Positron Annihilation Localization by Nanoscale Magnetization Yaser H
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
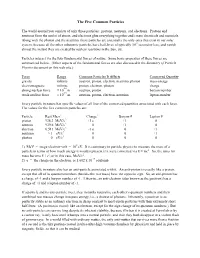
The Five Common Particles
The Five Common Particles The world around you consists of only three particles: protons, neutrons, and electrons. Protons and neutrons form the nuclei of atoms, and electrons glue everything together and create chemicals and materials. Along with the photon and the neutrino, these particles are essentially the only ones that exist in our solar system, because all the other subatomic particles have half-lives of typically 10-9 second or less, and vanish almost the instant they are created by nuclear reactions in the Sun, etc. Particles interact via the four fundamental forces of nature. Some basic properties of these forces are summarized below. (Other aspects of the fundamental forces are also discussed in the Summary of Particle Physics document on this web site.) Force Range Common Particles It Affects Conserved Quantity gravity infinite neutron, proton, electron, neutrino, photon mass-energy electromagnetic infinite proton, electron, photon charge -14 strong nuclear force ≈ 10 m neutron, proton baryon number -15 weak nuclear force ≈ 10 m neutron, proton, electron, neutrino lepton number Every particle in nature has specific values of all four of the conserved quantities associated with each force. The values for the five common particles are: Particle Rest Mass1 Charge2 Baryon # Lepton # proton 938.3 MeV/c2 +1 e +1 0 neutron 939.6 MeV/c2 0 +1 0 electron 0.511 MeV/c2 -1 e 0 +1 neutrino ≈ 1 eV/c2 0 0 +1 photon 0 eV/c2 0 0 0 1) MeV = mega-electron-volt = 106 eV. It is customary in particle physics to measure the mass of a particle in terms of how much energy it would represent if it were converted via E = mc2. -

1.1. Introduction the Phenomenon of Positron Annihilation Spectroscopy
PRINCIPLES OF POSITRON ANNIHILATION Chapter-1 __________________________________________________________________________________________ 1.1. Introduction The phenomenon of positron annihilation spectroscopy (PAS) has been utilized as nuclear method to probe a variety of material properties as well as to research problems in solid state physics. The field of solid state investigation with positrons started in the early fifties, when it was recognized that information could be obtained about the properties of solids by studying the annihilation of a positron and an electron as given by Dumond et al. [1] and Bendetti and Roichings [2]. In particular, the discovery of the interaction of positrons with defects in crystal solids by Mckenize et al. [3] has given a strong impetus to a further elaboration of the PAS. Currently, PAS is amongst the best nuclear methods, and its most recent developments are documented in the proceedings of the latest positron annihilation conferences [4-8]. PAS is successfully applied for the investigation of electron characteristics and defect structures present in materials, magnetic structures of solids, plastic deformation at low and high temperature, and phase transformations in alloys, semiconductors, polymers, porous material, etc. Its applications extend from advanced problems of solid state physics and materials science to industrial use. It is also widely used in chemistry, biology, and medicine (e.g. locating tumors). As the process of measurement does not mostly influence the properties of the investigated sample, PAS is a non-destructive testing approach that allows the subsequent study of a sample by other methods. As experimental equipment for many applications, PAS is commercially produced and is relatively cheap, thus, increasingly more research laboratories are using PAS for basic research, diagnostics of machine parts working in hard conditions, and for characterization of high-tech materials. -

