Olefination Reactions Lecture Notes OTBS OPMB O
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supporting Information a Convenient Chromatography-Free Method For
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry This journal is © The Royal Society of Chemistry 2012 Supporting Information A Convenient Chromatography-Free Method for the Purification of Alkenes Produced in the Wittig Reaction Peter A. Byrne, Kamalraj V. Rajendran, Jimmy Muldoon and Declan G. Gilheany Centre for Synthesis and Chemical Biology, School of Chemistry and Chemical Biology, University College Dublin, Belfield, Dublin 4, Ireland 1. General Experimental 2 2. Synthesis of phosphonium salts 4 3. Procedures for Wittig reactions & phosphine oxide removal 11 4. Characterisation of purified alkenes 15 5. Reduction of phosphine chalcogenides 34 6. Synthesis & isolation of neomenthyl chloride and reduction of phosphine oxide-by-product 37 7. NMR spectra of phosphonium salts 42 8. NMR spectra of purified alkenes and regenerated phosphines from Wittig reactions 56 9. NMR spectra of phosphines produced by reduction of phosphine chalcogenides 85 10. NMR spectra of purified products from Appel-type reactions 90 11. References 91 1 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry This journal is © The Royal Society of Chemistry 2012 1. General Experimental All chemicals were supplied by Aldrich, with the exception of Zeoprep silica, 2- methylbenzaldehyde ( o-tolualdehyde, Fluka), ( tert -butoxycarbonylmethyl)- -1 triphenylphosphonium bromide (Fluka), 1 mol L LiAlH 4 in THF (Acros Organics) and Merck standardised alumina 90. All chemicals were used without further purification except diethyl ether, toluene, and THF, which were processed through an Innovative Technology Inc. Pure Solv-400-3-MD solvent purification (Grubbs still) system and stored in Strauss flasks under a nitrogen atmosphere, and ethyl acetate and dichloromethane, which were degassed by passing a stream of dry nitrogen gas (oxygen- free) through the solvent for one hour for the purposes of work-ups in phosphine syntheses. -

Studies Directed Towards the Stereoselective Total Synthesis of Miyakolide
Studies Directed Towards the Stereoselective Total Synthesis of Miyakolide by Jinhua Song Submitted to the Department of Chemistry in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy in Organic Chemistry at the Massachusetts Institute of Technology February, 1999 @1999 Jinhua Song All rights Reserved The author hereby grants MIT permissions to reproduce and to distribute publicly paper and electronic copies of this thesis document in whole or in part. Signature of Author: Department of Chemistry September 25, 1998 Certified by: Professor Satoru Masamune A. C. Cope Professor of Chemistry Thesis Supervisor Accepted by:, ProfessotDietmar Seyferth, Chairman Departmental Committee on Graduate Students MASSACHUSETTS INSTITUTE OF TECHNOLOGY LrL J This doctoral thesis has been examined by a committee of the Department of Chemistry as follows: Professor Timothy M. Swager Chairman Professor Satoru Masamune Thesis Supervisor Professor Rick L. Danheiser , 2 Studies Directed Towards the Stereoselective Total Synthesis of Miyakolide by Jinhua Song Submitted to the Department of Chemistry on September 25, 1998, in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy in Organic Chemistry Abstract Presented are the stereoselective syntheses of the A (C18-C28), B (C14-C17), C (C6-C13), D (Cl-C5), C'D' (C1-C13) fragments and the efficient coupling of B and C'D' fragments of the marine natural product miyakolide, a 24-membered polyketide macrolide which exhibits anti-cancer activity. Fragment A was synthesized from the chiral aldehyde 4-4 through the successful application of the newly developed boron mediated anti-selective aldol methodology using the chiral ester 3-4. -

