History of the Discovery of Sulfaquinoxaline As a Coccidiostat
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
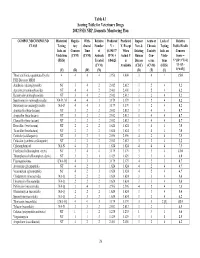
2002 FSIS National Residue Program, Section 4
Table 4.1 Scoring Table for Veterinary Drugs 2002 FSIS NRP, Domestic Monitoring Plan COMPOUND/COMPOUND Historical Regula- With- Relative Predicted Predicted Impact Acute or Lack of Relative CLASS Testing tory drawal Number V = V, Except New & Chronic Testing Public Health Info. on Concern Time of (0.19437* When Existing Toxicity Info. on Concern Violations (CVM) (CVM) Animals R*N) + Actual V Human Con- Viola- Score = (FSIS) Treated 0.84625 is Disease cerns tions V*[(D+3*T)/4] (CVM) Available (CDC) (CVM) (FSIS) *{1+[(L- (V) (R) (W) (N) (D) (T) (L) 1)*0.05]} Those antibiotics quantitated by the 4 4 4 4 3.956 4.000 3 4 1 15.0 FSIS Bioassay MRM Amikacin (aminoglycoside) NT 3 4 2 2.012 2.012 3 2 4 5.2 Apramycin (aminoglycoside) NT 4 4 2 2.401 2.401 3 2 4 6.2 Kanamycin (aminoglycoside) NT 3 4 2 2.012 2.012 3 2 4 5.2 Spectinomycin (aminoglycoside) NA-D, M 4 4 3 3.179 3.179 3 2 4 8.2 Streptomycin (aminoglycoside) NA-D 4 4 3 3.179 3.179 3 2 4 8.2 Amoxicillin (beta-lactam) NT 3 2 2 2.012 2.012 3 4 4 8.7 Ampicillin (beta-lactam) NT 3 2 2 2.012 2.012 3 4 4 8.7 Cloxacillin (beta-lactam) NT 3 2 2 2.012 2.012 3 4 4 8.7 Hetacillin (beta-lactam) NT 2 2 2 1.624 1.624 3 4 4 7.0 Ticarcillin (beta-lactam) NT 2 2 2 1.624 1.624 3 4 4 7.0 Ceftiofur (cefalosporin) NT 3 2 3 2.596 2.596 4 2 4 7.5 Cefazolin (synthetic cefalosporin) NT 3 2 2 2.012 2.012 3 2 4 5.2 Chloramphenicol NA-N 4 2 1 1.624 1.624 4 4 4 7.5 Florfenicol (chloramphen. -

Prevalence of Cryptosporidium Spp. \(Eucoccidiorida
Article available at http://www.parasite-journal.org or http://dx.doi.org/10.1051/parasite/2007144335 PREVALENCE OF CRYPTOSPORIDIUM SPP. (EUCOCCIDIORIDA: CRYPTOSPORIIDAE) IN SEVEN SPECIES OF FARM ANIMALS IN TUNISIA SOLTANE R.*, GUYOT K.**, DEI-CAS E.** & AYADI A.* Summary: Résumé : PRÉVALENCE DE CRYPTOSPORIDIUM SPP. (EUCOCCIDIORIDA : CRYPTOSPORIIDAE) CHEZ SEPT ESPÈCES D’ANIMAUX DE FERME EN TUNISIE 1,001 faecal samples were obtained from 89 sheep (lambs and adult), 184 goats, 190 horses, 178 rabbits, 110 camels, 1001 prélèvements fécaux ont été obtenus à partir de 89 moutons, 200 broiler chicken and 50 turkeys housed in farms from different 184 chèvres, 190 chevaux, 178 lapins, 110 chameaux, localities in Tunisia. All samples were analysed for 200 poulets et 50 dindes élevés dans des fermes de différentes Cryptosporidium oocysts by microscopic examination of smears localités en Tunisie. Tous les prélèvements ont été analysés pour la stained by modified Ziehl Neelsen technique. The parasite was recherche de Cryptosporidium par examen microscopique des detected in ten lambs and adult sheep (11.2 %) and nine broiler frottis colorés au Ziehl Neelsen modifié. Le parasite a été détecté chicken (4.5 %). Molecular characterization, performed in four chez dix ovins (11,2 %) et neuf poulets (4,5 %). La caractérisation animals, identified C. bovis in three lambs and C. meleagridis in moléculaire réalisée pour quatre isolats a identifié C. bovis chez one broiler chicken. This work is the first report on trois agneaux et C. meleagridis chez un poulet. Ce travail est le Cryptosporidium in farm animals in Tunisia. premier rapport sur Cryptosporidium chez des animaux de ferme en Tunisie. -

