Neemmc Guidelines for Tablet Crushing and Administration Via Enteral Feeding Tubes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
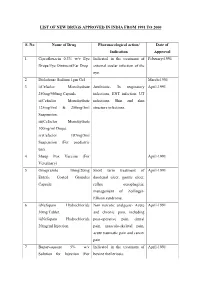
List of New Drugs Approved in India from 1991 to 2000
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

SPIRONOLACTONE Spironolactone – Oral (Common Brand Name
SPIRONOLACTONE Spironolactone – oral (common brand name: Aldactone) Uses: Spironolactone is used to treat high blood pressure. Lowering high blood pressure helps prevent strokes, heart attacks, and kidney problems. It is also used to treat swelling (edema) caused by certain conditions (e.g., congestive heart failure) by removing excess fluid and improving symptoms such as breathing problems. This medication is also used to treat low potassium levels and conditions in which the body is making too much of a natural chemical (aldosterone). Spironolactone is known as a “water pill” (potassium-sparing diuretic). Other uses: This medication has also been used to treat acne in women, female pattern hair loss, and excessive hair growth (hirsutism), especially in women with polycystic ovary disease. Side effects: Drowsiness, lightheadedness, stomach upset, diarrhea, nausea, vomiting, or headache may occur. To minimize lightheadedness, get up slowly when rising from a seated or lying position. If any of these effects persist or worsen, notify your doctor promptly. Tell your doctor immediately if any of these unlikely but serious side effects occur; dizziness, increased thirst, change in the amount of urine, mental/mood chances, unusual fatigue/weakness, muscle spasms, menstrual period changes, sexual function problems. This medication may lead to high levels of potassium, especially in patients with kidney problems. If not treated, very high potassium levels can be fatal. Tell your doctor immediately if you notice any of the following unlikely but serious side effects: slow/irregular heartbeat, muscle weakness. Precautions: Before taking spironolactone, tell your doctor or pharmacist if you are allergic to it; or if you have any other allergies. -

Fosavance, INN-Alendronic Acid and Colecalciferol
EMA/175858/2015 EMEA/H/C/000619 EPAR summary for the public Fosavance alendronic acid and colecalciferol This is a summary of the European public assessment report (EPAR) for Fosavance. It explains how the Committee for Medicinal Products for Human Use (CHMP) assessed the medicine to reach its opinion in favour of granting a marketing authorisation and its recommendations on the conditions of use for Fosavance. What is Fosavance? Fosavance is a medicine that contains two active substances: alendronic acid and colecalciferol (vitamin D3). It is available as tablets (70 mg alendronic acid and 2,800 international units [IU] colecalciferol; 70 mg alendronic acid and 5,600 IU colecalciferol). What is Fosavance used for? Fosavance (containing either 2,800 or 5,600 IU colecalciferol) is used to treat osteoporosis (a disease that makes bones fragile) in women who have been through the menopause and are at risk of low vitamin D levels. Fosavance 70 mg/5,600 IU is for use in patients who are not taking vitamin D supplements. Fosavance reduces the risk of fractures (broken bones) in the spine and the hip. The medicine can only be obtained with a prescription. How is Fosavance used? The recommended dose of Fosavance is one tablet once a week. It is intended for long-term use. The patient must take the tablet with a full glass of water (but not mineral water), at least 30 minutes before any food, drink or other medicines (including antacids, calcium supplements and vitamins). To avoid irritation of the oesophagus (the tube that leads from the mouth to the stomach), the patient should not lie down until after their first food of the day, which should be at least 30 minutes after taking the tablet. -

Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and Its Application
Infect Dis Ther (2017) 6:57–67 DOI 10.1007/s40121-016-0144-8 REVIEW Use of Ceftaroline Fosamil in Children: Review of Current Knowledge and its Application Juwon Yim . Leah M. Molloy . Jason G. Newland Received: November 10, 2016 / Published online: December 30, 2016 Ó The Author(s) 2016. This article is published with open access at Springerlink.com ABSTRACT infections, CABP caused by penicillin- and ceftriaxone-resistant S. pneumoniae and Ceftaroline is a novel cephalosporin recently resistant Gram-positive infections that fail approved in children for treatment of acute first-line antimicrobial agents. However, bacterial skin and soft tissue infections and limited data are available on tolerability in community-acquired bacterial pneumonia neonates and infants younger than 2 months (CABP) caused by methicillin-resistant of age, and on pharmacokinetic characteristics Staphylococcus aureus, Streptococcus pneumoniae in children with chronic medical conditions and other susceptible bacteria. With a favorable and those with invasive, complicated tolerability profile and efficacy proven in infections. In this review, the microbiological pediatric patients and excellent in vitro profile of ceftaroline, its mechanism of action, activity against resistant Gram-positive and and pharmacokinetic profile will be presented. Gram-negative bacteria, ceftaroline may serve Additionally, clinical evidence for use in as a therapeutic option for polymicrobial pediatric patients and proposed place in therapy is discussed. Enhanced content To view enhanced content for this article go to http://www.medengine.com/Redeem/ 1F47F0601BB3F2DD. Keywords: Antibiotic resistance; Ceftaroline J. Yim (&) fosamil; Children; Methicillin-resistant St. John Hospital and Medical Center, Detroit, MI, Staphylococcus aureus; Streptococcus pneumoniae USA e-mail: [email protected] L. -

Adrovance, INN-Alendronic Acid
EMA/194587/2011 EMEA/H/C/000759 EPAR summary for the public Adrovance alendronic acid / colecalciferol This is a summary of the European public assessment report (EPAR) for Adrovance. It explains how the Committee for Medicinal Products for Human Use (CHMP) assessed the medicine to reach its opinion in favour of granting a marketing authorisation and its recommendations on the conditions of use for Adrovance. What is Adrovance? Adrovance is a medicine that contains two active substances: alendronic acid and colecalciferol (vitamin D3). It is available as white tablets (capsule-shaped: 70 mg alendronic acid and 2,800 international units [IU] colecalciferol; rectangular: 70 mg alendronic acid and 5,600 IU colecalciferol). What is Adrovance used for? Adrovance (containing either 2,800 or 5,600 IU colecalciferol) is used to treat osteoporosis (a disease that makes bones fragile) in women who have been through the menopause and are at risk of low vitamin D levels. Adrovance 70 mg/5,600 IU is for use in patients who are not taking vitamin D supplements. Adrovance reduces the risk of broken bones in the spine and the hip. The medicine can only be obtained with a prescription. How is Adrovance used? The recommended dose of Adrovance is one tablet once a week. It is intended for long-term use. The patient must take the tablet with a full glass of water (but not mineral water), at least 30 minutes before any food, drink or other medicines (including antacids, calcium supplements and vitamins). To avoid irritation of the oesophagus (gullet), the patient should not lie down until after their first food of the day, which should be at least 30 minutes after taking the tablet. -

