(Ephemeroptera) in Lotic Karst Habitats
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

9 a New Record of Burrowing Mayfly, Anthopotamus Neglectus Neglectus
Ohio Biological Survey Notes 10: 9–12, 2021. © Ohio Biological Survey, Inc. A New Record of Burrowing Mayfly, Anthopotamus neglectus neglectus (Traver, 1935) (Ephemeroptera: Potamanthidae), from Ohio, USA DONALD H. DEAN1 1Departments of Entomology and Chemistry & Biochemistry, 484 W. 12th Ave., The Ohio State University, Columbus, OH USA 43214. E-mail: [email protected] Abstract: A new state record for a mayfly (Ephemeroptera) was collected on the Olentangy River, Delaware County, Ohio, USA. Anthopotamus neglectus Traver (1935) were collected as nymphs and subsequently reared to adults. Keywords: Olentangy River, Delaware County, Ohio Introduction The neglected hackle-gilled burrowing mayfly, or the golden (or yellow) drake to fly fishers, Anthopotamus neglectus was first described by Traver (1935) as Potomanthus neglectus. Bae and McCafferty (1991) reorganized the family Potomanthidae and placed the taxon in a new genus, Anthopotamus McCafferty and Bae (1990). They further divided the species into two subspecies, A. neglectus neglectus and A. neglectus disjunctus. The geographic range of the former species was originally given as a small circle centered in New York. The latter species was centered in the south-central United States. More recently, A. neglectus neglectus has been reported in eastern North America including Ontario, Alabama, Arkansas, Maryland, Missouri, Mississippi, New York, Oklahoma, Tennessee, Virginia, and West Virginia (Randolph, 2002). The online database NatureServe Explorer (2019) lists the range of A. neglectus neglectus as previously stated, with the addition of Georgia and Pennsylvania (but it includes the caveat “Distribution data for U.S. states and Canadian provinces is known to be incomplete or has not been reviewed for this taxon”). -
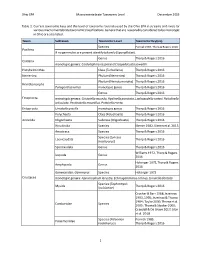
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Pennak 1989, Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Crocker & Barr 1968; Jezerinac 1993, 1995; Jezerinac & Thoma 1984; Taylor 2000; Thoma et al. Cambaridae Species 2005; Thoma & Stocker 2009; Crandall & De Grave 2017; Glon et al. 2018 Species (Palaemon Pennak 1989, Palaemonidae kadiakensis) Thorp & Rogers 2016 1 Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites Genus Morse et al. -

Adhesion Performance in the Eggs of the Philippine Leaf Insect Phyllium Philippinicum (Phasmatodea: Phylliidae)
insects Article Adhesion Performance in the Eggs of the Philippine Leaf Insect Phyllium philippinicum (Phasmatodea: Phylliidae) Thies H. Büscher * , Elise Quigley and Stanislav N. Gorb Department of Functional Morphology and Biomechanics, Institute of Zoology, Kiel University, Am Botanischen Garten 9, 24118 Kiel, Germany; [email protected] (E.Q.); [email protected] (S.N.G.) * Correspondence: [email protected] Received: 12 June 2020; Accepted: 25 June 2020; Published: 28 June 2020 Abstract: Leaf insects (Phasmatodea: Phylliidae) exhibit perfect crypsis imitating leaves. Although the special appearance of the eggs of the species Phyllium philippinicum, which imitate plant seeds, has received attention in different taxonomic studies, the attachment capability of the eggs remains rather anecdotical. Weherein elucidate the specialized attachment mechanism of the eggs of this species and provide the first experimental approach to systematically characterize the functional properties of their adhesion by using different microscopy techniques and attachment force measurements on substrates with differing degrees of roughness and surface chemistry, as well as repetitive attachment/detachment cycles while under the influence of water contact. We found that a combination of folded exochorionic structures (pinnae) and a film of adhesive secretion contribute to attachment, which both respond to water. Adhesion is initiated by the glue, which becomes fluid through hydration, enabling adaption to the surface profile. Hierarchically structured pinnae support the spreading of the glue and reinforcement of the film. This combination aids the egg’s surface in adapting to the surface roughness, yet the attachment strength is additionally influenced by the egg’s surface chemistry, favoring hydrophilic substrates. -

