Molecular Diversification of Salamanders of The
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Other Contributions
Other Contributions NATURE NOTES Amphibia: Caudata Ambystoma ordinarium. Predation by a Black-necked Gartersnake (Thamnophis cyrtopsis). The Michoacán Stream Salamander (Ambystoma ordinarium) is a facultatively paedomorphic ambystomatid species. Paedomorphic adults and larvae are found in montane streams, while metamorphic adults are terrestrial, remaining near natal streams (Ruiz-Martínez et al., 2014). Streams inhabited by this species are immersed in pine, pine-oak, and fir for- ests in the central part of the Trans-Mexican Volcanic Belt (Luna-Vega et al., 2007). All known localities where A. ordinarium has been recorded are situated between the vicinity of Lake Patzcuaro in the north-central portion of the state of Michoacan and Tianguistenco in the western part of the state of México (Ruiz-Martínez et al., 2014). This species is considered Endangered by the IUCN (IUCN, 2015), is protected by the government of Mexico, under the category Pr (special protection) (AmphibiaWeb; accessed 1April 2016), and Wilson et al. (2013) scored it at the upper end of the medium vulnerability level. Data available on the life history and biology of A. ordinarium is restricted to the species description (Taylor, 1940), distribution (Shaffer, 1984; Anderson and Worthington, 1971), diet composition (Alvarado-Díaz et al., 2002), phylogeny (Weisrock et al., 2006) and the effect of habitat quality on diet diversity (Ruiz-Martínez et al., 2014). We did not find predation records on this species in the literature, and in this note we present information on a predation attack on an adult neotenic A. ordinarium by a Thamnophis cyrtopsis. On 13 July 2010 at 1300 h, while conducting an ecological study of A. -

Amphibian Alliance for Zero Extinction Sites in Chiapas and Oaxaca
Amphibian Alliance for Zero Extinction Sites in Chiapas and Oaxaca John F. Lamoreux, Meghan W. McKnight, and Rodolfo Cabrera Hernandez Occasional Paper of the IUCN Species Survival Commission No. 53 Amphibian Alliance for Zero Extinction Sites in Chiapas and Oaxaca John F. Lamoreux, Meghan W. McKnight, and Rodolfo Cabrera Hernandez Occasional Paper of the IUCN Species Survival Commission No. 53 The designation of geographical entities in this book, and the presentation of the material, do not imply the expression of any opinion whatsoever on the part of IUCN concerning the legal status of any country, territory, or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. The views expressed in this publication do not necessarily reflect those of IUCN or other participating organizations. Published by: IUCN, Gland, Switzerland Copyright: © 2015 International Union for Conservation of Nature and Natural Resources Reproduction of this publication for educational or other non-commercial purposes is authorized without prior written permission from the copyright holder provided the source is fully acknowledged. Reproduction of this publication for resale or other commercial purposes is prohibited without prior written permission of the copyright holder. Citation: Lamoreux, J. F., McKnight, M. W., and R. Cabrera Hernandez (2015). Amphibian Alliance for Zero Extinction Sites in Chiapas and Oaxaca. Gland, Switzerland: IUCN. xxiv + 320pp. ISBN: 978-2-8317-1717-3 DOI: 10.2305/IUCN.CH.2015.SSC-OP.53.en Cover photographs: Totontepec landscape; new Plectrohyla species, Ixalotriton niger, Concepción Pápalo, Thorius minutissimus, Craugastor pozo (panels, left to right) Back cover photograph: Collecting in Chamula, Chiapas Photo credits: The cover photographs were taken by the authors under grant agreements with the two main project funders: NGS and CEPF. -

Multi-National Conservation of Alligator Lizards
MULTI-NATIONAL CONSERVATION OF ALLIGATOR LIZARDS: APPLIED SOCIOECOLOGICAL LESSONS FROM A FLAGSHIP GROUP by ADAM G. CLAUSE (Under the Direction of John Maerz) ABSTRACT The Anthropocene is defined by unprecedented human influence on the biosphere. Integrative conservation recognizes this inextricable coupling of human and natural systems, and mobilizes multiple epistemologies to seek equitable, enduring solutions to complex socioecological issues. Although a central motivation of global conservation practice is to protect at-risk species, such organisms may be the subject of competing social perspectives that can impede robust interventions. Furthermore, imperiled species are often chronically understudied, which prevents the immediate application of data-driven quantitative modeling approaches in conservation decision making. Instead, real-world management goals are regularly prioritized on the basis of expert opinion. Here, I explore how an organismal natural history perspective, when grounded in a critique of established human judgements, can help resolve socioecological conflicts and contextualize perceived threats related to threatened species conservation and policy development. To achieve this, I leverage a multi-national system anchored by a diverse, enigmatic, and often endangered New World clade: alligator lizards. Using a threat analysis and status assessment, I show that one recent petition to list a California alligator lizard, Elgaria panamintina, under the US Endangered Species Act often contradicts the best available science. -