Chapter 3 the Fundamentals of Nuclear Physics Outline Natural
Outline Chapter 3 The Fundamentals of Nuclear • Terms: activity, half life, average life • Nuclear disintegration schemes Physics • Parent-daughter relationships Radiation Dosimetry I • Activation of isotopes Text: H.E Johns and J.R. Cunningham, The physics of radiology, 4th ed. http://www.utoledo.edu/med/depts/radther Natural radioactivity Activity • Activity – number of disintegrations per unit time; • Particles inside a nucleus are in constant motion; directly proportional to the number of atoms can escape if acquire enough energy present • Most lighter atoms with Z<82 (lead) have at least N Average one stable isotope t / ta A N N0e lifetime • All atoms with Z > 82 are radioactive and t disintegrate until a stable isotope is formed ta= 1.44 th • Artificial radioactivity: nucleus can be made A N e0.693t / th A 2t / th unstable upon bombardment with neutrons, high 0 0 Half-life energy protons, etc. • Units: Bq = 1/s, Ci=3.7x 1010 Bq Activity Activity Emitted radiation 1 Example 1 Example 1A • A prostate implant has a half-life of 17 days. • A prostate implant has a half-life of 17 days. If the What percent of the dose is delivered in the first initial dose rate is 10cGy/h, what is the total dose day? N N delivered? t /th t 2 or e Dtotal D0tavg N0 N0 A. 0.5 A. 9 0.693t 0.693t B. 2 t /th 1/17 t 2 2 0.96 B. 29 D D e th dt D h e th C. 4 total 0 0 0.693 0.693t /th 0.6931/17 C. -

The W and Z at LEP
50YEARSOFCERN The W and Z at LEP 1954-2004 The Large Electron Positron collider made significant contributions to the process of establishing the Standard Model as the basis for matter and 5 forces, and also built a platform for physics scenarios beyond the model. The Standard Model of particle physics is arguably one of the greatest achievements in physics in the 20th century. Within this framework the electroweak interactions, as introduced by Sheldon Glashow, Abdus Salam and Steven Weinberg, are formulated as an SU(2)xll(l) gauge field theory with the masses of the fundamental particles generated by the Higgs mechanism. Both of the first two crucial steps in establishing experimentally the electroweak part of the Standard Model occurred at CERN. These were the discovery of neutral currents in neutrino scattering by the Gargamelle collab oration in 1973, and only a decade later the discovery by the UA1 and UA2 collaborations of the W and Z gauge bosons in proton- antiproton collisions at the converted Super Proton Synchrotron (CERN Courier May 2003 p26 and April 2004 pl3). Establishing the theory at the quantum level was the next logical step, following the pioneering theoretical work of Gerard 't Hooft Fig. 1. The cover page and Martinus Veltman. Such experimental proof is a necessary (left) of the seminal requirement for a theory describing phenomena in the microscopic CERN yellow report world. At the same time, performing experimental analyses with high 76-18 on the physics precision also opens windows to new physics phenomena at much potential of a 200 GeV higher energy scales, which can be accessed indirectly through e+e~ collider, and the virtual effects. -

A Relativistic One Pion Exchange Model of Proton-Neutron Electron-Positron Pair Production
Utah State University DigitalCommons@USU All Graduate Theses and Dissertations Graduate Studies 5-1973 A Relativistic One Pion Exchange Model of Proton-Neutron Electron-Positron Pair Production William A. Peterson Utah State University Follow this and additional works at: https://digitalcommons.usu.edu/etd Part of the Physics Commons Recommended Citation Peterson, William A., "A Relativistic One Pion Exchange Model of Proton-Neutron Electron-Positron Pair Production" (1973). All Graduate Theses and Dissertations. 3674. https://digitalcommons.usu.edu/etd/3674 This Dissertation is brought to you for free and open access by the Graduate Studies at DigitalCommons@USU. It has been accepted for inclusion in All Graduate Theses and Dissertations by an authorized administrator of DigitalCommons@USU. For more information, please contact [email protected]. ii ACKNOWLEDGMENTS The author sincerely appreciates the advice and encouragement of Dr. Jack E. Chatelain who suggested this problem and guided its progress. The author would also like to extend his deepest gratitude to Dr. V. G. Lind and Dr. Ackele y Miller for their support during the years of graduate school. William A. Pete r son iii TABLE OF CONTENTS Page ACKNOWLEDGMENTS • ii LIST OF TABLES • v LIST OF FIGURES. vi ABSTRACT. ix Chapter I. INTRODUCTION. • . • • II. FORMULATION OF THE PROBLEM. 7 Green's Function Treatment of the Dirac Equation. • • • . • . • • • 7 Application of Feynman Graph Rules to Pair Production in Neutron- Proton Collisions 11 III. DIFFERENTIAL CROSS SECTION. 29 Symmetric Coplanar Case 29 Frequency Distributions 44 BIBLIOGRAPHY. 72 APPENDIXES •• 75 Appendix A. Notation and Definitions 76 Appendix B. Expressi on for Cross Section. 79 Appendix C. -