Research Paper Comparative Study of The
Internat. Rev. Hydrobiol. 95 2010 1 58–71 DOI: 10.1002/iroh.200811197 MANUEL JESÚS LÓPEZ-RODRÍGUEZ, JOSÉ MANUEL TIERNO DE FIGUEROA and JAVIER ALBA-TERCEDOR Departamento de Biología Animal. Facultad de Ciencias. Universidad de Granada. 18071, Granada, Spain; e-mail: [email protected] Research Paper Comparative Study of the Nymphal Biology of Two Coexisting Species of Mayflies (Insecta: Ephemeroptera) in a Mediterranean Stream in Southern Europe key words: mayflies, life cycle, feeding, secondary production, niche overlap Abstract We study the life history, nymphal feeding and secondary production of two leptophlebiid mayfly species (Habrophlebia eldae and Paraleptophlebia submarginata). They cohabit in a Mediterranean stream and present a very high niche overlap in terms of trophic resources. The life cycle was esti- mated using size-frequency analysis of samples taken throughout a year. Both species have a similar but displaced period of the nymphal development. Secondary production was calculated by means of the size-frequency method. Annual secondary production of P. submarginata is much higher than that of H. eldae (1.95 g DW m–2 year–1 vs. 0.17 g DW m–2 year–1), and presents a quite similar annual P/B ratio, but slightly higher in P. submarginata (6.97 in P. submarginata and 9.21 in H. eldae). The study of the gut contents revealed that they are mainly detritivores but, when larger they feed also on CPOM from leaves fallen in the stream. They present an almost total niche overlap in terms of food acquisition. However the previously mentioned shift in trophic resources utilization with size makes it possible that, because no similar size classes of each species are present at the same time, niche segregation exists between the two species. -

Microsoft Outlook
Joey Steil From: Leslie Jordan <[email protected]> Sent: Tuesday, September 25, 2018 1:13 PM To: Angela Ruberto Subject: Potential Environmental Beneficial Users of Surface Water in Your GSA Attachments: Paso Basin - County of San Luis Obispo Groundwater Sustainabilit_detail.xls; Field_Descriptions.xlsx; Freshwater_Species_Data_Sources.xls; FW_Paper_PLOSONE.pdf; FW_Paper_PLOSONE_S1.pdf; FW_Paper_PLOSONE_S2.pdf; FW_Paper_PLOSONE_S3.pdf; FW_Paper_PLOSONE_S4.pdf CALIFORNIA WATER | GROUNDWATER To: GSAs We write to provide a starting point for addressing environmental beneficial users of surface water, as required under the Sustainable Groundwater Management Act (SGMA). SGMA seeks to achieve sustainability, which is defined as the absence of several undesirable results, including “depletions of interconnected surface water that have significant and unreasonable adverse impacts on beneficial users of surface water” (Water Code §10721). The Nature Conservancy (TNC) is a science-based, nonprofit organization with a mission to conserve the lands and waters on which all life depends. Like humans, plants and animals often rely on groundwater for survival, which is why TNC helped develop, and is now helping to implement, SGMA. Earlier this year, we launched the Groundwater Resource Hub, which is an online resource intended to help make it easier and cheaper to address environmental requirements under SGMA. As a first step in addressing when depletions might have an adverse impact, The Nature Conservancy recommends identifying the beneficial users of surface water, which include environmental users. This is a critical step, as it is impossible to define “significant and unreasonable adverse impacts” without knowing what is being impacted. To make this easy, we are providing this letter and the accompanying documents as the best available science on the freshwater species within the boundary of your groundwater sustainability agency (GSA). -