The Synthesis and Applications of N-Alkenyl Aziridines
The Synthesis and Applications of N-Alkenyl Aziridines by Nicholas A. Afagh A thesis submitted in conformity with the requirements for the degree of Master of Science Department of Chemistry University of Toronto © Copyright by Nicholas A. Afagh 2010 The Synthesis and Applications of N-Alkenyl Aziridines Nicholas A. Afagh Master of Science Department of Chemistry University of Toronto 2010 Abstract N-alkenyl aziridines are a unique class of molecules that do not behave as typical enamines as a result of the inability of the nitrogen atom lone-pair of electrons to delocalize. The attenuated nucleophilicity of these enamines presents opportunities for the selective functionalization and reactivity not available to classical enamines. An operationally simple and mild copper-mediated coupling has been developed that facilitates the preparation of a broad range of N-alkenyl aziridines not available through existing methods. The preparation and reactivity of highly- functionalized N-alkenyl aziridines are reported. Also reported is the application of the chemoselective amine/aldehyde/alkyne (A 3) multicomponent coupling involving amphoteric aziridine aldehydes as the aldehyde component. This coupling allows access to propargyl amines with pendent aziridine functionality. ii Acknowledgments First and foremost, I would like to thank my supervisor, Professor Andrei K. Yudin for his continuous support and encouragement over the past two years. His wealth of knowledge and profound insight into all matters chemistry made for many interesting discussions. In addition, I would like to thank all the members of the Yudin group past and present with whom I have had the distinct pleasure of working alongside and shared many late evenings. -

Radical Approaches to Alangium and Mitragyna Alkaloids
Radical Approaches to Alangium and Mitragyna Alkaloids A Thesis Submitted for a PhD University of York Department of Chemistry 2010 Matthew James Palframan Abstract The work presented in this thesis has focused on the development of novel and concise syntheses of Alangium and Mitragyna alkaloids, and especial approaches towards (±)-protoemetinol (a), which is a key precursor of a range of Alangium alkaloids such as psychotrine (b) and deoxytubulosine (c). The approaches include the use of a key radical cyclisation to form the tri-cyclic core. O O O N N N O O O H H H H H H O N NH N Protoemetinol OH HO a Psychotrine Deoxytubulosine b c Chapter 1 gives a general overview of radical chemistry and it focuses on the application of radical intermolecular and intramolecular reactions in synthesis. Consideration is given to the mediator of radical reactions from the classic organotin reagents, to more recently developed alternative hydrides. An overview of previous synthetic approaches to a range of Alangium and Mitragyna alkaloids is then explored. Chapter 2 follows on from previous work within our group, involving the use of phosphorus hydride radical addition reactions, to alkenes or dienes, followed by a subsequent Horner-Wadsworth-Emmons reaction. It was expected that the tri-cyclic core of the Alangium alkaloids could be prepared by cyclisation of a 1,7-diene, using a phosphorus hydride to afford the phosphonate or phosphonothioate, however this approach was unsuccessful and it highlighted some limitations of the methodology. Chapter 3 explores the radical and ionic chemistry of a range of silanes. -

Properties of Silicon Hafensteiner
Baran Lab Properties of Silicon Hafensteiner Si vs. C Siliconium Ion - Si is less electronegative than C - Not believed to exist in any reaction in solution - More facile nucleophilic addition at Si center J. Y. Corey, J. Am. Chem. Soc. 1975, 97, 3237 - Pentacoordinate Si compounds have been observed Average BDE (kcal/mol) MeSiF4 NEt4 Ph3SiF2 NR4 C–C C–Si Si–Si C–F Si–F 83 76 53 116 135 - Lack of cation justified by high rate of bimolecular reactivity at Si C–O Si–O C–H Si–H Mechanism of TMS Deprotection 86 108 83 76 OTMS O Average Bond Lengths (Å) C–C C–Si C–O Si–O 1.54 1.87 1.43 1.66 Workup Si Si Silicon forms weak p-Bonds O O F F NBu4 p - C–C = 65 kcal/mol p - C–Si = 36 kcal/mol Pentavalent Silicon Baran Lab Properties of Silicon Hafensteiner Nucleophilic addition to Si b-Silicon effect and Solvolysis F RO–SiMe3 RO F–SiMe3 SiMe3 Me H H vs. Me3C H Me3C H OSiMe O Li 3 H OCOCF H OCOCF MeLi 3 3 A B Me-SiMe3 12 kA / kB = 2.4 x 10 Duhamel et al. J. Org. Chem. 1996, 61, 2232 H H SiMe Me b-Silicon Effect 3 vs. Me3C H Me3C H - Silicon stabalizes b-carbocations H OCOCF3 H OCOCF3 - Stabalization is a result of hyperconjugation 4 kA / kB = 4 x 10 SiR3 CR3 Evidence for Stepwise mechanism vs. Me3Si SiMe2Ph SiMe2Ph A B Me3Si SiMe2Ph *A is more stable than B by 38 kcal/mol * Me3Si SiMe2Ph Me3Si Jorgensen, JACS, 1986,107, 1496 Product ratios are equal from either starting material suggesting common intermediate cation Baran Lab Properties of Silicon Hafensteiner Evidence for Rapid Nucleophilic Attack Extraordinary Metallation Me SiMe3 SiMe3 Li Si t-BuLi Me SnCl4 Cl Me3Si Cl Me2Si Cl SiMe MeO OMe 3 OMe OMe Me2Si Cl vs. -