Neutrino Masses-How to Add Them to the Standard Model
he Oscillating Neutrino The Oscillating Neutrino of spatial coordinates) has the property of interchanging the two states eR and eL. Neutrino Masses What about the neutrino? The right-handed neutrino has never been observed, How to add them to the Standard Model and it is not known whether that particle state and the left-handed antineutrino c exist. In the Standard Model, the field ne , which would create those states, is not Stuart Raby and Richard Slansky included. Instead, the neutrino is associated with only two types of ripples (particle states) and is defined by a single field ne: n annihilates a left-handed electron neutrino n or creates a right-handed he Standard Model includes a set of particles—the quarks and leptons e eL electron antineutrino n . —and their interactions. The quarks and leptons are spin-1/2 particles, or weR fermions. They fall into three families that differ only in the masses of the T The left-handed electron neutrino has fermion number N = +1, and the right- member particles. The origin of those masses is one of the greatest unsolved handed electron antineutrino has fermion number N = 21. This description of the mysteries of particle physics. The greatest success of the Standard Model is the neutrino is not invariant under the parity operation. Parity interchanges left-handed description of the forces of nature in terms of local symmetries. The three families and right-handed particles, but we just said that, in the Standard Model, the right- of quarks and leptons transform identically under these local symmetries, and thus handed neutrino does not exist. -

Can Any Anti-Particle Make a Probe?
NEWS AND VIEWS Can any anti-particle make a probe? Positron probes (of the solid state) are already a booming business and may soon be applied to the study of liquids. Can anti-proton probes be far away? EACH new particle of matter discovered from zero to some maximum. So what is new? Two things, both in the may most immediately be of interest to The trick of making a mono-energetic same issue of Physical Review Letters (2 those with theories to check (or invent, as beam of reasonable but still low energy September). First, E. Gramsch, KG.Lynn, the case may be), but one thing is certain: usually entails some interaction with a J. Throwe and I Kanazawa, from the somebody will eventually use the particle solid within which the energy of incident Brookhaven National Laboratory, have as a probe for some purpose or another. positrons is first reduced to the average extended the use of positrons as probes And that is likely to be true even if the thermal energy, whereupon some propor from solids to liquids, and with surprising newly discovered particle is inherently tion of the incident particles is afterwards results (67, 1,282; 1991). The incentive unstable, or if it interacts with and is de- re-emitted by scattering, but with an en has been to see whether positron probes stroyed by some component of the envi- ergy that is a measure of the work function can throw light on the structure of liquids, ronment in which it finds itself. What - the potential difference between the for which purpose they have studied matters is merely that the particle should interior and the exteriorofthe solid. -

