MIAMI UNIVERSITY the Graduate School
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Monitoring of Water Level Fluctuations of Darbandikhan Lake Using Remote Sensing Techniques
1 Plant Archives Vol. 20, Supplement 2, 2020 pp. 901-906 e-ISSN:2581-6063 (online), ISSN:0972-5210 MONITORING OF WATER LEVEL FLUCTUATIONS OF DARBANDIKHAN LAKE USING REMOTE SENSING TECHNIQUES Dalshad R. Azeez* 1, Fuad M. Ahmad 2 and Dashne A.K. Karim 2 1College of Agriculture, Kirkuk University, Iraq 2College of Agricultural Engineering, Salahaddin University, Iraq *Corresponding author : [email protected] Abstract Darbandikhan Reservoir Dam is located on the Sirwan (Diyala) River, about 230 km northeast of Baghdad and 65 km southeast of Sulaimaniyah - Iraq. Its borders extend from latitude 35° 06' 58 ''- 35° 21' 07 ''N and longitude 45° 40' 59'' -45° 44' 42'' E. In order to monitor the fluctuation in the level of this lake, Landsat Satellite images were collected for 10 years included, 1984, 1990, 1995, 2000, 2005, 2010, 2015, 2017, 2018 and 2019 for the same time period. Next, the classification of satellite images and the measurement of areas were done using ArcGIS 10.2. In order to study the effect of droughts and wet conditions on water levels in the lake, the standardized precipitation index (SPI) method proposed by Mckee, 1993 was used. In the current study, the 12 months’ time scale SPI values (SPI-12) that was considered individually for the years 1984-2017 were estimated. Each period starts from January and ends in December. It was found that the water level in Darbandikhan Lake has experienced periodic changes during the period from 1984 to 2018. The results also showed that there were some gradual drought trends in the study area according to precipitation changes during the years studied, where severe drought dominated several parts of the study area, and the worst were in the years 1995, 2000, 2915 and 2017. -

Eliminating Violence Against Women
ELIMINATING VIOLENCE AGAINST WOMEN PERSPECTIVES ON HONOR-RELATED VIOLENCE IN THE IRAQI KURDISTAN REGION, SULAIMANIYA GOVERNORATE By Tanyel B. Taysi With Contributions from Norul M. Rashid Martin Bohnstedt ASUDA & UNAMI HUMAN RIGHTS OFFICE: ELIMINATING VIOLENCE AGAINST WOMEN FOREWORD ......................................................................................................................................3 I. INTRODUCTION.............................................................................................................................4 II. INTERNATIONAL, REGIONAL AND NATIONAL HUMAN RIGHTS FRAMEWORKS .......................8 III. HONOR-RELATED VIOLENCE..................................................................................................14 IV. CONTEXTUAL OVERVIEW OF WOMEN’S POSITION IN IRAQI KURDISTAN ............................16 V. FINDINGS ...................................................................................................................................19 VI. SUMMARY ................................................................................................................................41 VII. CONCLUSIONS AND RECOMMENDATIONS .............................................................................43 APPENDIX.......................................................................................................................................48 Honor-related Violence in the Kurdistan Region Page 2 ASUDA & UNAMI HUMAN RIGHTS OFFICE: ELIMINATING VIOLENCE AGAINST WOMEN FOREWORD Honor-related -

Official General Report on Northern Iraq (April 2000) Contents Page
Official general report on Northern Iraq (April 2000) Contents Page 1. Introduction 4 2. Information on the country 6 2.1. Basic facts 6 2.1.1. Country and people 6 2.1.2. History 8 2.2. System of government 17 2.3. Political developments 20 2.3.1. Internal relations 20 2.3.2. External forces 31 2.4. Security situation 36 2.5. Social and economic situation 48 2.6. Conclusions 53 3. Human rights 55 3.1. Safeguards 55 3.1.1. Constitution 55 3.1.2. Other national legislation 55 3.1.3. Conventions 56 3.2. Monitoring 56 3.3. Respect and violations 58 3.3.1. Freedom of opinion 58 3.3.2. Freedom of association and of assembly 59 3.3.3. Freedom of religion 60 3.3.4. Freedom of movement 73 3.3.5. Judicial process 83 3.3.6. Arrest and detention 84 3.3.7. Maltreatment and torture 87 3.3.8. Extra-judicial executions and murders 87 10804/00 dre/LG/mc 2 DG H I EN 3.3.9. Death penalty 87 3.4. Position of specific groups 88 3.4.1. Turkmens 88 3.4.2. Staff of international organisations 91 3.4.3. Conscripts, deserters and servicemen 96 3.4.4. Independent intellectuals and journalists 98 3.4.5. Prominent political activists 99 3.4.6. Fayli Kurds 99 3.4.7. Women 101 3.4.8. Orphaned minors 104 3.5. Summary 104 4. Refugees and displaced persons 106 4.1. Motives 106 4.2. -

