Application of Squarate Ester Cascade Reactions to The
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
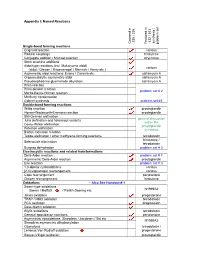
Appendix I: Named Reactions Single-Bond Forming Reactions Co
Appendix I: Named Reactions 235 / 335 432 / 533 synthesis / / synthesis Covered in Covered Featured in problem set problem Single-bond forming reactions Grignard reaction various Radical couplings hirstutene Conjugate addition / Michael reaction strychnine Stork enamine additions Aldol-type reactions (incl. Mukaiyama aldol) various (aldol / Claisen / Knoevenagel / Mannich / Henry etc.) Asymmetric aldol reactions: Evans / Carreira etc. saframycin A Organocatalytic asymmetric aldol saframycin A Pseudoephedrine glycinamide alkylation saframycin A Prins reaction Prins-pinacol reaction problem set # 2 Morita-Baylis-Hillman reaction McMurry condensation Gabriel synthesis problem set #3 Double-bond forming reactions Wittig reaction prostaglandin Horner-Wadsworth-Emmons reaction prostaglandin Still-Gennari olefination general discussion Julia olefination and heteroaryl variants within the Corey-Winter olefination prostaglandin Peterson olefination synthesis Barton extrusion reaction Tebbe olefination / other methylene-forming reactions tetrodotoxin hirstutene / Selenoxide elimination tetrodotoxin Burgess dehydration problem set # 3 Electrocyclic reactions and related transformations Diels-Alder reaction problem set # 1 Asymmetric Diels-Alder reaction prostaglandin Ene reaction problem set # 3 1,3-dipolar cycloadditions various [2,3] sigmatropic rearrangement various Cope rearrangement periplanone Claisen rearrangement hirstutene Oxidations – Also See Handout # 1 Swern-type oxidations (Swern / Moffatt / Parikh-Doering etc. N1999A2 Jones oxidation -

Elimination Reactions Are Described
Introduction In this module, different types of elimination reactions are described. From a practical standpoint, elimination reactions widely used for the generation of double and triple bonds in compounds from a saturated precursor molecule. The presence of a good leaving group is a prerequisite in most elimination reactions. Traditional classification of elimination reactions, in terms of the molecularity of the reaction is employed. How the changes in the nature of the substrate as well as reaction conditions affect the mechanism of elimination are subsequently discussed. The stereochemical requirements for elimination in a given substrate and its consequence in the product stereochemistry is emphasized. ELIMINATION REACTIONS Objective and Outline beta-eliminations E1, E2 and E1cB mechanisms Stereochemical considerations of these reactions Examples of E1, E2 and E1cB reactions Alpha eliminations and generation of carbene I. Basics Elimination reactions involve the loss of fragments or groups from a molecule to generate multiple bonds. A generalized equation is shown below for 1,2-elimination wherein the X and Y from two adjacent carbon atoms are removed, elimination C C C C -XY X Y Three major types of elimination reactions are: α-elimination: two atoms or groups are removed from the same atom. It is also known as 1,1-elimination. H R R C X C + HX R Both H and X are removed from carbon atom here R Carbene β-elimination: loss of atoms or groups on adjacent atoms. It is also H H known as 1,2- elimination. R C C R R HC CH R X H γ-elimination: loss of atoms or groups from the 1st and 3rd positions as shown below. -