HIGH ENERGY ASTROPHYSICS - Lecture 10
HIGH ENERGY ASTROPHYSICS - Lecture 10 PD Frank Rieger ITA & MPIK Heidelberg Wednesday 1 Pair Plasmas in Astrophysics 1 Overview • Pair Production γ + γ ! e+ + e− (threshold) • Compactness parameter • Example I: "internal" γγ-absorption in AGN (Mkn 421) • Example II: EBL & limited transparency of the Universe to TeV photons • Pair Annihilation e+ + e− ! γ + γ (no threshold) • Example: Annihilation-in-flight (energetics) 2 2 Two-Photon Pair Production Generation of e+e−-pairs in environments with very high radiation energy density: γ + γ ! e+ + e− • Reaction threshold for pair production (see lecture 9): 2 2 e1e2 − p1p2 cos θ = m1m2c + δm c (m1 + m2 + 0:5 δm) With mi = 0; ei := i=c = pi (photons) and δm = 2me: 2 4 2 4 12(1 − cos θ) = 0:5 (δm) c = 2mec 2 4 ) 12 = mec for producing e+e− at rest in head-on collision (lab frame angle cos θ = −1). 12 • Example: for TeV photon 1 = 10 eV, interaction possible with soft photons 2 2 ≥ (0:511) eV = 0:26 eV (infrared photons). 3 • Cross-section for pair-production: πr2 1 + β∗ σ (β∗) = 0 (1 − β∗2) 2β∗(β∗2 − 2) + (3 − β∗4) ln γγ 2 1 − β∗ with β∗ = v∗=c velocity of electron (positron) in centre of momentum frame, 2 and πr0 = 3σT =8. In terms of photon energies and collision angle θ (cf. above): ∗2 ∗ ∗ 2 2 ∗2 −1=2 12(1 − cos θ) = 2Ee and Ee = γ mec = mec (1 − β ) ∗ where Ee =total energy of electron (positron) in CoM frame, so 1 (1 − cos θ) = 2γ∗2m2c4 = 2m2c4 1 2 e e (1 − β∗2) s 2m2c4 ) β∗ = 1 − e 12(1 − cos θ) 4 1 + - "+" --> e + e T ! / 0.1 "" ! 0.01 1 10 100 #1 #2 (1-cos$) Figure 1: Cross-section for γγ-pair production in units of the Thomson cross-section σT as a function of 2 2 interacting photon energies (1=mec )(2=mec ) (1−cos θ). -

Collisions at Very High Energy
” . SLAC-PUB-4601 . April 1988 P-/E) - Theory of e+e- Collisions at Very High Energy MICHAEL E. PESKIN* - - - Stanford Linear Accelerator Center Stanford University, Stanford, California 94309 - Lectures presented at the SLAC Summer Institute on Particle Physics Stanford, California, August 10 - August 21, 1987 z-- -. - - * Work supported by the Department of Energy, contract DE-AC03-76SF00515. 1. Introduction The past fifteen years of high-energy physics have seen the successful elucida- - tion of the strong, weak, and electromagnetic interactions and the explanation of all of these forces in terms of the gauge theories of the standard model. We are now beginning the last stage of this chapter in physics, the era of direct experi- - mentation on the weak vector bosons. Experiments at the CERN @ collider have _ isolated the W and 2 bosons and confirmed the standard model expectations for their masses. By the end of the decade, the new colliders SLC and LEP will have carried out precision measurements of the properties of the 2 boson, and we have good reason to hope that this will complete the experimental underpinning of the structure of the weak interactions. Of course, the fact that we have answered some important questions about the working of Nature does not mean that we have exhausted our questions. Far from r it! Every advance in fundamental physics brings with it new puzzles. And every advance sets deeper in relief those very mysterious issues, such as the origin of the mass of electron, which have puzzled generations of physicists and still seem out of - reach of our understanding. -

The Quark Model and Deep Inelastic Scattering
The quark model and deep inelastic scattering Contents 1 Introduction 2 1.1 Pions . 2 1.2 Baryon number conservation . 3 1.3 Delta baryons . 3 2 Linear Accelerators 4 3 Symmetries 5 3.1 Baryons . 5 3.2 Mesons . 6 3.3 Quark flow diagrams . 7 3.4 Strangeness . 8 3.5 Pseudoscalar octet . 9 3.6 Baryon octet . 9 4 Colour 10 5 Heavier quarks 13 6 Charmonium 14 7 Hadron decays 16 Appendices 18 .A Isospin x 18 .B Discovery of the Omega x 19 1 The quark model and deep inelastic scattering 1 Symmetry, patterns and substructure The proton and the neutron have rather similar masses. They are distinguished from 2 one another by at least their different electromagnetic interactions, since the proton mp = 938:3 MeV=c is charged, while the neutron is electrically neutral, but they have identical properties 2 mn = 939:6 MeV=c under the strong interaction. This provokes the question as to whether the proton and neutron might have some sort of common substructure. The substructure hypothesis can be investigated by searching for other similar patterns of multiplets of particles. There exists a zoo of other strongly-interacting particles. Exotic particles are ob- served coming from the upper atmosphere in cosmic rays. They can also be created in the labortatory, provided that we can create beams of sufficient energy. The Quark Model allows us to apply a classification to those many strongly interacting states, and to understand the constituents from which they are made. 1.1 Pions The lightest strongly interacting particles are the pions (π). -