(Alveolata) As Inferred from Hsp90 and Actin Phylogenies1
J. Phycol. 40, 341–350 (2004) r 2004 Phycological Society of America DOI: 10.1111/j.1529-8817.2004.03129.x EARLY EVOLUTIONARY HISTORY OF DINOFLAGELLATES AND APICOMPLEXANS (ALVEOLATA) AS INFERRED FROM HSP90 AND ACTIN PHYLOGENIES1 Brian S. Leander2 and Patrick J. Keeling Canadian Institute for Advanced Research, Program in Evolutionary Biology, Departments of Botany and Zoology, University of British Columbia, Vancouver, British Columbia, Canada Three extremely diverse groups of unicellular The Alveolata is one of the most biologically diverse eukaryotes comprise the Alveolata: ciliates, dino- supergroups of eukaryotic microorganisms, consisting flagellates, and apicomplexans. The vast phenotypic of ciliates, dinoflagellates, apicomplexans, and several distances between the three groups along with the minor lineages. Although molecular phylogenies un- enigmatic distribution of plastids and the economic equivocally support the monophyly of alveolates, and medical importance of several representative members of the group share only a few derived species (e.g. Plasmodium, Toxoplasma, Perkinsus, and morphological features, such as distinctive patterns of Pfiesteria) have stimulated a great deal of specula- cortical vesicles (syn. alveoli or amphiesmal vesicles) tion on the early evolutionary history of alveolates. subtending the plasma membrane and presumptive A robust phylogenetic framework for alveolate pinocytotic structures, called ‘‘micropores’’ (Cavalier- diversity will provide the context necessary for Smith 1993, Siddall et al. 1997, Patterson -

NEW CLASSIFICATION for Toxoplasma Gondii
LETTER TO THE EDITOR NEW CLASSIFICATION FOR Toxoplasma gondii Claudio Bruno Silva de Oliveira Dear Editor, I have been following the latest publications in the field of parasitology and have noticed that, despite the changes in the group that host the parasite Toxoplasma gondii that have been suggested since 2012 (Adl et al. 2012), many articles in several journals have not been updated (Liempi et al. 2014; Ning et al. 2015; Lorenzi et al. 2016). This can be explained by a certain protectionism regarding the form that has been used for several years. However, it is important to consider that this can also be due to some unfamiliarity with the current classification of eukaryotes and protozoa. Classically this protozoan is classified within the Protista kingdom, Apicomplexa phylum, Sporozoasida class, Eucoccidiorida order, Sarcocystidae family, and Toxoplasma genus (Current et al. 1990). This classification has been used for many decades, and it is well accepted by research groups in the area. Recently, however, Adl et al. (2012) seeking to standardize and organize different groups of eukaryotes, mainly protists, suggested a new classification based on phylogenetic and ultra-structural similarity. Now Toxoplasma gondii appears within a super group called SAR comprising: Stramenopiles, Alveolata, and Rhizaria. More precisely, it appears inside the Alveolata group (first group); among Alveolata it is classified as Apicomplexa (second group); among the Apicomplexa it is classified as Conoidasida (third group); among these it is classified as Coccidia (fourth group); and finally among the Coccidia it is part of Eimeriorina group (fifth group), along with other associated parasites such as Cyclospora and Neospora. -

Comparative Transcriptome Analysis of Second- and Third-Generation Merozoites of Eimeria Necatrix
Su et al. Parasites & Vectors (2017) 10:388 DOI 10.1186/s13071-017-2325-z RESEARCH Open Access Comparative transcriptome analysis of second- and third-generation merozoites of Eimeria necatrix Shijie Su1,2,3, Zhaofeng Hou1,2,3, Dandan Liu1,2,3, Chuanli Jia1,2,3, Lele Wang1,2,3, Jinjun Xu1,2,3 and Jianping Tao1,2,3* Abstract Background: Eimeria is a common genus of apicomplexan parasites that infect diverse vertebrates, most notably poultry, causing serious disease and economic losses. Eimeria species have complex life-cycles consisting of three developmental stages. However, the molecular basis of the Eimeria reproductive mode switch remains an enigma. Methods: Total RNA extracted from second- (MZ-2) and third-generation merozoites (MZ-3) of Eimeria necatrix was subjected to transcriptome analysis using RNA sequencing (RNA-seq) followed by qRT-PCR validation. Results: A total of 6977 and 6901 unigenes were obtained from MZ-2 and MZ-3, respectively. Approximately 2053 genes were differentially expressed genes (DEGs) between MZ-2 and MZ-3. Compared with MZ-2, 837 genes were upregulated and 1216 genes were downregulated in MZ-3. Approximately 95 genes in MZ-2 and 48 genes in MZ-3 were further identified to have stage-specific expression. Gene ontology category and KEGG analysis suggested that 216 upregulated genes in MZ-2 were annotated by 70 GO assignments, 242 upregulated genes were associated with 188 signal pathways, while 321 upregulated genes in MZ-3 were annotated by 56 GO assignments, 322 upregulated genes were associated with 168 signal pathways. The molecular functions of upregulated genes in MZ-2 were mainly enriched for protein degradation and amino acid metabolism. -