CT DEEP Family-Level Identification Guide for Riffle-Dwelling Macroinvertebrates of Connecticut
CT DEEP Family-Level Identification Guide for Riffle-Dwelling Macroinvertebrates of Connecticut Seventh Edition Spring 2013 Authors and Acknowledgements Michael Beauchene produced the First Edition and revised the Second and Third Editions. Christopher Sullivan revised the Fourth and Fifth Editions. Erin McCollum developed the Sixth Edition with editorial assistance from Michael Beauchene. The First through Sixth Editions were developed and revised for use with Project SEARCH, a program formerly coordinated by CTDEEP but presently inactive. This Seventh Edition has been slightly modified for use by Connecticut high school students participating in the Connecticut Envirothon Aquatic Ecology workshop. Original drawings provided by Michael Beauchene and by the Volunteer Stream Monitoring Partnership at the University of Minnesota’s Water Resources Center. This page intentionally left blank. About the Key Scope of the Key This key is intended to assist Connecticut Envirothon students in the identification of aquatic benthic macroinvertebrates. As such, it is targeted toward organisms that are most commonly found in the riffle microhabitats of Connecticut streams. When conducting an actual field study of riffle dwelling macroinvertebrates, there may be an organism collected at a site in Connecticut that will not be found in this key. In this case, you should utilize another reference guide to identify the organism. Several useful guides are listed below. AQUATIC ENTOMOLOGY by Patrick McCafferty A GUIDE TO COMMON FRESHWATER INVERTEBRATES OF NORTH AMERICA by J. Reese Voshell, Jr. AN INTRODUCTION TO THE AQUATIC INSECTS OF NORTH AMERICA by R.W. Merritt and K.W. Cummins Most organisms will be keyed to the family level, however several will not be identified beyond the Kingdom Animalia phylum, class, or order. -

Annual Newsletter and Bibliography of the International Society of Plecopterologists PERLA NO. 28, 2010
PERLA Annual Newsletter and Bibliography of The International Society of Plecopterologists Pteronarcella regularis (Hagen), Mt. Shasta City Park, California, USA. Photograph by Bill P. Stark PERLA NO. 28, 2010 Department of Bioagricultural Sciences and Pest Management Colorado State University Fort Collins, Colorado 80523 USA PERLA Annual Newsletter and Bibliography of the International Society of Plecopterologists Available on Request to the Managing Editor MANAGING EDITOR: Boris C. Kondratieff Department of Bioagricultural Sciences And Pest Management Colorado State University Fort Collins, Colorado 80523 USA E-mail: [email protected] EDITORIAL BOARD: Richard W. Baumann Department of Biology and Monte L. Bean Life Science Museum Brigham Young University Provo, Utah 84602 USA E-mail: [email protected] J. Manuel Tierno de Figueroa Dpto. de Biología Animal Facultad de Ciencias Universidad de Granada 18071 Granada, SPAIN E-mail: [email protected] Kenneth W. Stewart Department of Biological Sciences University of North Texas Denton, Texas 76203, USA E-mail: [email protected] Shigekazu Uchida Aichi Institute of Technology 1247 Yagusa Toyota 470-0392, JAPAN E-mail: [email protected] Peter Zwick Schwarzer Stock 9 D-36110 Schlitz, GERMANY E-mail: [email protected] 2 TABLE OF CONTENTS Subscription policy……………………………………………………………………….4 Publication of the Proceedings of the International Joint Meeting on Ephemeroptera and Plecoptera 2008…………………………………….………………….………….…5 Ninth North American Plecoptera Symposium………………………………………….6 -