19.13 the Wittig Alkene Synthesis 933
19_BRCLoudon_pgs5-0.qxd 12/9/08 11:41 AM Page 933 19.13 THE WITTIG ALKENE SYNTHESIS 933 PROBLEMS 19.32 Draw the structures of all aldehydes or ketones that could in principle give the following product after application of either the Wolff–Kishner or Clemmensen reduction. H3C CH2CH(CH3)2 L L 19.33 Outline a synthesis of 1,4-dimethoxy-2-propylbenzene from hydroquinone (p-hydroxyphenol) and any other reagents. 19.13 THE WITTIG ALKENE SYNTHESIS Our tour through aldehyde and ketone chemistry started with simple additions; then addition followed by substitution (acetal formation); then additions followed by elimination (imine and enamine formation). Another addition–elimination reaction, called the Wittig alkene synthe- sis, is an important method for preparing alkenes from aldehydes and ketones. An example of the Wittig alkene synthesis is the preparation of methylenecyclohexane from cyclohexanone. 1 1 A | A | O CH_ 2 PPh3 CH2 Ph3P O _ (19.70) 1 + 3 anL ylid + L 1 3 triphenylphosphine cyclohexanone methylenecyclohexane oxide The Wittig synthesis is especially important because it gives alkenes in which the position of the double bond is unambiguous; in other words, the Wittig synthesis is completely regios- elective.Itcanbeusedforthepreparationofalkenesthatwouldbedifficulttoprepareby other reactions. For example, methylenecyclohexane, which is readily prepared by the Wittig synthesis (Eq. 19.70), cannot be prepared by dehydration of 1-methylcyclohexanol; 1- methylcyclohexene is obtained instead, because alcohol dehydration gives the alkene iso- mer(s) in which the double bond has the greatest number of alkyl substituents (Sec. 10.1). OH " H2SO4 " CH3 H2O CH3 L + 1-methylcyclohexene H2SO4 (19.71) A CH2 (little or none formed) methylenecyclohexane The nucleophile in the Wittig alkene synthesis is a type of ylid. -

Syllabus CHEM 6352 2014
CHEM 6352 Organic Reactions & Synthesis Fall 2014 Jeremy A. May Office: 5025 SERC Office hours: T/Th 10-11 am or by appointment (email me) Email: [email protected] Website: http://mynsm.uh.edu/groups/maygroup/wiki/b24dc/Classes.html Lectures: 154 Fleming Tuesdays and Thursdays 8:30–10:00. August 26–December 6, 2014. Homework Session Saturdays 3:00 pm to 5:30 pm in Fleming 154/160/162. No class November 27–29, 2014 (Thanksgiving recess); Oct. 31st is last day to withdraw Optional Texts (on reserve at MD Anderson Library) Zweifel, G.; Nantz, M. “Modern Organic Synthesis: An Introduction” March, J. “Advanced Organic Chemistry” Corey, E. J.; Cheng, X.-M. “The Logic of Chemical Synthesis” Warren, S. “Designing Organic Syntheses: A Programmed Introduction to the Synthon Approach” Kürti, L.; Czakó, B. “Strategic Applications of Named Reactions in Organic Synthesis” Grossman, R. “The Art of Writing Reasonable Organic Reaction Mechanisms” Model Sets: Students are strongly encouraged to purchase at least one set. HGS biochemistry molecular model sets are recommended and are available at Research Stores in the Old Science Building. Other relevant texts and references: Greene; Wuts. “Protective Groups in Organic Synthesis” Nicolaou, K.C.; Sorensen, E. “Classics in Total Synthesis” Nicolaou, K.C.; Snyder, S. “Classics in Total Synthesis II” Larock, R. C. "Comprehensive Organic Transformations" Hartwig, J. “Organotransition Metal Chemistry: From Bonding to Catalysis” Tsuji, J. “Palladium Reagents and Catalysts” Hegedus, L. “Transition Metals in the Synthesis of Complex Organic Molecules” Problem Sets: Problem Sets will be distributed on Tuesdays (or before) and are due by the next Saturday at the Homework Session. -
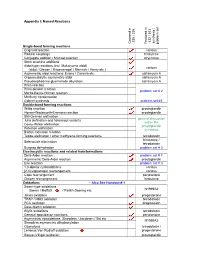
Appendix I: Named Reactions Single-Bond Forming Reactions Co
Appendix I: Named Reactions 235 / 335 432 / 533 synthesis / / synthesis Covered in Covered Featured in problem set problem Single-bond forming reactions Grignard reaction various Radical couplings hirstutene Conjugate addition / Michael reaction strychnine Stork enamine additions Aldol-type reactions (incl. Mukaiyama aldol) various (aldol / Claisen / Knoevenagel / Mannich / Henry etc.) Asymmetric aldol reactions: Evans / Carreira etc. saframycin A Organocatalytic asymmetric aldol saframycin A Pseudoephedrine glycinamide alkylation saframycin A Prins reaction Prins-pinacol reaction problem set # 2 Morita-Baylis-Hillman reaction McMurry condensation Gabriel synthesis problem set #3 Double-bond forming reactions Wittig reaction prostaglandin Horner-Wadsworth-Emmons reaction prostaglandin Still-Gennari olefination general discussion Julia olefination and heteroaryl variants within the Corey-Winter olefination prostaglandin Peterson olefination synthesis Barton extrusion reaction Tebbe olefination / other methylene-forming reactions tetrodotoxin hirstutene / Selenoxide elimination tetrodotoxin Burgess dehydration problem set # 3 Electrocyclic reactions and related transformations Diels-Alder reaction problem set # 1 Asymmetric Diels-Alder reaction prostaglandin Ene reaction problem set # 3 1,3-dipolar cycloadditions various [2,3] sigmatropic rearrangement various Cope rearrangement periplanone Claisen rearrangement hirstutene Oxidations – Also See Handout # 1 Swern-type oxidations (Swern / Moffatt / Parikh-Doering etc. N1999A2 Jones oxidation -