Cyclospora Cayetanensis and Cyclosporiasis: an Update
microorganisms Review Cyclospora cayetanensis and Cyclosporiasis: An Update Sonia Almeria 1 , Hediye N. Cinar 1 and Jitender P. Dubey 2,* 1 Department of Health and Human Services, Food and Drug Administration, Center for Food Safety and Nutrition (CFSAN), Office of Applied Research and Safety Assessment (OARSA), Division of Virulence Assessment, Laurel, MD 20708, USA 2 Animal Parasitic Disease Laboratory, United States Department of Agriculture, Agricultural Research Service, Beltsville Agricultural Research Center, Building 1001, BARC-East, Beltsville, MD 20705-2350, USA * Correspondence: [email protected] Received: 19 July 2019; Accepted: 2 September 2019; Published: 4 September 2019 Abstract: Cyclospora cayetanensis is a coccidian parasite of humans, with a direct fecal–oral transmission cycle. It is globally distributed and an important cause of foodborne outbreaks of enteric disease in many developed countries, mostly associated with the consumption of contaminated fresh produce. Because oocysts are excreted unsporulated and need to sporulate in the environment, direct person-to-person transmission is unlikely. Infection by C. cayetanensis is remarkably seasonal worldwide, although it varies by geographical regions. Most susceptible populations are children, foreigners, and immunocompromised patients in endemic countries, while in industrialized countries, C. cayetanensis affects people of any age. The risk of infection in developed countries is associated with travel to endemic areas and the domestic consumption of contaminated food, mainly fresh produce imported from endemic regions. Water and soil contaminated with fecal matter may act as a vehicle of transmission for C. cayetanensis infection. The disease is self-limiting in most immunocompetent patients, but it may present as a severe, protracted or chronic diarrhea in some cases, and may colonize extra-intestinal organs in immunocompromised patients. -

2019-6654 Final Report of an Audit Carried out In
Ref. Ares(2019)7826731 - 19/12/2019 EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Health and food audits and analysis DG(SANTE) 2019-6654 FINAL REPORT OF AN AUDIT CARRIED OUT IN BELARUS FROM 13 TO 24 MAY 2019 IN ORDER TO EVALUATE THE CONTROL OF RESIDUES AND CONTAMINANTS IN LIVE ANIMALS AND ANIMAL PRODUCTS INCLUDING CONTROLS ON VETERINARY MEDICINAL PRODUCTS Executive Summary This report describes the outcome of an audit carried out in Belarus from 13 to 24 May 2019 as part of the European Commission’s Directorate-General for Health and Food Safety planned work programme. The objective of the audit was to evaluate the effectiveness of official controls on residues and contaminants in live animals and animal products eligible for export to the European Union (EU). The audit assessed the implementation of the residue monitoring plan and also covered the authorisation, distribution and use of veterinary medicinal products, given that these areas have an impact on the monitoring of residues. Attention was also paid to examining the implementation of corrective actions indicated in response to specific recommendations made in the report of the previous residues audit to Belarus. The planning of the residue monitoring largely follows the principles of Directive 96/23/EC and covers for the most part an appropriate range of substances. The plan is nevertheless weakened by the fact that action levels for several substances across all commodities (including those for which listing has been requested) are not aligned with EU maximum residue limits, thus the plan would not be sufficient to demonstrate that commodities eligible for export to the EU would comply with such limits where they are lower that national limits. -