Appendix A: Potentially Inappropriate Prescriptions (Pips) for Older People (Modified from ‘STOPP/START 2’ O’Mahony Et Al 2014)
Appendix A: Potentially Inappropriate Prescriptions (PIPs) for older people (modified from ‘STOPP/START 2’ O’Mahony et al 2014) Consider holding (or deprescribing - consult with patient): 1. Any drug prescribed without an evidence-based clinical indication 2. Any drug prescribed beyond the recommended duration, where well-defined 3. Any duplicate drug class (optimise monotherapy) Avoid hazardous combinations e.g.: 1. The Triple Whammy: NSAID + ACE/ARB + diuretic in all ≥ 65 year olds (NHS Scotland 2015) 2. Sick Day Rules drugs: Metformin or ACEi/ARB or a diuretic or NSAID in ≥ 65 year olds presenting with dehydration and/or acute kidney injury (AKI) (NHS Scotland 2015) 3. Anticholinergic Burden (ACB): Any additional medicine with anticholinergic properties when already on an Anticholinergic/antimuscarinic (listed overleaf) in > 65 year olds (risk of falls, increased anticholinergic toxicity: confusion, agitation, acute glaucoma, urinary retention, constipation). The following are known to contribute to the ACB: Amantadine Antidepressants, tricyclic: Amitriptyline, Clomipramine, Dosulepin, Doxepin, Imipramine, Nortriptyline, Trimipramine and SSRIs: Fluoxetine, Paroxetine Antihistamines, first generation (sedating): Clemastine, Chlorphenamine, Cyproheptadine, Diphenhydramine/-hydrinate, Hydroxyzine, Promethazine; also Cetirizine, Loratidine Antipsychotics: especially Clozapine, Fluphenazine, Haloperidol, Olanzepine, and phenothiazines e.g. Prochlorperazine, Trifluoperazine Baclofen Carbamazepine Disopyramide Loperamide Oxcarbazepine Pethidine -

CASODEX (Bicalutamide)
HIGHLIGHTS OF PRESCRIBING INFORMATION • Gynecomastia and breast pain have been reported during treatment with These highlights do not include all the information needed to use CASODEX 150 mg when used as a single agent. (5.3) CASODEX® safely and effectively. See full prescribing information for • CASODEX is used in combination with an LHRH agonist. LHRH CASODEX. agonists have been shown to cause a reduction in glucose tolerance in CASODEX® (bicalutamide) tablet, for oral use males. Consideration should be given to monitoring blood glucose in Initial U.S. Approval: 1995 patients receiving CASODEX in combination with LHRH agonists. (5.4) -------------------------- RECENT MAJOR CHANGES -------------------------- • Monitoring Prostate Specific Antigen (PSA) is recommended. Evaluate Warnings and Precautions (5.2) 10/2017 for clinical progression if PSA increases. (5.5) --------------------------- INDICATIONS AND USAGE -------------------------- ------------------------------ ADVERSE REACTIONS ----------------------------- • CASODEX 50 mg is an androgen receptor inhibitor indicated for use in Adverse reactions that occurred in more than 10% of patients receiving combination therapy with a luteinizing hormone-releasing hormone CASODEX plus an LHRH-A were: hot flashes, pain (including general, back, (LHRH) analog for the treatment of Stage D2 metastatic carcinoma of pelvic and abdominal), asthenia, constipation, infection, nausea, peripheral the prostate. (1) edema, dyspnea, diarrhea, hematuria, nocturia, and anemia. (6.1) • CASODEX 150 mg daily is not approved for use alone or with other treatments. (1) To report SUSPECTED ADVERSE REACTIONS, contact AstraZeneca Pharmaceuticals LP at 1-800-236-9933 or FDA at 1-800-FDA-1088 or ---------------------- DOSAGE AND ADMINISTRATION ---------------------- www.fda.gov/medwatch The recommended dose for CASODEX therapy in combination with an LHRH analog is one 50 mg tablet once daily (morning or evening). -

Crohn's & Colitis Program, Patient Information Guide
Inflammatory Bowel Disease Program Patient Information Guide Page # Contents ..........................................................................................................................................1 Welcome! Welcome Letter ....................................................................................................................3 Important Things to Know Up Front ....................................................................................4 Meet Your Crohn’s & Colitis Team .....................................................................................6 How to Contact Us Crohn’s & Colitis Clinic Schedule .....................................................................................11 Physician and Nurse Contact Information..........................................................................12 Appointment Scheduling and Other Contact Information..................................................13 The Basics of Inflammatory Bowel Disease (IBD) Basic Information about Inflammatory Bowel Disease (IBD) ...........................................14 Frequently Asked Questions about Inflammatory Bowel Disease (IBD) ..........................18 Testing in IBD Colonoscopy and Flexible Sigmoidoscopy ........................................................................20 Upper Endoscopy ...............................................................................................................21 Capsule Endoscopy and Deep Enteroscopy .......................................................................22 -