Chapter 4 Ephemeroptera
Guide to Aquatic Invertebrates of the Upper Midwest | 2004 CHAPTER 4 EPHEMEROPTERA (Mayflies) EPHEMEROPTERA Citation: Bouchard, R.W., Jr. 2004. Guide to aquatic macroinvertebrates of the Upper Midwest. Water Resources Center, University of Minnesota, St. Paul, MN. 208 pp. Chapter 4 | EPHEMEROPTERA 47 Guide to Aquatic Invertebrates of the Upper Midwest | 2004 ORDER EPHEMEROPTERA Mayflies 4 Mayfly larvae are found in a variety of locations including lakes, wetlands, streams, and rivers; however, they are most common and diverse in lotic habitats. They are common and abundant in stream riffles and pools, at lake margins and in some cases lake bottoms. All mayfly larvae are aquatic with terrestrial adults. In most mayfly species the adult only lives for 1-2 days. Consequently, the majority of a mayfly’s life is spent in the water as a larva. The adult lifespan is so short there is no need for the insect to feed and therefore the adult does not possess functional mouthparts. Mayflies are often an indicator of good water quality because most mayflies are relatively intolerant of pollution. Mayflies are also an important food source for fish. Ephemeroptera Morphology Most mayflies have three caudal filaments (tails) (Figure 3.1) although in some taxa the terminal filament (middle tail) is greatly reduced and there appear to be only two caudal filaments (only one genus actually lacks the terminal filament). Mayflies have gills on the dorsal surface of the abdomen (Figure 3.1), but the number and shape of these gills vary widely between taxa. All mayflies possess only one EPHEMEROPTERA tarsal claw at the end of each leg (Figure 3.1). -
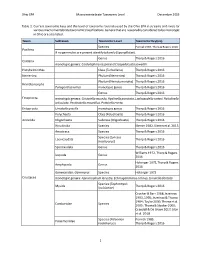
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Pennak 1989, Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Crocker & Barr 1968; Jezerinac 1993, 1995; Jezerinac & Thoma 1984; Taylor 2000; Thoma et al. Cambaridae Species 2005; Thoma & Stocker 2009; Crandall & De Grave 2017; Glon et al. 2018 Species (Palaemon Pennak 1989, Palaemonidae kadiakensis) Thorp & Rogers 2016 1 Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites Genus Morse et al. -

CHAPTER 4: EPHEMEROPTERA (Mayflies)
Guide to Aquatic Invertebrate Families of Mongolia | 2009 CHAPTER 4 EPHEMEROPTERA (Mayflies) EPHEMEROPTERA Draft June 17, 2009 Chapter 4 | EPHEMEROPTERA 45 Guide to Aquatic Invertebrate Families of Mongolia | 2009 ORDER EPHEMEROPTERA Mayflies 4 Mayfly larvae are found in a variety of locations including lakes, wetlands, streams, and rivers, but they are most common and diverse in lotic habitats. They are common and abundant in stream riffles and pools, at lake margins and in some cases lake bottoms. All mayfly larvae are aquatic with terrestrial adults. In most mayfly species the adult only lives for 1-2 days. Consequently, the majority of a mayfly’s life is spent in the water as a larva. The adult lifespan is so short there is no need for the insect to feed and therefore the adult does not possess functional mouthparts. Mayflies are often an indicator of good water quality because most mayflies are relatively intolerant of pollution. Mayflies are also an important food source for fish. Ephemeroptera Morphology Most mayflies have three caudal filaments (tails) (Figure 4.1) although in some taxa the terminal filament (middle tail) is greatly reduced and there appear to be only two caudal filaments (only one genus actually lacks the terminal filament). Mayflies have gills on the dorsal surface of the abdomen (Figure 4.1), but the number and shape of these gills vary widely between taxa. All mayflies possess only one tarsal claw at the end of each leg (Figure 4.1). Characters such as gill shape, gill position, and tarsal claw shape are used to separate different mayfly families. -