Application of Complex Aldol Reactions to the Total Synthesis of Phorboxazole B
J. Am. Chem. Soc. 2000, 122, 10033-10046 10033 Application of Complex Aldol Reactions to the Total Synthesis of Phorboxazole B David A. Evans,* Duke M. Fitch,1 Thomas E. Smith, and Victor J. Cee Contribution from the Department of Chemistry and Chemical Biology, HarVard UniVersity, Cambridge, Massachusetts 02138 ReceiVed June 29, 2000 Abstract: The synthesis of phorboxazole B has been accomplished in 27 linear steps and an overall yield of 12.6%. The absolute stereochemistry of the C4-C12,C33-C38, and C13-C19 fragments was established utilizing catalytic asymmetric aldol methodology, while the absolute stereochemistry of the C20-C32 fragment was derived from an auxiliary-based asymmetric aldol reaction. All remaining chirality was incorporated through internal asymmetric induction, with the exception of the C43 stereocenter which was derived from (R)-trityl glycidol. Key fragment couplings include a stereoselective double stereodifferentiating aldol reaction, a metalated oxazole alkylation, an oxazole-stabilized Wittig olefination, and a chelation-controlled addition of the fully elaborated alkenyl metal side chain. Introduction and HT29 (3.31 × 10-10 M), as well as leukemia CCRF-CBM (2.45 × 10-10 M), prostate cancer PC-3 (3.54 × 10-10 M), and Phorboxazoles A (1)andB(2) are marine natural products breast cancer MCF7 cell lines (5.62 × 10-10 M).2b In addition isolated from a recently discovered species of Indian Ocean to this impressive anticancer activity, phorboxazoles A and B sponge (genus Phorbas sp.) near Muiron Island, Western also exhibit potent in vitro antifungal activity against Candida Australia.2 These substances are representative of a new class albicans and Saccharomyces carlsbergensis at 0.1 µg/disk in of macrolides differing only in their C hydroxyl-bearing 13 the agar disk diffusion assay.2a stereocenters. -

Olefination Reactions Lecture Notes OTBS OPMB O
Olefination Reactions Lecture Notes OTBS OPMB O Key Reviews: O Me O Me XX Wittig Reaction K. C. Nicolaou and co-workers, Ann. 1997, 1283. Horner-Wadsworth-Emmons and Tebbe Olefinations S. E. Kelly, Comprehensive Org. Synth. 1991, Vol. 1, 729. Peterson Olefination D. J. Ager, Synthesis 1984, 384. Julia (Julia-Lythgoe) Olefination B. M. Trost, Bull. Chem. Soc. Jpn. 1988, 61, 107. Wittig Olefination: Background and Principles R1 n-BuLi, Ph P 3 O X LDA, R R1 LiHMDS PPh + 1 3 X Ph3P + R1 Ph3P pKa = 18-20 ylide when R = alkyl, H Ph Ph Ph Ph Ph R1 P Ph P -[Ph3P=O] O R1 O R strong bond 1 formation drives reaction oxaphosphatane betaine G. Wittig and G. Schollkopf, Chem. Ber. 1954, 87, 1318. Wittig Olefination: Background and Principles Stereoselectivity with non-stabilized ylides Me OMe Me Ph3P Ph3P Ph3P Me Ph3P Ph3P Not stable; must be made in situ and used immediately Wittig Olefination: Background and Principles Stereoselectivity with non-stabilized ylides Me OMe Me Ph3P Ph3P Ph3P Me Ph3P Ph3P Not stable; must be made in situ and used immediately Addition to carbonyl is an irreversible and concerted [2+2] cycloaddition such that the R groups on the aldehyde and the ylide are as far apart as possible PPh3 Ph3P O H H H O + Me Me Ph3P R O R H -[Ph3P=O] As the size of the R groups increases, selectivity for Z-alkene increases Me Nonpolar solvents favor initial addition Polar solvents favor the elimination Z-alkene Wittig Olefination: Background and Principles Stereoselectivity with stabilized ylides Me O O Ph P Ph3P 3 Ph3P Me OEt semistabilized Incredibly stable; not moisture sensitive, can be chromatographed Price for stability is lower reactivity: reacts well with aldehydes, slowly with ketones Wittig Olefination: Background and Principles Stereoselectivity with stabilized ylides Me O O Ph P Ph3P 3 Ph3P Me OEt semistabilized Incredibly stable; not moisture sensitive, can be chromatographed Price for stability is lower reactivity: reacts well with aldehydes, slowly with ketones Initial addition to carbonyl is reversible so the thermodynamic elimination product results. -