Pseudoeurycea Naucampatepetl. the Cofre De Perote Salamander Is Endemic to the Sierra Madre Oriental of Eastern Mexico. This
Pseudoeurycea naucampatepetl. The Cofre de Perote salamander is endemic to the Sierra Madre Oriental of eastern Mexico. This relatively large salamander (reported to attain a total length of 150 mm) is recorded only from, “a narrow ridge extending east from Cofre de Perote and terminating [on] a small peak (Cerro Volcancillo) at the type locality,” in central Veracruz, at elevations from 2,500 to 3,000 m (Amphibian Species of the World website). Pseudoeurycea naucampatepetl has been assigned to the P. bellii complex of the P. bellii group (Raffaëlli 2007) and is considered most closely related to P. gigantea, a species endemic to the La specimens and has not been seen for 20 years, despite thorough surveys in 2003 and 2004 (EDGE; www.edgeofexistence.org), and thus it might be extinct. The habitat at the type locality (pine-oak forest with abundant bunch grass) lies within Lower Montane Wet Forest (Wilson and Johnson 2010; IUCN Red List website [accessed 21 April 2013]). The known specimens were “found beneath the surface of roadside banks” (www.edgeofexistence.org) along the road to Las Lajas Microwave Station, 15 kilometers (by road) south of Highway 140 from Las Vigas, Veracruz (Amphibian Species of the World website). This species is terrestrial and presumed to reproduce by direct development. Pseudoeurycea naucampatepetl is placed as number 89 in the top 100 Evolutionarily Distinct and Globally Endangered amphib- ians (EDGE; www.edgeofexistence.org). We calculated this animal’s EVS as 17, which is in the middle of the high vulnerability category (see text for explanation), and its IUCN status has been assessed as Critically Endangered. -

Assessing Environmental Variables Across Plethodontid Salamanders
Lauren Mellenthin1, Erica Baken1, Dr. Dean Adams1 1Iowa State University, Ames, Iowa Stereochilus marginatus Pseudotriton ruber Pseudotriton montanus Gyrinophilus subterraneus Gyrinophilus porphyriticus Gyrinophilus palleucus Gyrinophilus gulolineatus Urspelerpes brucei Eurycea tynerensis Eurycea spelaea Eurycea multiplicata Introduction Eurycea waterlooensis Eurycea rathbuni Materials & Methods Eurycea sosorum Eurycea tridentifera Eurycea pterophila Eurycea neotenes Eurycea nana Eurycea troglodytes Eurycea latitans Eurycea naufragia • Arboreality has evolved at least 5 times within Plethodontid salamanders. [1] Eurycea tonkawae Eurycea chisholmensis Climate Variables & Eurycea quadridigitata Polygons & Point Data Eurycea wallacei Eurycea lucifuga Eurycea longicauda Eurycea guttolineata MAXENT Modeling Eurycea bislineata Eurycea wilderae Eurycea cirrigera • Yet no morphological differences separate arboreal and terrestrial species. [1] Eurycea junaluska Hemidactylium scutatum Batrachoseps robustus Batrachoseps wrighti Batrachoseps campi Batrachoseps attenuatus Batrachoseps pacificus Batrachoseps major Batrachoseps luciae Batrachoseps minor Batrachoseps incognitus • There is minimal range overlap between the two microhabitat types. Batrachoseps gavilanensis Batrachoseps gabrieli Batrachoseps stebbinsi Batrachoseps relictus Batrachoseps simatus Batrachoseps nigriventris Batrachoseps gregarius Preliminary results discovered that 71% of the arboreal species distribution Batrachoseps diabolicus Batrachoseps regius Batrachoseps kawia Dendrotriton -