In Baltic Amber 85
Overview and descriptions of fossil stoneflies (Plecoptera) in Baltic amber 85 Entomologie heute 22 (2010): 85-97 Overview and Descriptions of Fossil Stoneflies (Plecoptera) in Baltic Amber Übersicht und Beschreibungen von fossilen Steinfliegen (Plecoptera) im Baltischen Bernstein CELESTINE CARUSO & WILFRIED WICHARD Summary: Three new fossil species of stoneflies (Plecoptera: Nemouridae and Leuctridae) from Eocene Baltic amber are being described: Zealeuctra cornuta n. sp., Lednia zilli n. sp., and Podmosta attenuata n. sp.. Extant species of these three genera are found in Eastern Asia and in the Nearctic region. It is very probably that the genera must have been widely spread across the northern hemisphere in the Cretaceous period, before Europe was an archipelago in Eocene. The current state of knowledge about the seventeen Plecoptera species of Baltic amber is shortly presented. Due to discovered homonymies, the following nomenclatural corrections are proposed: Leuctra fusca Pictet, 1856 in Leuctra electrofusca Caruso & Wichard, 2010 and Nemoura affinis Berendt, 1856 in Nemoura electroaffinis Caruso & Wichard, 2010. Keywords: Fossil insects, fossil Plecoptera, Eocene, paleobiogeography Zusammenfassung: In dieser Arbeit werden drei neue fossile Steinfliegen-Arten (Plecoptera: Nemouridae und Leuctridae) des Baltischen Bernsteins beschrieben: Zealeuctra cornuta n. sp., Lednia zilli n. sp., Podmosta attenuata n. sp.. Rezente Arten der drei Gattungen sind in Ostasien und in der nearktischen Region nachgewiesen. Sehr wahrscheinlich breiteten sich die Gattungen in der Krei- dezeit über die nördliche Hemisphäre aus, noch bevor Europa im Eozän ein Archipel war. Der gegenwärtige Kenntnisstand über die siebzehn Plecoptera Arten des Baltischen Bernsteins wird kurz dargelegt. Wegen bestehender Homonymien werden folgende nomenklatorische Korrekturen vorgenommen: Leuctra fusca Pictet, 1856 in Leuctra electrofusca Caruso & Wichard, 2010 und Nemoura affinis Berendt, 1856 in Nemoura electroaffinis Caruso & Wichard, 2010. -

ARTHROPODA Subphylum Hexapoda Protura, Springtails, Diplura, and Insects
NINE Phylum ARTHROPODA SUBPHYLUM HEXAPODA Protura, springtails, Diplura, and insects ROD P. MACFARLANE, PETER A. MADDISON, IAN G. ANDREW, JOCELYN A. BERRY, PETER M. JOHNS, ROBERT J. B. HOARE, MARIE-CLAUDE LARIVIÈRE, PENELOPE GREENSLADE, ROSA C. HENDERSON, COURTenaY N. SMITHERS, RicarDO L. PALMA, JOHN B. WARD, ROBERT L. C. PILGRIM, DaVID R. TOWNS, IAN McLELLAN, DAVID A. J. TEULON, TERRY R. HITCHINGS, VICTOR F. EASTOP, NICHOLAS A. MARTIN, MURRAY J. FLETCHER, MARLON A. W. STUFKENS, PAMELA J. DALE, Daniel BURCKHARDT, THOMAS R. BUCKLEY, STEVEN A. TREWICK defining feature of the Hexapoda, as the name suggests, is six legs. Also, the body comprises a head, thorax, and abdomen. The number A of abdominal segments varies, however; there are only six in the Collembola (springtails), 9–12 in the Protura, and 10 in the Diplura, whereas in all other hexapods there are strictly 11. Insects are now regarded as comprising only those hexapods with 11 abdominal segments. Whereas crustaceans are the dominant group of arthropods in the sea, hexapods prevail on land, in numbers and biomass. Altogether, the Hexapoda constitutes the most diverse group of animals – the estimated number of described species worldwide is just over 900,000, with the beetles (order Coleoptera) comprising more than a third of these. Today, the Hexapoda is considered to contain four classes – the Insecta, and the Protura, Collembola, and Diplura. The latter three classes were formerly allied with the insect orders Archaeognatha (jumping bristletails) and Thysanura (silverfish) as the insect subclass Apterygota (‘wingless’). The Apterygota is now regarded as an artificial assemblage (Bitsch & Bitsch 2000). -