Elimination Reactions Are Described
Introduction In this module, different types of elimination reactions are described. From a practical standpoint, elimination reactions widely used for the generation of double and triple bonds in compounds from a saturated precursor molecule. The presence of a good leaving group is a prerequisite in most elimination reactions. Traditional classification of elimination reactions, in terms of the molecularity of the reaction is employed. How the changes in the nature of the substrate as well as reaction conditions affect the mechanism of elimination are subsequently discussed. The stereochemical requirements for elimination in a given substrate and its consequence in the product stereochemistry is emphasized. ELIMINATION REACTIONS Objective and Outline beta-eliminations E1, E2 and E1cB mechanisms Stereochemical considerations of these reactions Examples of E1, E2 and E1cB reactions Alpha eliminations and generation of carbene I. Basics Elimination reactions involve the loss of fragments or groups from a molecule to generate multiple bonds. A generalized equation is shown below for 1,2-elimination wherein the X and Y from two adjacent carbon atoms are removed, elimination C C C C -XY X Y Three major types of elimination reactions are: α-elimination: two atoms or groups are removed from the same atom. It is also known as 1,1-elimination. H R R C X C + HX R Both H and X are removed from carbon atom here R Carbene β-elimination: loss of atoms or groups on adjacent atoms. It is also H H known as 1,2- elimination. R C C R R HC CH R X H γ-elimination: loss of atoms or groups from the 1st and 3rd positions as shown below. -

Organic & Biomolecular Chemistry
Organic & Biomolecular Chemistry View Article Online PAPER View Journal | View Issue Diastereoselective synthesis of trisubstituted olefins using a silicon-tether ring-closing Cite this: Org. Biomol. Chem., 2020, 18, 2297 metathesis strategy† Stéphane Wittmann,‡ Alexander F. Tiniakos and Joëlle Prunet * The diastereoselective synthesis of trisubstituted olefins with concomitant C–C bond formation is still adifficult challenge, and olefin metathesis reactions for the formation of such alkenes are usually not high yielding or/and diastereoselective. Herein we report an efficient and diastereoselective synthesis of trisubstituted olefins flanked by an allylic alcohol, by a silicon-tether ring-closing metathesis strategy. Both E-andZ-trisubstituted alkenes were synthesised, depending on the method employed Received 30th November 2019, to cleave the silicon tether. Furthermore, this methodology features a novel Peterson olefination for Accepted 4th March 2020 the synthesis of allyldimethylsilanes. These versatile intermediates were also converted into the DOI: 10.1039/c9ob02563d corresponding allylchlorodimethylsilanes, which are not easily accessible in high yields by other Creative Commons Attribution 3.0 Unported Licence. rsc.li/obc methods. Introduction However, there are much fewer examples of RCM reactions with O–Si–C tethers. The reported formations of trisubstituted The trisubstituted E olefin motif with a methyl substituent is olefins from allyl silanes lead to bicyclic products,8 and the present in numerous polyketide natural products. Among others focus on vinyl silanes.9 We propose a strategy for an these, callipeltoside A1 and dolabelide C2 possess another efficient and diastereoselective synthesis of trisubstituted This article is licensed under a common feature: an allylic alkoxy group on the lone substitu- olefins flanked by an allylic alcohol, which involves a RCM ent of the trisusbstituted olefin (highlighted in red in reaction with a O–Si–C tether. -