Results of the Global Amphibian Assessment for Bolivia
Results of the Global Amphibian Assessment for Bolivia Diversity Number of Rank in Latin World Rank2 Percentage of Species in Bolivia America and the the World’s Caribbean1 Diversity All Amphibians 201 7 15 3.5 % Frogs & Toads 197 7 14 3.9 % Salamanders 1 13 68 0.2 % Caecilians 3 11 20 1.8 % 1 Out of 44 countries and territories. 2 Out of 192 countries and territories. Threatened Species (Threatened species are in one of the categories in italics.) IUCN Categories Number of Species in IUCN Categories Number of Species in Bolivia Bolivia Extinct 0 Near Threatened 6 Critically Endangered 5 Least Concern 161 Endangered 6 Data Deficient 13 Vulnerable 10 Number and percent of Bolivian species that are threatened: 21 (10%) Extinct Species: none Species that are Critically Endangered and possibly extinct (species that are missing but for which sufficient information is lacking to declare them as extinct): Frogs: Gastrotheca lauzuricae, Hyla chlorostea, Eleutherodactylus zongoensis Role of Protected Areas: Of the 21 threatened species in Bolivia, 76% occur in at least one protected area. Local Experts: Steffen Reichle, The Nature Conservancy, TEL (591-3) 348 0766, 348 0767 Claudia Cortez, Colección Boliviana de Fauna, TEL (591-3) 272 1152 For More Information: Dr. Bruce Young, NatureServe, Costa Rica, TEL (506) 645-6231 Global Amphibian Assessment website: www.globalamphibians.org A detailed analysis of the results for the Americas: www.natureserve.org Results of the Global Amphibian Assessment for Bolivia Map of Amphibian Diversity Source: Global Amphibian Assessment Map of the Distribution of Threatened Species Source: Global Amphibian Assessment Results of the Global Amphibian Assessment for Brazil Diversity Number of Rank in Latin World Rank2 Percentage of Species in Brazil America and the the World’s Caribbean1 Diversity All Amphibians 731 1 1 12.7 % Frogs & Toads 704 1 1 13.9 % Salamanders 1 13 68 0.2 % Caecilians 26 2 2 15.5 % 1 Out of 44 countries and territories. -

About the Book the Format Acknowledgments
About the Book For more than ten years I have been working on a book on bryophyte ecology and was joined by Heinjo During, who has been very helpful in critiquing multiple versions of the chapters. But as the book progressed, the field of bryophyte ecology progressed faster. No chapter ever seemed to stay finished, hence the decision to publish online. Furthermore, rather than being a textbook, it is evolving into an encyclopedia that would be at least three volumes. Having reached the age when I could retire whenever I wanted to, I no longer needed be so concerned with the publish or perish paradigm. In keeping with the sharing nature of bryologists, and the need to educate the non-bryologists about the nature and role of bryophytes in the ecosystem, it seemed my personal goals could best be accomplished by publishing online. This has several advantages for me. I can choose the format I want, I can include lots of color images, and I can post chapters or parts of chapters as I complete them and update later if I find it important. Throughout the book I have posed questions. I have even attempt to offer hypotheses for many of these. It is my hope that these questions and hypotheses will inspire students of all ages to attempt to answer these. Some are simple and could even be done by elementary school children. Others are suitable for undergraduate projects. And some will take lifelong work or a large team of researchers around the world. Have fun with them! The Format The decision to publish Bryophyte Ecology as an ebook occurred after I had a publisher, and I am sure I have not thought of all the complexities of publishing as I complete things, rather than in the order of the planned organization. -