Adhesion Performance in the Eggs of the Philippine Leaf Insect Phyllium Philippinicum (Phasmatodea: Phylliidae)
insects Article Adhesion Performance in the Eggs of the Philippine Leaf Insect Phyllium philippinicum (Phasmatodea: Phylliidae) Thies H. Büscher * , Elise Quigley and Stanislav N. Gorb Department of Functional Morphology and Biomechanics, Institute of Zoology, Kiel University, Am Botanischen Garten 9, 24118 Kiel, Germany; [email protected] (E.Q.); [email protected] (S.N.G.) * Correspondence: [email protected] Received: 12 June 2020; Accepted: 25 June 2020; Published: 28 June 2020 Abstract: Leaf insects (Phasmatodea: Phylliidae) exhibit perfect crypsis imitating leaves. Although the special appearance of the eggs of the species Phyllium philippinicum, which imitate plant seeds, has received attention in different taxonomic studies, the attachment capability of the eggs remains rather anecdotical. Weherein elucidate the specialized attachment mechanism of the eggs of this species and provide the first experimental approach to systematically characterize the functional properties of their adhesion by using different microscopy techniques and attachment force measurements on substrates with differing degrees of roughness and surface chemistry, as well as repetitive attachment/detachment cycles while under the influence of water contact. We found that a combination of folded exochorionic structures (pinnae) and a film of adhesive secretion contribute to attachment, which both respond to water. Adhesion is initiated by the glue, which becomes fluid through hydration, enabling adaption to the surface profile. Hierarchically structured pinnae support the spreading of the glue and reinforcement of the film. This combination aids the egg’s surface in adapting to the surface roughness, yet the attachment strength is additionally influenced by the egg’s surface chemistry, favoring hydrophilic substrates. -

DATA COLLECTION SURVEY on WATER RESOURCE MANAGEMENT and AGRICULTURE IRRIGATION in the REPUBLIC of IRAQ FINAL REPORT April 2016 the REPUBLIC of IRAQ
DATA COLLECTION SURVEY ON WATER RESOURCE MANAGEMENT AND AGRICULTURE IRRIGATION IN THE REPUBLIC OF IRAQ FINAL REPORT April 2016 REPORT IRAQ FINAL THE REPUBLIC OF IN IRRIGATION AGRICULTURE AND RESOURCE MANAGEMENT WATER ON COLLECTION SURVEY DATA THE REPUBLIC OF IRAQ DATA COLLECTION SURVEY ON WATER RESOURCE MANAGEMENT AND AGRICULTURE IRRIGATION IN THE REPUBLIC OF IRAQ FINAL REPORT April 2016 Japan International Cooperation Agency (JICA) NTC International Co., Ltd. 7R JR 16-008 英文 118331.402802.28.4.14 作業;藤川 Directorate Map Dohuk N Albil Nineveh Kiekuk As-Sulaymaniyyah Salah ad-Din Tigris river Euphrates river Bagdad Diyala Al-Anbar Babil Wasit Karbala Misan Al-Qadisiyan Al-Najaf Dhi Qar Al-Basrah Al-Muthanna Legend Irrigation Area International boundary Governorate boundary River Location Map of Irrigation Areas ( ii ) Photographs Kick-off meeting with MoWR officials at the conference Explanation to D.G. Directorate of Legal and Contracts of room of MoWR MoWR on the project formulation (Conference room at Both parties exchange observations of Inception report. MoWR) Kick-off meeting with MoA officials at the office of MoA Meeting with MoP at office of D.G. Planning Both parties exchange observations of Inception report. Both parties discussed about project formulation Courtesy call to the Minister of MoA Meeting with representatives of WUA assisted by the JICA JICA side explained the progress of the irrigation sector loan technical cooperation project Phase 1. and further project formulation process. (Conference room of MoWR) ( iii ) Office of AL-Zaidiya WUA AL-Zaidiya WUA office Site field work to investigate WUA activities during the JICA team conducted hearing investigation on water second field survey (Dhi-Qar District) management, farming practice of WUA (Dhi-Qar District) Piet Ghzayel WUA Piet Ghzayel WUA Photo shows the eastern portion of the farmland.