University of California
UC Riverside UC Riverside Electronic Theses and Dissertations Title Towards a Catalytic Asymmetric Cope Rearrangement and the Synthesis and Self-Assembly of Metal-Coordinated Hosts Permalink https://escholarship.org/uc/item/4gc654st Author Moehlig, Melissa Padilla Publication Date 2013 Peer reviewed|Thesis/dissertation eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA RIVERSIDE Towards a Catalytic Asymmetric Cope Rearrangement and the Synthesis and Self- Assembly of Metal-Coordinated Hosts A Dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Chemistry by Melissa Padilla Moehlig December 2013 Dissertation Committee: Dr. Richard J. Hooley, Chairperson Dr. Catharine H. Larsen Dr. Michael C. Pirrung Copyright by Melissa Padilla Moehlig 2013 The Dissertation of Melissa Padilla Moehlig is approved: Committee Chairperson University of California, Riverside ACKNOWLEDGEMENTS Graduate school has been one of the most rewarding and yet the most exhausting and stressful times of my life. It would not have survived without the help of several people. I would like to thank Dr. Courtney Meyet, Dr. Katherine Djernes, and Yoo-Jin Ghang for their friendship and all the laughs that were necessary to keep me sane. I would like to thank Michael Young, Hou Ung, and Jay-Ar Bendo for our morning coffee breaks, they were crucial to get my day started. I would like to thank Prof. Larsen for teaching me to be independent. I would like to thank Prof. Hooley for all his guidance over the past five years. You are truly a great mentor and I don’t think I would have survived graduate school without your help and advice. -

A Acetophenones, 10 Horner–Emmons Olefination, 7 Acrylonitrile
Index A Alkenylmetals, 38 Acetophenones, 10 Alkenylsilanes, 18 Horner–Emmons olefination, 7 Alkenylstannanes, 18 Acrylonitrile, Z-selective cross metathesis, 46 α-Alkoxycyclopentenone, 25 Acylgermanes, 18 (E)-β-Alkoxy divinyl ketones, 25 Acyl phosphonates, olefination, Co(TPP), 160 α-Alkoxyketones, 12 Acylsilanes, 18 Alkyl aryl ketones, 16 Acylstannanes, 18 Alkylidenephosphoranes, olefination, 208 Africanol, 187 Alkynes, conjugated dienes, 98 Aldehydes, 197 Z-alkenes, 40, 43 Fe(II)(TTP), olefination, 151 hydrofluorination, 60 Mo-based catalytic olefination, 149 hydrofunctionalizations, 134 MTO-catalyzed olefination, 149 hydrometalation, 43 olefination, 6 2-Alkynoate, conjugated dienes, 109 diazocarbonyl reagents, 161 Alkynoates, 108 oxygen-substituted phosphorus Alkynyl ketones, 16 ylides, 156 Allenamides, 126 Ph3As, 157 Allenoates, 108 Rh-catalyzed methylenation, 157 one-pot Wittig reaction, 213 Ru-catalyzed olefination, 157 Allylation, 54 Aldimines, olefination, 28 Allyl ethers, chiral, 258 Alkaloids, 171 N-Allylhydrazones, 107 Alkene cross metathesis, 178 Allylic alcohols, trisubstituted, 207 Alkene metathesis, catalytic Allylic carbonates, one-pot Wittig enantioselective, 187 reaction, 213 catalytic Z-selective, 189 Allylic gem-difluorides, 67 selective, 163 Allylic substitution, 54 selective relay, 174 Allylidenetriethylphosphoranes, 205 Alkenes, 197 Allylsulfides, multisubstituted, synthesis, 239 desulfurization, 16 tetrasubstituted, 1 Amino acid, (Z)-fluoroalkenyl moiety, 83 Z-Alkenes, stereoselective synthesis, 33 α-Aminoketones, 14 Alkenylboronic -