Studies in Directed Catalytic Asymmetric Hydroboration of 1,2-Disubstituted Unsaturated Amide
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Student Research Projects, Dissertations, and Chemistry, Department of Theses - Chemistry Department Summer 7-2017 STUDIES IN DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2-DISUBSTITUTED UNSATURATED AMIDE Shuyang Zhang University of Nebraska - Lincoln, [email protected] Follow this and additional works at: http://digitalcommons.unl.edu/chemistrydiss Part of the Organic Chemistry Commons Zhang, Shuyang, "STUDIES IN DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2-DISUBSTITUTED UNSATURATED AMIDE" (2017). Student Research Projects, Dissertations, and Theses - Chemistry Department. 85. http://digitalcommons.unl.edu/chemistrydiss/85 This Article is brought to you for free and open access by the Chemistry, Department of at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Student Research Projects, Dissertations, and Theses - Chemistry Department by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. STUDIES IN DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2- DISUBSTITUTED UNSATURATED AMIDE by Shuyang Zhang A THESIS Presented to the Faculty of The Graduate College at the University of Nebraska In Partial Fulfillment of Requirements For the Degree of Master of Science Major: Chemistry Under the Supervision of Professor James M. Takacs Lincoln, Nebraska July 2017 DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2- DISUBSTITUTED ALKENES Shuyang Zhang, M.S. University of Nebraska 2017 Advisor: Professor James M. Takacs The Rh-catalyzed, substrate directed catalytic asymmetric hydroboration of γ,δ-unsaturated amides provides a direct route to enantioenriched acyclic secondary γ- borylated carbonyl derivatives with high regio- and enantioselectivity. The catalytic condition optimization and substrate scope study is discussed, including the effects in catalytical asymmetric hydroboration on pre-installed chiral γ,δ-unsaturated amides. -

The Wittig Reaction
UC Berkeley College of Chemistry Chemistry 112B Organic Chemistry The Wittig Reaction Author: Jonathan Melville Graduate Student Instructor: Rebecca Triano March 13, 2014 1 Introduction The Wittig reaction, discovered in 1954 by Georg Wittig, is one of the most common tech- niques used for the stereoselective preparation of alkenes. Broadly speaking, the reaction allows for the formation of an alkene product and a triphenylphosphine oxide side product from the reaction of an aldehyde or ketone and a \Wittig reagent" (a triphenylphospho- nium ylide). In our experiment, we formed methyl (2E)-3-(2-nitrophenyl)acrylate and triphenylphosphine oxide from the reaction of 2-nitrobenzaldehyde and methyl (triph- enylphosphoranylidene) acetate. The key step of the mechanism of the ylide reaction is the nucleophilic addition of the ylide to the electrophilic carbonyl group, forming a 4-membered ring that dissociates into the product molecules. The stereoselectivity of the reaction is predicated on the stability of triphenylphosphonium ylide, which determines which of two ring intermediates form: the sterically-unfavored cis intermediate that forms via a fast yet reversible process, or the slow, irreversible trans intermediate. Because ylides contain (by definition) adjacent positive and negative charges (a positive on the phosphonium, and a negative on the carbon adjacent to the residue), R groups that can better stabilize the adjacent negative charge produce more stable ylides. We can thus classify three distinct levels of stabiliza- tion of the ylide by its residue: neutral substituents that do not stabilize negative charge (like alkyl groups or hydrogens) produce an unstabilized ylide that preferentially forms cis product, whereas electron-withdrawing residues (like nitro or ester groups) stabilize the charge and preferentially form trans product, while \semi-stabilized" substituents (generally aromatic rings) produce a mixture of the two.