Minelli-Et-Al(Eds)
ZOOTAXA 1950 Updating the Linnaean Heritage: Names as Tools for Thinking about Animals and Plants ALESSANDRO MINELLI, LUCIO BONATO & GIUSEPPE FUSCO (EDS) Magnolia Press Auckland, New Zealand ALESSANDRO MINELLI, LUCIO BONATO & GIUSEPPE FUSCO (EDS) Updating the Linnaean Heritage: Names as Tools for Thinking about Animals and Plants (Zootaxa 1950) 163 pp.; 30 cm. 5 Dec. 2008 ISBN 978-1-86977-297-0 (paperback) ISBN 978-1-86977-298-7 (Online edition) FIRST PUBLISHED IN 2008 BY Magnolia Press P.O. Box 41-383 Auckland 1346 New Zealand e-mail: [email protected] http://www.mapress.com/zootaxa/ © 2008 Magnolia Press All rights reserved. No part of this publication may be reproduced, stored, transmitted or disseminated, in any form, or by any means, without prior written permission from the publisher, to whom all requests to reproduce copyright material should be directed in writing. This authorization does not extend to any other kind of copying, by any means, in any form, and for any purpose other than private research use. ISSN 1175-5326 (Print edition) ISSN 1175-5334 (Online edition) Zootaxa 1950: 3–4 (2008) ISSN 1175-5326 (print edition) www.mapress.com/zootaxa/ ZOOTAXA Copyright © 2008 · Magnolia Press ISSN 1175-5334 (online edition) Updating the Linnaean Heritage: Names as Tools for Thinking about Animals and Plants ALESSANDRO MINELLI FLS, LUCIO BONATO & GIUSEPPE FUSCO (EDS) Department of Biology, University of Padova, Via Ugo Bassi 58B, I 35131 Padova, Italy Email: [email protected], [email protected], [email protected] Table of contents 4 Preface ALESSANDRO MINELLI FLS, LUCIO BONATO, GIUSEPPE FUSCO (ITALY) 5 Actual usage of biological nomenclature and its implications for data integrators; a national, regional and global perspective CHARLES HUSSEY (UK), YDE DE JONG (THE NETHERLANDS), DAVID REMSEN (DENMARK) 9 The Linnean foundations of zoological and botanical nomenclature OTTO KRAUS (GERMANY) 21 Zoological vs. -
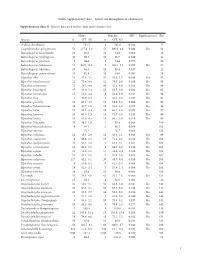
I Online Supplementary Data – Sexual Size Dimorphism in Salamanders
Online Supplementary data – Sexual size dimorphism in salamanders Supplementary data S1. Species data used in this study and references list. Males Females SSD Significant test Ref Species n SVL±SD n SVL±SD Andrias davidianus 2 532.5 8 383.0 -0.280 12 Cryptobranchus alleganiensis 53 277.4±5.2 52 300.9±3.4 0.084 Yes 61 Batrachuperus karlschmidti 10 80.0 10 84.8 0.060 26 Batrachuperus londongensis 20 98.6 10 96.7 -0.019 12 Batrachuperus pinchonii 5 69.6 5 74.6 0.070 26 Batrachuperus taibaiensis 11 92.9±12.1 9 102.1±7.1 0.099 Yes 27 Batrachuperus tibetanus 10 94.5 10 92.8 -0.017 12 Batrachuperus yenyuadensis 10 82.8 10 74.8 -0.096 26 Hynobius abei 24 57.8±2.1 34 55.0±1.2 -0.048 Yes 92 Hynobius amakusaensis 22 75.4±4.8 12 76.5±3.6 0.014 No 93 Hynobius arisanensis 72 54.3±4.8 40 55.2±4.8 0.016 No 94 Hynobius boulengeri 37 83.0±5.4 15 91.5±3.8 0.102 Yes 95 Hynobius formosanus 15 53.0±4.4 8 52.4±3.9 -0.011 No 94 Hynobius fuca 4 50.9±2.8 3 52.8±2.0 0.037 No 94 Hynobius glacialis 12 63.1±4.7 11 58.9±5.2 -0.066 No 94 Hynobius hidamontanus 39 47.7±1.0 15 51.3±1.2 0.075 Yes 96 Hynobius katoi 12 58.4±3.3 10 62.7±1.6 0.073 Yes 97 Hynobius kimurae 20 63.0±1.5 15 72.7±2.0 0.153 Yes 98 Hynobius leechii 70 61.6±4.5 18 66.5±5.9 0.079 Yes 99 Hynobius lichenatus 37 58.5±1.9 2 53.8 -0.080 100 Hynobius maoershanensis 4 86.1 2 80.1 -0.069 101 Hynobius naevius 72.1 76.7 0.063 102 Hynobius nebulosus 14 48.3±2.9 12 50.4±2.1 0.043 Yes 96 Hynobius osumiensis 9 68.4±3.1 15 70.2±3.0 0.026 No 103 Hynobius quelpaertensis 41 52.5±3.8 4 61.3±4.1 0.167 Yes 104 Hynobius -