New Application of the Julia Olefination for the Synthesis of Tyr-Gly E-Alkene and Carba Isostere Pseudopeptides Cécile Charrier, Laurent Ettouati, Joelle Paris
New application of the Julia olefination for the synthesis of Tyr-Gly E-alkene and carba isostere pseudopeptides Cécile Charrier, Laurent Ettouati, Joelle Paris To cite this version: Cécile Charrier, Laurent Ettouati, Joelle Paris. New application of the Julia olefination for the synthesis of Tyr-Gly E-alkene and carba isostere pseudopeptides. Tetrahedron Letters, Elsevier, 1999, 40 (31), pp.5705 - 5707. 10.1016/S0040-4039(99)01139-9. hal-01651962 HAL Id: hal-01651962 https://hal.archives-ouvertes.fr/hal-01651962 Submitted on 15 Feb 2018 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. New Application of The Julia Olefination for The Synthesis of Tyr-Gly E-Alkene and Carba Isostere Pseudopeptides Cécile CHARRIER, Laurent ETTOUATI* and Joelle PARIS Université Claude Bernard Lyon I, Institut des Sciences Pharmaceutiques et Biologiques, Laboratoire de Chimie Thérapeutique, 8 avenue Rockefeller F-69373 Lyon cedex 08, France Abstract: A new application of the Julia olefination to the synthesis of TyrΨ[E-CH=CH]Gly and TyrΨ[CH2CH2]Gly pseudopeptides is described -

Advanced Organic Chemistry PART A: Structure and Mechanisms PART B: Reactions and Synthesis Advanced Organic FIFTH Chemistry EDITION
Advanced Organic FIFTH Chemistry EDITION Part B: Reactions and Synthesis Advanced Organic Chemistry PART A: Structure and Mechanisms PART B: Reactions and Synthesis Advanced Organic FIFTH Chemistry EDITION Part B: Reactions and Synthesis FRANCIS A. CAREY and RICHARD J. SUNDBERG University of Virginia Charlottesville, Virginia 123 Francis A. Carey Richard J. Sundberg Department of Chemistry Department of Chemistry University of Virginia University of Virginia Charlottesville, VA 22904 Charlottesville, VA 22904 Library of Congress Control Number: 2006939782 ISBN-13: 978-0-387-68350-8 (hard cover) e-ISBN-13: 978-0-387-44899-2 ISBN-13: 978-0-387-68354-6 (soft cover) Printed on acid-free paper. ©2007 Springer Science+Business Media, LLC All rights reserved. This work may not be translated or copied in whole or in part without the written permission of the publisher (Springer Science+Business Media, LLC, 233 Spring Street, New York, NY 10013, USA), except for brief excerpts in connection with reviews or scholarly analysis. Use in connection with any form of information storage and retrieval, electronic adaptation, computer software, or by similar or dissimilar methodology now know or hereafter developed is forbidden. The use in this publication of trade names, trademarks, service marks and similar terms, even if they are not identified as such, is not to be taken as an expression of opinion as to whether or not they are subject to proprietary rights. 98765432(corrected 2nd printing, 2008) springer.com Preface The methods of organic synthesis have continued to advance rapidly and we have made an effort to reflect those advances in this Fifth Edition. -