COVID-19—The Potential Beneficial Therapeutic Effects of Spironolactone During SARS-Cov-2 Infection
pharmaceuticals Review COVID-19—The Potential Beneficial Therapeutic Effects of Spironolactone during SARS-CoV-2 Infection Katarzyna Kotfis 1,* , Kacper Lechowicz 1 , Sylwester Drozd˙ zal˙ 2 , Paulina Nied´zwiedzka-Rystwej 3 , Tomasz K. Wojdacz 4, Ewelina Grywalska 5 , Jowita Biernawska 6, Magda Wi´sniewska 7 and Miłosz Parczewski 8 1 Department of Anesthesiology, Intensive Therapy and Acute Intoxications, Pomeranian Medical University in Szczecin, 70-111 Szczecin, Poland; [email protected] 2 Department of Pharmacokinetics and Monitored Therapy, Pomeranian Medical University, 70-111 Szczecin, Poland; [email protected] 3 Institute of Biology, University of Szczecin, 71-412 Szczecin, Poland; [email protected] 4 Independent Clinical Epigenetics Laboratory, Pomeranian Medical University, 71-252 Szczecin, Poland; [email protected] 5 Department of Clinical Immunology and Immunotherapy, Medical University of Lublin, 20-093 Lublin, Poland; [email protected] 6 Department of Anesthesiology and Intensive Therapy, Pomeranian Medical University in Szczecin, 71-252 Szczecin, Poland; [email protected] 7 Clinical Department of Nephrology, Transplantology and Internal Medicine, Pomeranian Medical University, 70-111 Szczecin, Poland; [email protected] 8 Department of Infectious, Tropical Diseases and Immune Deficiency, Pomeranian Medical University in Szczecin, 71-455 Szczecin, Poland; [email protected] * Correspondence: katarzyna.kotfi[email protected]; Tel.: +48-91-466-11-44 Abstract: In March 2020, coronavirus disease 2019 (COVID-19) caused by SARS-CoV-2 was declared Citation: Kotfis, K.; Lechowicz, K.; a global pandemic by the World Health Organization (WHO). The clinical course of the disease is Drozd˙ zal,˙ S.; Nied´zwiedzka-Rystwej, unpredictable but may lead to severe acute respiratory infection (SARI) and pneumonia leading to P.; Wojdacz, T.K.; Grywalska, E.; acute respiratory distress syndrome (ARDS). -

Medication: Amitriptyline 10 Mg
Amitriptyline (Elavil) COMPLEX CHRONIC DISEASES PROGRAM Medication Handout Date: May 15, 2018 Medication: Amitriptyline 10 mg What is Amitriptyline? Amitriptyline belongs to a group of medications called tricyclic antidepressants (TCAs) that were first used to treat depression. It works by altering the levels of certain neuro- transmitters in the brain such as noradrenalin and serotonin. They have since been found to be effective for many different uses such as: pain, helping with sleep quality (but is not a sleeping pill), irritable bowel syndrome (with diarrhea), migraine prevention, and interstitial cystitis. Expected Benefit: Usually takes several weeks to notice a benefit You may not notice a benefit until you get to a dose of 25 mg Watch for possible side effects: This list of side effects is important for you to be aware of; however, it is also important to remember that not all side effects happen to all people. Many of these less serious side effects will improve over the first few days of taking the medications. If you have problems with these side effects talk with your doctor or pharmacist: Dry mouth – this is the most common side; the others are much less frequent Hangover effect – too sleepy in the morning Blurred vision Urinary retention, trouble with urination Tiredness, dizziness that is more than usual Diarrhea or constipation Stopping the medication: Do NOT stop taking this medication suddenly without asking your doctor – this medication is usually decreased slowly (at higher doses) before it is stopped completely How to use this medication: Take this medication with or without food Dosing Schedule: Start with 5 mg (½ tablet) 2 hrs before bed Increase dose according to table below Many patients can only tolerate 20 or 30 mg If you don’t have dry mouth or side effects, you can continue slowly increasing the dose to a maximum of 70 mg You can stay at a lower dose (stop increasing) if you get side effects (usually dry mouth). -