Área De Estudio
Wildfire effects on macroinvertebrate communities in Mediterranean streams Efectes dels incendis forestals sobre las comunitats de macroinvertebrats en rius mediterranis Iraima Verkaik ADVERTIMENT. La consulta d’aquesta tesi queda condicionada a l’acceptació de les següents condicions d'ús: La difusió d’aquesta tesi per mitjà del servei TDX (www.tesisenxarxa.net) ha estat autoritzada pels titulars dels drets de propietat intel·lectual únicament per a usos privats emmarcats en activitats d’investigació i docència. No s’autoritza la seva reproducció amb finalitats de lucre ni la seva difusió i posada a disposició des d’un lloc aliè al servei TDX. No s’autoritza la presentació del seu contingut en una finestra o marc aliè a TDX (framing). Aquesta reserva de drets afecta tant al resum de presentació de la tesi com als seus continguts. En la utilització o cita de parts de la tesi és obligat indicar el nom de la persona autora. ADVERTENCIA. La consulta de esta tesis queda condicionada a la aceptación de las siguientes condiciones de uso: La difusión de esta tesis por medio del servicio TDR (www.tesisenred.net) ha sido autorizada por los titulares de los derechos de propiedad intelectual únicamente para usos privados enmarcados en actividades de investigación y docencia. No se autoriza su reproducción con finalidades de lucro ni su difusión y puesta a disposición desde un sitio ajeno al servicio TDR. No se autoriza la presentación de su contenido en una ventana o marco ajeno a TDR (framing). Esta reserva de derechos afecta tanto al resumen de presentación de la tesis como a sus contenidos. -
![Complementary Description of Habrophlebia Vaillantorum Thomas, 1986 in Comparison with H.Fusca (Curtis, 1834) [Ephemeroptera, Leptophlebiidae]](https://docslib.b-cdn.net/cover/2552/complementary-description-of-habrophlebia-vaillantorum-thomas-1986-in-comparison-with-h-fusca-curtis-1834-ephemeroptera-leptophlebiidae-1842552.webp)
Complementary Description of Habrophlebia Vaillantorum Thomas, 1986 in Comparison with H.Fusca (Curtis, 1834) [Ephemeroptera, Leptophlebiidae]
Ephemera, 1999, Vol. 1 (1) : 9-21 Complementary description of Habrophlebia vaillantorum Thomas, 1986 in comparison with H.fusca (Curtis, 1834) [Ephemeroptera, Leptophlebiidae] by Alain THOMAS*, Elda GAINO** and Virginie MARIE* * Laboratoire d'Hydrobiologie, UMR CESAC, 118, Route de Narbonne, F - 31062 Toulouse Cedex 4, France ** Istituto di Zoologia, Via Elce di Sotto, I - 06123 Perugia, Italia Keywords : Habrophlebia, systematics, morphology, adults, last-instar larva, egg, SEM. The male adult of Habrophlebia vaillantorum is described for the first time. The larvae mainly differ from those of H. fusca by the morphology of the mouthparts (maxillae, labium), femoral and tibial bristles, tarsal claws, marginal spines of abdominal terga, and gills, the latter being adapted to flowing water in H. vaillan torum. Feathery bristles on maxillary palpi and internai side oftibiae (filtering structure) are reduced or ab sent in H. vaillantorum in comparison with H. fusca. Scanning electron microscopy (SEM) investigation permitted to highlight the chorionic pattern in both spe cies (the egg of H.fusca being also described for the first time). Among the various characteristics useful for a proper diagnosis, the most outstanding feature is represented by the ribs, less numerous and wider in H. fus ca than in H. vaillantorum. Description complémentaire d'Habrophlebia vaillantorum Thomas, 1986, comparative avec H. fusca (Curtis, 1984) [Ephemeroptera, Leptophlebiidae] Mots clés: Habrophlebia, systématique, morphologie, adultes, larve au dernier stade, oeuf, MEB. L'adulte mâle d'Habrophlebia vaillantorum est décrit pour la première fois. Les larves diffèrent principa lement de celles d' H. fusca par la morphologie des pièces buccales (maxilles, labium), des soies des fémurs et des tibias, des griffes tarsales, des épines marginales des tergites abdominaux et des branchies, ces der nières étant adaptées aux eaux courantes chez H. -