Advanced Functonal Group Transformatons and Reactons C
Advanced Func+onal Group Transformaons and Reacons C-X where X is an heteroatom (N,O,halogen) C=C (double bonds) C-C (single bonds) Amides and esters are two of the most fundamental and significant functional groups in chemistry, biochemistry, and materials science. Therefore, many dehydrocondensing reagents for synthesizing amide and ester bonds from carboxylic acids and amines or alcohols have been developed over the past century. March 20, 2014 (1) MontalbeE, C. A. G. N.; Falque, V. Tetrahedron 2005, 61, 10827−10852. (2) El-Faham, A.; Albericio, F. Chem. Rev. 2011, 111, 6557−6602. (3) Paabiraman, V. R.; Bode, J. W. Nature 2011, 480, 471−479. Dehydraon reacons In the following some examples for ac4vaon of carboxylic acids towards nucleophiles are discussed: Nu: Oxygen and nitrogen nucleophiles DCC amide couplings Carboxylic acids are relavely unreac4ve with nucleophiles due high lying LUMO and bad leaving group (OH). A solu4on is the use of reagents which selec4vely ac4vate a carboxyl group towards nucleophilic subs4tu4on. Carbodiimides such as N,N‘- dicyclohexylcarbodiimide (DCC) are frequently used for amide and ester bond formaon. As a driving force for the reac4on serves the formaon of urea (DCU). However, in the case of pep4de synthesis the ac4vaon can leads to racemizaon of stereocenter at α-C posi4on during the reac4on. Therefore reagents as HOBt, HBTU etc. were introduced as co-coupling agents. They react faster with the ac4vated intermediate than the racemizaon at α-C posion occurs. Other coupling agents frequently used: Acid chloride using DMF/oxalyl chloride The standard way to ac4vate a carboxyl group is to convert it in to an acid chloride. -

The Merck Index
Browse Organic Name Reactions ● Preface ● 4CC ● Acetoacetic Ester Condensation ● Acetoacetic Ester Synthesis ● Acyloin Condensation ● Addition ● Akabori Amino Acid Reactions ● Alder (see Diels-Alder Reaction) ● Alder-Ene Reaction ● Aldol Reaction (Condensation) ● Algar-Flynn-Oyamada Reaction ● Allan-Robinson Reaction ● Allylic Rearrangements ● Aluminum Alkoxide Reduction ● Aluminum Alkoxide Reduction (see Meerwein-Ponndorf-Verley Reduction) ● Amadori Rearrangement ● Amidine and Ortho Ester Synthesis ● Aniline Rearrangement ● Arbuzov (see Michaelis-Arbuzov Reaction) ● Arens-van Dorp Synthesis ● Arndt-Eistert Synthesis ● Auwers Synthesis ● Babayan (see Favorskii-Babayan Synthesis) ● Bachmann (see Gomberg-Bachmann Reaction) ● Bäcklund (see Ramberg-Bäcklund Reaction) ● Baeyer-Drewson Indigo Synthesis ● Baeyer-Villiger Reaction ● Baker-Venkataraman Rearrangement ● Bakshi (see Corey-Bakshi-Shibata Reduction) ● Balz-Schiemann Reaction ● Bamberger Rearrangement ● Bamford-Stevens Reaction ● Barbier(-type) Reaction ● Barbier-Wieland Degradation ● Bart Reaction ● Barton Decarboxylation ● Barton Deoxygenation ● Barton Olefin Synthesis ● Barton Reaction http://themerckindex.cambridgesoft.com/TheMerckIndex/NameReactions/TOC.asp (1 of 17)19/4/2005 20:00:21 Browse Organic Name Reactions ● Barton-Kellogg Reaction ● Barton-McCombie Reaction ● Barton-Zard Reaction ● Baudisch Reaction ● Bauer (see Haller-Bauer Reaction) ● Baumann (see Schotten-Baumann Reaction) ● Baylis-Hillman Reaction ● Béchamp Reduction ● Beckmann Fragmentation ● Beckmann Rearrangement -