A Novel Rhoptry Protein As Candidate Vaccine Against Eimeria Tenella Infection
Article A Novel Rhoptry Protein as Candidate Vaccine against Eimeria tenella Infection Xingju Song 1,2, Xu Yang 1,2, Taotao Zhang 1,2, Jing Liu 1,2 and Qun Liu 1,2,* 1 National Animal Protozoa Laboratory, College of Veterinary Medicine, China Agricultural University, Beijing 100083, China; [email protected] (X.S.); [email protected] (X.Y.); [email protected] (T.Z.); [email protected] (J.L.) 2 Key Laboratory of Animal Epidemiology of the Ministry of Agriculture, College of Veterinary Medicine, China Agricultural University, Beijing 100083, China * Correspondence: [email protected] Received: 25 July 2020; Accepted: 10 August 2020; Published: 12 August 2020 Abstract: Eimeria tenella (E. tenella) is a highly pathogenic and prevalent species of Eimeria that infects chickens, and it causes a considerable disease burden worldwide. The secreted proteins and surface antigens of E. tenella at the sporozoite stage play an essential role in the host–parasite interaction, which involves attachment and invasion, and these interactions are considered vaccine candidates based on the strategy of cutting off the invasion pathway to interrupt infection. We selected two highly expressed surface antigens (SAGs; Et-SAG13 and Et-SAG) and two highly expressed secreted antigens (rhoptry kinases Eten5-A, Et-ROPK-Eten5-A and dense granule 12, Et-GRA12) at the sporozoite stage. Et-ROPK-Eten5-A and Et-GRA12 were two unexplored proteins. Et-ROPK-Eten5-A was an E. tenella-specific rhoptry (ROP) protein and distributed in the apical pole of sporozoites and merozoites. Et-GRA12 was scattered in granular form at the sporozoite stage. -

DETECTION of TOXOPLASMA GONDII in FRESH PRODUCE By
DETECTION OF TOXOPLASMA GONDII IN FRESH PRODUCE by DANAYA AMINA BETHEA (Under the Direction of Ynes R. Ortega) ABSTRACT Toxoplasma gondii is an intracellular obligate protozoan parasite that causes toxoplasmosis in humans. This study aimed to detect and genotype T. gondii in fresh produce. Six PCR assays using various Toxoplasma gene targets were examined for their suitability in vegetable matrices. The SAG2 PCR was selected for this study. Collectively, DNA was extracted from 818 vegetable samples acquired from 16 markets in Atlanta, GA, U.S. and 5 in Lima, Peru. Nested polymerase chain reaction was conducted using characterized primers for T. gondii SAG2 locus. PCR products were purified and sequenced. Detection via PCR showed that 2.5% and 0.79% of U.S and Peru samples tested positive, respectively. The detection of T. gondii in fresh produce indicates that it may play a role in the prevalence of toxoplasmosis in the U.S and Peru. It also emphasizes the need to develop methodologies to prevent dissemination and inactivation of viable oocysts in the environment. INDEX WORDS: Toxoplasma gondii, Fresh Produce, PCR, Genotyping, SAG2, Peru, Georgia DETECTION OF TOXOPLASMA GONDII IN FRESH PRODUCE by DANAYA AMINA BETHEA B.S., Clark Atlanta University, 2011 A Thesis Submitted to the Graduate Faculty of The University of Georgia in Partial Fulfillment of the Requirements for the Degree MASTER OF SCIENCE ATHENS, GEORGIA 2014 © 2014 Danaya Amina Bethea All Rights Reserved DETECTION OF TOXOPLASMA GONDII IN FRESH PRODUCE by DANAYA AMINA BETHEA Major Professor: Ynes R. Ortega Committee: Jennifer Cannon Joseph F. Frank Electronic Version Approved: Julie Coffield Interim Dean of the Graduate School The University of Georgia August 2014 DEDICATION I would like to dedicate this manuscript to my family and friends, for their immeasurable love and support and my boyfriend Roderique John, for always being there for me. -

The Organic Chemistry of Drug Synthesis
The Organic Chemistry of Drug Synthesis VOLUME 2 DANIEL LEDNICER Mead Johnson and Company Evansville, Indiana LESTER A. MITSCHER The University of Kansas School of Pharmacy Department of Medicinal Chemistry Lawrence, Kansas A WILEY-INTERSCIENCE PUBLICATION JOHN WILEY AND SONS, New York • Chichester • Brisbane • Toronto Copyright © 1980 by John Wiley & Sons, Inc. All rights reserved. Published simultaneously in Canada. Reproduction or translation of any part of this work beyond that permitted by Sections 107 or 108 of the 1976 United States Copyright Act without the permission of the copyright owner is unlawful. Requests for permission or further information should be addressed to the Permissions Department, John Wiley & Sons, Inc. Library of Congress Cataloging in Publication Data: Lednicer, Daniel, 1929- The organic chemistry of drug synthesis. "A Wiley-lnterscience publication." 1. Chemistry, Medical and pharmaceutical. 2. Drugs. 3. Chemistry, Organic. I. Mitscher, Lester A., joint author. II. Title. RS421 .L423 615M 91 76-28387 ISBN 0-471-04392-3 Printed in the United States of America 10 987654321 It is our pleasure again to dedicate a book to our helpmeets: Beryle and Betty. "Has it ever occurred to you that medicinal chemists are just like compulsive gamblers: the next compound will be the real winner." R. L. Clark at the 16th National Medicinal Chemistry Symposium, June, 1978. vii Preface The reception accorded "Organic Chemistry of Drug Synthesis11 seems to us to indicate widespread interest in the organic chemistry involved in the search for new pharmaceutical agents. We are only too aware of the fact that the book deals with a limited segment of the field; the earlier volume cannot be considered either comprehensive or completely up to date. -