The Lamb Shift in Classical Terms Jean Louis Van Belle, Drs, Maec, Baec, Bphil 2 April 2020 Email: [email protected] Summary
Explaining the Lamb shift in classical terms Jean Louis Van Belle, Drs, MAEc, BAEc, BPhil 2 April 2020 Email: [email protected] Summary Any realist model of an atom must explain its properties in terms of its parts: the electrons and protons. We, therefore, need a realist model of an electron and a proton. Such model must explain their properties, including their mass, radius and magnetic moment – and the anomaly therein, of course. Indeed, these properties are not to be thought of as mysterious intrinsic properties of a pointlike or dimensionless particle: the model should generate them. We think our ring current model does that rather convincingly. In this paper, we take the next logical step. We relate these models to the four quantum numbers that define electron orbitals. In the process, we also offer the basics of a classical explanation of the Lamb shift. This should complete our realist interpretation of quantum physics. Contents Introduction: the Lamb shift and proton spin flips ....................................................................................... 1 Electron states, orbitals and energy levels ................................................................................................... 3 Spin and orbital angular momentum of protons and electrons ................................................................... 6 Was there a need for a new theory? .......................................................................................................... 10 The four quantum numbers for electron -

D MESON PRODUCTION in E+E ANNIHILATION* Petros-Afentoulis Rapidis Stanford Linear Accelerator Center Stanford University Stanfor
SLAC-220 UC-34d (E) D MESON PRODUCTION IN e+e ANNIHILATION* Petros-Afentoulis Rapidis Stanford Linear Accelerator Center Stanford University Stanford, California 94305 June 1979 Prepared for the Department of Energy under contract number DE-AC03-76SF00515 Printed in the United States of America . Available from National Technical Information Service, U .S . Department of Commerce, 5285 Port Royal Road, Springfield, VA 22161 . Price : Printed Copy $6 .50 ; Microfiche $3 .00 . * Ph .D . dissertation . ABSTRACT The production of D mesons in e+e annihilation for the center-of- mass energy range 3 .7 to 7 .0 GeV has been studied with the MARK :I magnetic detector at the Stanford Positron Electron Accelerating Rings facility . We observed a resonance in the total cross-section for hadron production in e+e annihilation at an energy just above the threshold for charm production. This resonance,which we name I", has a mass of 3772 ± 6 MeV/c 2 , a total width of 28 ± 5 MeV/c 2 , a partial width to electron pairs of 345 ± 85 eV/c 2 , and decays almost exclusively into DD pairs . The •" provides a rich source of background-free and kinematically well defined D mesons for study . From the study of D mesons produced in the decay of the *" we have determined the masses of the D ° and D+ mesons to be 1863 .3 ± 0 .9 MeV/c 2 and 1868 .3 ± 0 .9 MeV/c 2 respectively . We also determined the branching fractions for D ° decay to K-7r+ , K°ir+e and K 1r+ur ir+ to be (2 .2 ± 0 .6)%, (4 .0 ± 1 .3)%, and (3 .2 ± 1 .1)% and the r+i+ + branching fractions for D + decay to K°7+ and K to be (1 .5 0 .6)1 and (3 .9 '~ 1 .0)% .