Adverse Drug Reactions Sample Chapter
Sample copyright Pharmaceutical Press www.pharmpress.com 5 Drug-induced skin reactions Anne Lee and John Thomson Introduction Cutaneous drug eruptions are one of the most common types of adverse reaction to drug therapy, with an overall incidence rate of 2–3% in hos- pitalised patients.1–3 Almost any medicine can induce skin reactions, and certain drug classes, such as non-steroidal anti-inflammatory drugs (NSAIDs), antibiotics and antiepileptics, have drug eruption rates approaching 1–5%.4 Although most drug-related skin eruptions are not serious, some are severe and potentially life-threatening. Serious reac- tions include angio-oedema, erythroderma, Stevens–Johnson syndrome and toxic epidermal necrolysis. Drug eruptions can also occur as part of a spectrum of multiorgan involvement, for example in drug-induced sys- temic lupus erythematosus (see Chapter 11). As with other types of drug reaction, the pathogenesis of these eruptions may be either immunological or non-immunological. Healthcare professionals should carefully evalu- ate all drug-associated rashes. It is important that skin reactions are identified and documented in the patient record so that their recurrence can be avoided. This chapter describes common, serious and distinctive cutaneous reactions (excluding contact dermatitis, which may be due to any external irritant, including drugs and excipients), with guidance on diagnosis and management. A cutaneous drug reaction should be suspected in any patient who develops a rash during a course of drug therapy. The reaction may be due to any medicine the patient is currently taking or has recently been exposed to, including prescribed and over-the-counter medicines, herbal or homoeopathic preparations, vaccines or contrast media. -

Inflammatory Bowel Disease Agents
Therapeutic Class Overview Inflammatory Bowel Disease Agents INTRODUCTION • Inflammatory bowel disease (IBD) is a spectrum of chronic idiopathic inflammatory intestinal conditions that cause gastrointestinal symptoms including diarrhea, abdominal pain, bleeding, fatigue, and weight loss. The exact cause of IBD is unknown; however, proposed etiologies involve a combination of infectious, genetic, and lifestyle factors (Bernstein et al 2015, Peppercorn 2019[a], Peppercorn 2020[c]). • Complications of IBD include hemorrhage, rectal fissures, fistulas, peri-rectal and intra-abdominal abscesses, and colon cancer. Possible extra-intestinal complications include hepatobiliary complications, anemia, arthritis and arthralgias, uveitis, skin lesions, and mood and anxiety disorders (Bernstein et al 2015). • Ulcerative colitis (UC) and Crohn’s disease (CD) are 2 forms of IBD that differ in pathophysiology and presentation; as a result of these differences, the approach to the treatment of each condition often differs (Peppercorn 2019[a]). • UC is characterized by recurrent episodes of inflammation of the mucosal layer of the colon. The inflammation, limited to the mucosa, commonly involves the rectum and may extend in a proximal and continuous fashion to affect other parts of the colon. The hallmark clinical symptom is an inflamed rectum with symptoms of urgency, bleeding, and tenesmus (Peppercorn 2020[c], Rubin et al 2019). • CD can involve any part of the gastrointestinal tract and is characterized by transmural inflammation and “skip areas.” Transmural inflammation may lead to fibrosis, strictures, sinus tracts, and fistulae (Peppercorn 2019[b]). • The immune system is known to play a critical role in the underlying pathogenesis of IBD. It is suggested that abnormal responses of both innate and adaptive immunity mechanisms induce aberrant intestinal tract inflammation in IBD patients (Geremia et al 2014).