United States Patent (19) 11) Patent Number: 5,001,115 Sloan (45) Date of Patent: Mar
United States Patent (19) 11) Patent Number: 5,001,115 Sloan (45) Date of Patent: Mar. 19, 1991 54 PRODRUGS OF BIOLOGICALLY ACTIVE Primary Examiner-Mukund J. Shah HYDROXYAROMATIC COMPOUNDS Assistant Examiner-E. L. Ward Attorney, Agent, or Firm-Kerkam, Stowell, Kondracki (75 Inventor: Kenneth B. Sloan, Gainesville, Fla. & Clarke 73 Assignee: University of Florida, Gainesville, Fla, (57) ABSTRACT (21) Appl. No.: 352,919 Prodrugs of bio-active hydroxyaromatic drugs having 22 Filed: May 17, 1989 the structural formula: A pharmaceutically acceptable prodrug of a biologi (51] Int. Cl. ............... A61K 31/70; A61K 31/595; cally active, therapeutically effective hydroxyaromatic A61K 31/535; A61K 31/255 52 U.S. C. ..................................... 514/34: 514/289; drug, said prodrug being selected from the group con 514/169; 514/373; 514/222.8; 514/328; sisting of, (A) compounds having the structural for 514/360; 514/603; 514/417; 514/425; 514/518; mula: 546/44; 54.6/176; 546/75; 544/2; 536/64; 548/123; 548/209; 548/256; 548/417 DRUG-O-CR'R''-2) 58 Field of Search ............... 514/169,289, 373, 417, 514/425, 222.8, 328,360, 518, 603, 34; 536/64; wherein: 564/82, 155; 552/626; 546/44, 176, 75; 544/2; DRUG -O- is the hydroxyaromatic O-dehydro 548/123, 209, 256, 477, 595 residue of said drug; 56 References Cited R" and R' may be the same or different and may be H, PUBLICATIONS alkyl, aryl or electron withdrawing groups; 2 is a displaceable leaving group; and Katritzky, et al. J. Chem. Soc. -

Phylogeny of Coccidia and Coevolution with Their Hosts
School of Doctoral Studies in Biological Sciences Faculty of Science Phylogeny of coccidia and coevolution with their hosts Ph.D. Thesis MVDr. Jana Supervisor: prof. RNDr. Václav Hypša, CSc. 12 This thesis should be cited as: Kvičerová J, 2012: Phylogeny of coccidia and coevolution with their hosts. Ph.D. Thesis Series, No. 3. University of South Bohemia, Faculty of Science, School of Doctoral Studies in Biological Sciences, České Budějovice, Czech Republic, 155 pp. Annotation The relationship among morphology, host specificity, geography and phylogeny has been one of the long-standing and frequently discussed issues in the field of parasitology. Since the morphological descriptions of parasites are often brief and incomplete and the degree of host specificity may be influenced by numerous factors, such analyses are methodologically difficult and require modern molecular methods. The presented study addresses several questions related to evolutionary relationships within a large and important group of apicomplexan parasites, coccidia, particularly Eimeria and Isospora species from various groups of small mammal hosts. At a population level, the pattern of intraspecific structure, genetic variability and genealogy in the populations of Eimeria spp. infecting field mice of the genus Apodemus is investigated with respect to host specificity and geographic distribution. Declaration [in Czech] Prohlašuji, že svoji disertační práci jsem vypracovala samostatně pouze s použitím pramenů a literatury uvedených v seznamu citované literatury. Prohlašuji, že v souladu s § 47b zákona č. 111/1998 Sb. v platném znění souhlasím se zveřejněním své disertační práce, a to v úpravě vzniklé vypuštěním vyznačených částí archivovaných Přírodovědeckou fakultou elektronickou cestou ve veřejně přístupné části databáze STAG provozované Jihočeskou univerzitou v Českých Budějovicích na jejích internetových stránkách, a to se zachováním mého autorského práva k odevzdanému textu této kvalifikační práce.