Filter-Feeding Habits of the Larvae of Anthopotamus \(Ephemeroptera
Annls Limnol. 28 (1) 1992 : 27-34 Filter-feeding habits of the larvae of Anthopotamus (Ephemeroptera : Potamanthidae) W.P. McCaffertyl Y.J. Bae1 Keywords : Ephemeroptera, Potamanthidae, Anthopotamus, filter feeding, behavior, morphology, detritus. A field and laboratory investigation of the food and feeding behavior of larvae of the potamanthid mayfly Anthopo tamus verticis (Say) was conducted from 1989 to 1991 on a population from the Tippecanoe River, Indiana (USA). Gut content analyses indicated that all size classes of larvae are detritivores, with over 95 % of food consisting of fine detrital particles. Videomacroscopy indicated that all size classes of larvae are filter feeders, able to utilize both active deposit filter feeding and passive seston filter feeding cycles in their interstitial microhabitat. Deposit filter feeding initially incor porates the removal of loosely deposited detritus with the forelegs. Seston filter feeding initially incorporates filtering by long setae on the forelegs and palps. Mandibular tusks are used to help remove detritus from the foreleg setae. A SEM examination of filtering setae indicated they are hairlike and bipectinate, being equipped with lateral rows of setu- les. The results show that previous assumptions that potamanthid larvae were collector/gatherers are erroneous. The results are applicable to congeners, and all potamanthids, including Palearctic and Oriental elements, are hypothesized to be filter feeders, differing only in some details of behavior. Le régime trophique de type filtreur des larves d'Anthopotamus (Ephemeroptera : Potamanthidae) Mots clés : Ephemeroptera, Potamanthidae, Anthopotamus, type trophique filtreur, comportement, morphologie, débris particulates. Une investigation sur le régime alimentaire et le comportement trophique des larves de l'Ephémère Potamanthidae Anthopotamus verticis (Say) a été menée à la fois sur le terrain et expérimentalement au laboratoire de 1989 à 1991, sur une population de la rivière Tippecanoe, Indiana (USA). -

1 New Ohio and Indiana Records of Aquatic Insects (Ephemeroptera
Ohio Biological Survey Notes 9: 1–15, 2019. © Ohio Biological Survey, Inc. New Ohio and Indiana Records of Aquatic Insects (Ephemeroptera, Plecoptera, Trichoptera, Coleoptera: Elmidae, Diptera: Chironomidae) MICHAEL J. BOLTON1, SARAH K. MACY2, R. EDWARD DEWALT3, AND LUKE M. JACOBUS4 1Ohio Environmental Protection Agency, Division of Surface Water, 4675 Homer Ohio Lane, Groveport, OH 43125, Michael.Bolton@epa. ohio.gov; 2Formerly with the Ohio Environmental Protection Agency; current e-mail: [email protected]; 3University of Illinois, Illinois Natural History Survey, 1816 S Oak St., Champaign, IL 61820, [email protected]; 4Indiana University–Purdue University Columbus, 4601 Central Avenue, Columbus, IN 47203, [email protected]. Abstract: New state records and additional locations for rarely collected species are reported for Ephemeroptera (mayflies), Plecoptera (stoneflies), Trichoptera (caddisflies), Coleoptera: Elmidae (riffle beetles), and Diptera: Chironomidae (chironomids, non-biting midges, midges). These specimen records result primarily from Ohio Environmental Protection Agency biomonitoring of Ohio streams and from records found in the Purdue University Entomological Research Collection and the Illinois Natural History Survey Insect Collection; a few records were derived from material housed in two other collections. New state records for Ohio consist of the mayflies Acentrella rallatoma Burian & Myers, Acerpenna pygmaea (Hagen), Anafroptilum album (McDunnough), Anafroptilum minor group species 1, Anafroptilum -

Ephemeroptera: Potamanthidae)
Hydrobiologia 288: 65-78, 1994. 0 1994 Kluwer Academic Publishers. Printed in Belgium. 65 Microhabitat of Anthopotamus verticis (Ephemeroptera: Potamanthidae) Y. J. Bae & W. P. McCafferty Department of Entomology, Purdue University, West Lafayette, IN 47907, USA Received 24 September 1992; in revised form 10 August 1993; accepted 14 September 1993 Key words:Anthopotamus verticis, burrowing mayflies, Ephemeroptera, microhabitat, substrate particle size Abstract A field and laboratory study was designed to elucidate the microhabitat of the North American river- ine mayfly Anthopotamus verticis (Say). Previous reports have suggested that these mayflies are surface sprawlers or clingers; but despite a flattened body shape that is often associated with sprawling or clinging benthos, our investigation showed that larvae of A. verticis burrow and inhabit the hyporheic biotope. Substrate particle size was found to be a primary limiting factor in the microdistribution of the larvae. When homogeneous substrates (either fine, medium, or coarse gravel, or small or large pebbles) were provided, small, mid-sized, and large larvae significantly preferred coarse gravel or small pebbles. Small larvae were better represented in coarse gravel, and large larvae were better represented in small pebbles. Vertical distribution was deeper in coarse gravel than in medium gravel, and smaller larvae were gen- erally found deeper than large larvae. In the field, larvae occur in gravel or pebbles (excluding coarse sand or smaller particles) or mixed substrates, often at the interface of large rocks and finer substrates. Videomacroscopic examination also indicated larvae to be interstitial dwellers. Although relatively crude burrowers, larvae do use their tusks to excavate substrate, and their gills to generate interstitial current. -