Chemistry 432 – Lecture Notes Updated: Spring 2016
Chemistry 432 – Lecture Notes Updated: Spring 2016 Course Organization: Things You Need to Know 1. Named Reactions and Reagents 2. Vocabulary 3. Concepts 4. HOW TO DO SYNTHESIS Nucleophiles and Electrophiles: The Basis of Organic Chemistry notes_01 Synthesis 1: Strychnine Woodward, 1954 - Nobel 1965 Classics I, 21 Reactions: · Fischer indole synthesis · Indole addition · Dieckmann condensation · Allylic rearrangement Concepts: · Retrosynthesis · Substructure Recognition notes_02 Properties: · A poison from Southeast Asian rainforests · Known in Europe from the 16th century · Isolation in 1818 (Pelletier and Caventou) · Structure determined in 1946; X-ray in 1956 · 6 contiguous stereocentres! Definition: Retrosynthesis - A technique for transforming the structure of a synthetic target into a sequence of simpler structures, along a pathway which ultimately leads to known or commercially available starting materials. notes_04 - E.J. Corey, Nobel 1990 Retrosynthesis: notes_03 Methodology: 1.1 Fischer Indole Synthesis: notes_05 - most commonly done with ZnCl2 Continuing Steps: notes_06 - How does the ozonolysis work here? - Let’s come back to that after the next section. Other Syntheses of Strychnine: Magnus, 1992 – J.Am.Chem.Soc. 1992, 4403. - 38 years from 1st to 2nd synthesis! Stork, 1992 – lecture (Iscia Porto, Italy). Kuehne, 1993 – J.Org.Chem. 1993, 7490. Overman, 1993 – J.Am.Chem.Soc. 1993, 9293. - 1st enantioselective synthesis Rawal, 1994 – J.Org.Chem. 1994, 2685. Martin, 1996 – J.Am.Chem.Soc. 1996, 9804. Bonjoch, 1999 – Angew.Chem.Int.Ed. 1999, 395. Vollhardt, 2000 – Org.Lett. 2000, 2479. Shibasaki, 2002 – J.Am.Chem.Soc. 2002, 14546. Mori, 2002 – Angew.Chem.Int.Ed. 2002, 1934. Bodwell, 2002 – Angew.Chem.Int.Ed. 2002, 3261. Fukuyama, 2004 – J.Am.Chem.Soc. -

Synthesis of Chiral, Enantiopure Allylic Amines by the Julia
molecules Article ArticleSynthesis of Chiral, Enantiopure Allylic Amines by Synthesisthe Julia Olefination of Chiral, Enantiopure of α-Amino AllylicEsters Amines by the Julia Olefination of α-Amino Esters Fabio Benedetti *, Federico Berti, Lidia Fanfoni, Michele Garbo, Giorgia Regini and FabioFulvia Benedetti Felluga * *, Federico Berti, Lidia Fanfoni, Michele Garbo, Giorgia Regini and Fulvia Felluga * Department of Chemical and Pharmaceutical Sciences, University of Trieste, via Giorgieri 1. 34127 Trieste, DepartmentItaly; [email protected] of Chemical (F.B.); and [email protected] Pharmaceutical Sciences, (L.F.); University [email protected] of Trieste, via Giorgieri (M.G.); 1. 34127 Trieste, Italy; [email protected]@phd.units.it (F.B.); [email protected] (G.R.) (L.F.); [email protected] (M.G.); [email protected]* Correspondence: [email protected] (G.R.) (F.B.); [email protected] (F.F.); Tel.: +39-040-5583919 (F.B.); * Correspondence:+39-040-5583924 (F.F.) [email protected] (F.B.); [email protected] (F.F.); Tel.: +39-040-5583919 (F.B.); +39-040-5583924 (F.F.) Academic Editors: Carlo Siciliano and Constantinos M. Athanassopoulos AcademicReceived: 24 Editors: May 2016; Carlo Accepted: Siciliano and14 Ju Constantinosne 2016; Published: M. Athanassopoulos 20 June 2016 Received: 24 May 2016; Accepted: 14 June 2016; Published: 21 June 2016 Abstract: The four-step conversion of a series of N-Boc-protected L-amino acid methyl esters into Abstract: The four-step conversion of a series of N-Boc-protected L-amino acid methyl esters into enantiopure N-Boc allylamines by a modified Julia olefination is described. Key steps include the enantiopure N-Boc allylamines by a modified Julia olefination is described.