3Systematics and Diversity of Extant Amphibians
Systematics and Diversity of 3 Extant Amphibians he three extant lissamphibian lineages (hereafter amples of classic systematics papers. We present widely referred to by the more common term amphibians) used common names of groups in addition to scientifi c Tare descendants of a common ancestor that lived names, noting also that herpetologists colloquially refer during (or soon after) the Late Carboniferous. Since the to most clades by their scientifi c name (e.g., ranids, am- three lineages diverged, each has evolved unique fea- bystomatids, typhlonectids). tures that defi ne the group; however, salamanders, frogs, A total of 7,303 species of amphibians are recognized and caecelians also share many traits that are evidence and new species—primarily tropical frogs and salaman- of their common ancestry. Two of the most defi nitive of ders—continue to be described. Frogs are far more di- these traits are: verse than salamanders and caecelians combined; more than 6,400 (~88%) of extant amphibian species are frogs, 1. Nearly all amphibians have complex life histories. almost 25% of which have been described in the past Most species undergo metamorphosis from an 15 years. Salamanders comprise more than 660 species, aquatic larva to a terrestrial adult, and even spe- and there are 200 species of caecilians. Amphibian diver- cies that lay terrestrial eggs require moist nest sity is not evenly distributed within families. For example, sites to prevent desiccation. Thus, regardless of more than 65% of extant salamanders are in the family the habitat of the adult, all species of amphibians Plethodontidae, and more than 50% of all frogs are in just are fundamentally tied to water. -

Environmental and Social Management Framework for the Integrated Transboundary Ridge-To-Reef Management of the Mesoamerican Reef System Project
Environmental and Social Management Framework for the Integrated Transboundary Ridge-to-Reef Management of the Mesoamerican Reef System project ii Contents Terms and Acronyms Used in this Report ................................................................................................................ iii Introduction .......................................................................................................................................................................... 1 Institutional Framework .................................................................................................................................................. 2 Budgetary Arrangements ............................................................................................................................................ 3 The Meso-American Reef Ecoregion ............................................................................................................................ 4 Legal Frameworks .............................................................................................................................................................. 6 Classification of Projects .................................................................................................................................................. 7 Scoping .................................................................................................................................................................................... 8 Priority Watersheds -

Phylogenetic Relationships Among the Salamanders of the Bolitoglossa Macrinii Species Group
Journal of Herpetology, Vol. 36, No. 3, pp. 356–366, 2002 Copyright 2002 Society for the Study of Amphibians and Reptiles Phylogenetic Relationships among the Salamanders of the Bolitoglossa macrinii Species Group (Amphibia: Plethodontidae), with Descriptions of Two New Species from Oaxaca (Me´xico) GABRIELA PARRA-OLEA,1 MARIO GARCI´A-PARI´S,2,3 AND DAVID B. WAKE2,4 1Museum of Comparative Zoology, Harvard University, Cambridge, MA 02138, USA, and Instituto de Biologia, UNAM AP 70-153, CP 04510, Ciudad Universitaria, Me´xico, D.F. 2Museum of Vertebrate Zoology, 3101 Valley Life Sciences Building, University of California, Berkeley, California 94720-3160, USA 3Museo Nacional de Ciencias Naturales, CSIC, Jose´Gutie´rrez Abascal 2, 28006 Madrid, Spain ABSTRACT.—The Bolitoglossa macrinii species group (restricted geographically to southern Oaxaca and southwestern Guerrero, Me´xico) is a monophyletic assemblage of five species, B. macrinii, Bolitoglossa riletti, Bolitoglossa hermosa, Bolitoglossa oaxacensis sp. nov., and Bolitoglossa zapoteca sp. nov. DNA se- quences totaling 1164 base pairs for the mitochondrial genes 16S ribosomal RNA and cytochrome b were analyzed to generate a phylogenetic hypothesis for the relationships within the group. Our hypothesis is in agreement with previous morphological and allozyme analyses. Divergence within the group is great (to 18.6% for cytochrome b, to 7.0% for 16S rRNA) and two clades are well supported: one including B. riletti, B. hermosa,andB. zapoteca and the other including B. macrinii and B. oaxacensis. These clades likely diverged beginning in mid-Miocene times. We describe two new species, B. oaxacensis, for inland Oaxacan populations previously assigned to B.