Ladislav Toncr
Univerzita Palackého v Olomouci Přírodovědecká fakulta Katedra zoologie a ornitologická laboratoř Ladislav Toncr Životní cykly a emergence alpského hmyzu čeledi Baetidae Bakalářská práce Studijní program: Biologie Studijní obor: Systematická biologie a ekologie Forma studia: prezenční Vedoucí práce: RNDr. Vladimír Uvíra Dr. Termín odevzdání práce: 22. 7. 2013 Olomouc 2013 © Ladislav Toncr, 2013 Prohlášení „Prohlašuji, ţe jsem předloţenou bakalářskou práci vypracoval samostatně za pouţití citované literatury.“ V Olomouci dne 30. 7. 2013 ………………………… Podpis Poděkování: V prvé řadě bych rád poděkoval RNDr. Vladimíru Uvírovi, Dr. za poskytnutí cenných rad, konzultací a odborné literatury. Velký dík patří i mé rodině a přítelkyni za obrovskou podporu při psaní této práce a samotném studiu. Bibliografická identifikace Jméno a příjmení autora: Ladislav Toncr Název práce: Ţivotní cykly a emergence alpského hmyzu čeledi Baetidae Typ práce: Bakalářská práce Pracoviště: Katedra zoologie a ornitologická laboratoř Vedoucí práce: RNDr. Vladimír Uvíra, Dr. Rok obhajoby práce: 2013 Abstrakt: Práce se zabývá ţivotními cykly jepic čeledi Baetidae od vajíčka přes larvu a subimago aţ po dospělce. Pozornost je zaměřena hlavně na působení enviromentálních faktorů na ţivotní cykly vybraných druhů jepic. Za nejdůleţitější činitele jsou povaţovány: teplota, fotoperiodicita a dostupná potrava. Dále se za významné povaţují působení pH a toxických látek, biologický drift a predace. Tyto faktory ovlivňují zejména počet generací za rok, coţ se můţe lišit u jednotlivých druhů na různých stanovištích. Část práce je věnována působení dlouhodobých klimatických změn na ţivotní cykly akvatického hmyzu. Klíčová slova: Ephemeroptera, Baetidae, ţivotní cyklus, emergence, subimago, environmentální faktory prostředí Bibliographical identification Autor’s first name and surname: Ladislav Toncr Title: Life cycles and the emergence of alpine insects of the family Baetidae Type of thesis: Bachelor thesis Department: Department of Zoology and laboratory of ornithology Supervisior: RNDr. -

DOCTORAL THESIS the Influence of Hydrogeology and the Devensian
DOCTORAL THESIS The influence of hydrogeology and the Devensian glaciation on hyporheic communities of the UK Dunscombe, Mark Award date: 2012 General rights Copyright and moral rights for the publications made accessible in the public portal are retained by the authors and/or other copyright owners and it is a condition of accessing publications that users recognise and abide by the legal requirements associated with these rights. • Users may download and print one copy of any publication from the public portal for the purpose of private study or research. • You may not further distribute the material or use it for any profit-making activity or commercial gain • You may freely distribute the URL identifying the publication in the public portal ? Take down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. Download date: 06. Oct. 2021 The influence of hydrogeology and the Devensian glaciation on hyporheic communities of the UK Mark Dunscombe BSc MSc A thesis submitted in partial fulfilment of the requirements for the degree of PhD Department of Life Sciences Roehampton University University of Surrey 2011 1 Abstract Ecologically, the hyporheic zone (HZ) performs numerous roles within stream ecosystems (e.g. habitat, refugia from floods and droughts, nutrient cycling, pollution attenuation) with the dominant control on these various roles sediment composition. Recently, the body of literature on the role of sediment in the HZ has grown, though these studies rarely extend beyond the reach scale with little regional scale research undertaken in England and Wales.