantly reduced in the climacteric syndrome. The latest data from literature present also certain dangers and side effects, which may result due to opipramol administration. Mania may be induced not only in bipolar patients treated with opipramol, but it can be an adverse drug reaction in generalized anxiety disorder. This analysis shows however that opipramol is an important drug still very useful in different clinical conditions.